Scalable Manufacturing of Brivaracetam Intermediates via Novel Pd-Catalyzed Hydrogenation
Scalable Manufacturing of Brivaracetam Intermediates via Novel Pd-Catalyzed Hydrogenation
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing complex active pharmaceutical ingredients (APIs), particularly for anti-epileptic drugs like Brivaracetam. A significant breakthrough in this domain is documented in Chinese patent CN108658831B, which details a robust and high-yielding process for producing 2-oxo-1-pyrrolidine derivatives. This technology addresses critical bottlenecks in the existing supply chain by introducing a streamlined catalytic hydrogenation step that eliminates the need for expensive chiral ligands while maintaining exceptional stereocontrol. For procurement leaders and R&D directors, this represents a pivotal shift towards more cost-effective and reliable manufacturing protocols for key epilepsy treatment intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the levorotatory product of Brivaracetam has been plagued by inefficiencies and high operational costs. As noted in the background of the patent, earlier methodologies such as those disclosed in international publication WO01/62726 relied on hydrogenation reactions using ammonium formate as a hydrogen donor in the presence of palladium on charcoal. These legacy processes suffered from critically low yields, often hovering around merely 50 percent, which is economically unsustainable for large-scale commercial production. Furthermore, alternative approaches described in patents like CN104892483A necessitated the use of sophisticated and costly chiral catalytic ligands to induce the necessary stereoselectivity. These harsh reaction conditions and expensive reagent requirements created significant barriers to entry, limiting the ability of manufacturers to produce high-purity intermediates at a competitive price point.
The Novel Approach
The innovative process outlined in CN108658831B fundamentally reimagines the reduction step by utilizing a simple yet highly effective Palladium-based catalyst system, specifically favoring Palladium Chloride (PdCl2). Instead of relying on transfer hydrogenation with ammonium formate, this method employs molecular hydrogen directly, which not only simplifies the reaction workup but also dramatically improves atom economy. By leveraging the chirality already present in the starting amino acid precursor, the reaction achieves high diastereoselectivity without the burden of additional chiral auxiliaries. This approach transforms a previously low-yielding bottleneck into a high-efficiency step, capable of delivering molar yields exceeding 90 percent under mild conditions, thereby setting a new standard for industrial feasibility.
Mechanistic Insights into Pd-Catalyzed Stereoselective Hydrogenation
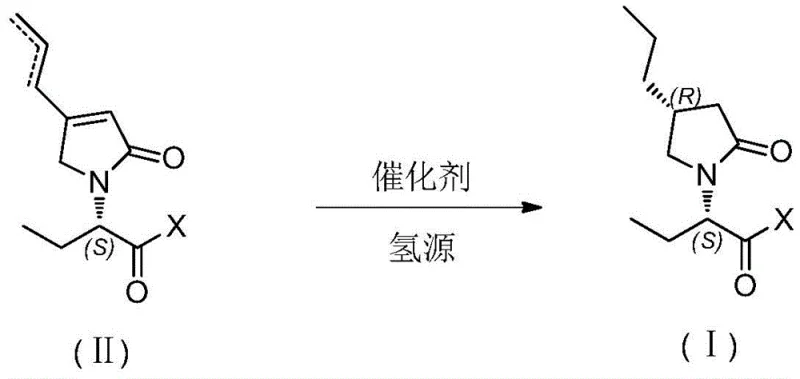
The core of this technological advancement lies in the precise mechanistic control exerted by the Palladium catalyst during the hydrogenation of the unsaturated pyrrolidone ring. The reaction proceeds through a heterogeneous or homogeneous catalytic cycle where molecular hydrogen is activated on the metal center and subsequently transferred to the olefinic bond of the substrate. Crucially, the stereochemical outcome is dictated by the existing chiral center at the C2 position, derived from the (S)-2-aminobutanoic acid moiety. This proximal chirality exerts a powerful directing effect, shielding one face of the double bond and forcing the hydrogen addition to occur from the opposite face, thus establishing the desired (4R) configuration with high fidelity.
Impurity control is inherently built into this mechanism due to the specificity of the catalyst and the mild reaction parameters. Unlike harsher reduction methods that might lead to over-reduction or racemization of the sensitive amide and lactam functionalities, the PdCl2 system operates selectively on the carbon-carbon double bond. The patent data indicates a diastereomeric ratio (d:e) of up to 98:2, demonstrating that the formation of the unwanted (4S) epimer is effectively suppressed. This high level of stereochemical purity minimizes the need for downstream purification steps such as recrystallization or chromatography, which are often major contributors to yield loss and processing time in pharmaceutical manufacturing.
How to Synthesize Brivaracetam Intermediate Efficiently
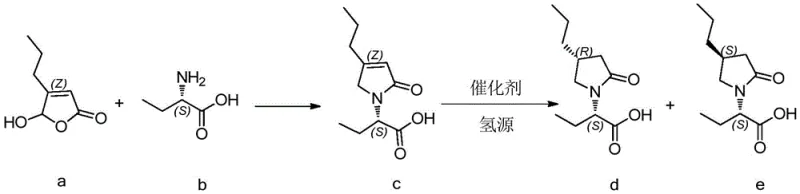
The synthesis begins with the condensation of (Z)-5-propylfuran-2(5H)-one and (S)-2-aminobutanoic acid to form the unsaturated intermediate, followed by the critical catalytic hydrogenation step. The process is designed to be operationally simple, utilizing common solvents like methanol and standard laboratory equipment, making it highly adaptable for both pilot and commercial scales. The following guide outlines the standardized procedure derived from the patent examples to ensure optimal yield and purity.
- Condense (Z)-5-propylfuran-2(5H)-one with (S)-2-aminobutanoic acid in methanol using triethylamine as a base.
- Perform catalytic hydrogenation on the resulting unsaturated intermediate using Palladium Chloride (PdCl2) and hydrogen gas.
- Isolate the final product through filtration, concentration, and pulping with n-hexane to achieve high diastereomeric purity.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain managers and procurement officers, the adoption of this patented process offers tangible strategic benefits that extend beyond mere technical metrics. By shifting away from complex chiral ligand systems and low-yield transfer hydrogenation, manufacturers can significantly reduce the cost of goods sold (COGS) associated with Brivaracetam production. The simplification of the reaction workflow reduces the dependency on specialized reagents that are subject to volatile market pricing and supply constraints, thereby enhancing the overall resilience of the supply chain against external disruptions.
- Cost Reduction in Manufacturing: The elimination of expensive chiral ligands represents a direct and substantial saving in raw material costs. Additionally, the dramatic improvement in reaction yield means that less starting material is required to produce the same amount of final product, effectively lowering the unit cost of the API intermediate. The use of molecular hydrogen, a commodity chemical, further replaces costlier hydrogen donors, contributing to a leaner and more economical production model that maximizes return on investment.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent output quality, which is critical for maintaining uninterrupted supply to downstream drug formulation partners. The mild reaction conditions reduce the risk of batch failures due to thermal runaway or sensitivity issues, leading to more predictable production schedules. This reliability allows procurement teams to negotiate better terms and secure long-term contracts with confidence, knowing that the manufacturing process is stable and scalable.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process aligns well with green chemistry principles by minimizing waste generation through higher efficiency. The avoidance of toxic or hazardous reagents simplifies waste treatment protocols and reduces the regulatory burden associated with chemical disposal. This makes the scale-up from kilogram to multi-ton production smoother and more compliant with increasingly stringent global environmental regulations, facilitating faster time-to-market for new generic or branded formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and specifications provided in the patent documentation to assist decision-makers in evaluating the technology's fit for their specific manufacturing needs.
Q: What is the primary advantage of the PdCl2 catalytic system over previous methods?
A: The primary advantage is the elimination of expensive chiral ligands. Previous methods required complex chiral catalysts to induce stereoselectivity, whereas this process relies on the inherent chirality of the starting amino acid and a simple, cost-effective Palladium Chloride catalyst to achieve high diastereoselectivity.
Q: How does this process improve yield compared to the prior art described in WO01/62726?
A: The prior art utilizing ammonium formate and palladium on charcoal reported yields as low as 50%. In contrast, the disclosed method utilizing molecular hydrogen and PdCl2 achieves molar yields exceeding 90%, drastically reducing material waste and production costs.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability. It operates under mild conditions (20-30°C), uses common solvents like methanol, and avoids hazardous or difficult-to-handle reagents, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brivaracetam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic processes like the one described in CN108658831B for the global epilepsy medication market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of high-quality intermediates. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Brivaracetam intermediate meets the highest international standards for safety and efficacy.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge technology for their product pipelines. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that optimize both your development timeline and commercial profitability.
