Revolutionizing 3-Trifluoromethyl-1,2,4-Triazole Production: A Cost-Effective DMF-Based Route for Commercial Scale-Up
Introduction to Patent CN114920707B: A Breakthrough in Heterocyclic Chemistry
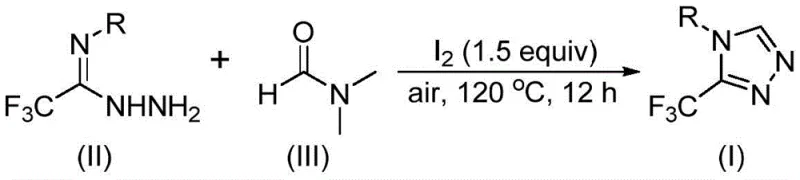
The pharmaceutical and agrochemical industries are constantly seeking more efficient routes to construct privileged scaffolds, particularly fluorinated heterocycles which are ubiquitous in modern drug design. Patent CN114920707B, published in April 2023, introduces a transformative preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology stands out by utilizing the common organic solvent N,N-dimethylformamide (DMF) not merely as a medium, but as a critical carbon source for the ring construction. For R&D directors and process chemists, this represents a paradigm shift from complex, multi-step sequences to a streamlined, one-pot tandem cyclization. The method operates under remarkably mild constraints regarding atmosphere, proceeding efficiently in air rather than requiring stringent inert gas protection, which significantly lowers the barrier for industrial adoption. By leveraging molecular iodine as a promoter, this route achieves high reaction efficiency while maintaining a broad substrate scope, allowing for the synthesis of diverse derivatives essential for SAR studies and commercial API production.
The strategic value of this patent lies in its ability to simplify the supply chain for high-purity pharmaceutical intermediates. Traditional syntheses of trifluoromethyl-triazoles often involve hazardous reagents, expensive catalysts, or sensitive conditions that complicate scale-up. In contrast, the disclosed method utilizes readily available starting materials—trifluoroethyliminohydrazide and DMF—to generate the core structure with impressive reliability. This innovation directly addresses the pain points of procurement managers looking for cost reduction in API manufacturing, as it eliminates the need for specialized C1 building blocks that are often costly and difficult to handle. Furthermore, the robustness of the reaction conditions suggests a high degree of reproducibility, a critical factor for supply chain heads concerned with continuity and quality control in large-scale production environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,2,4-triazole rings, especially those bearing trifluoromethyl groups, has been fraught with synthetic challenges that hinder efficient commercialization. Conventional methodologies frequently rely on the condensation of hydrazines with nitriles or amidines, often requiring harsh thermal conditions or strong acidic/basic environments that can degrade sensitive functional groups. A significant bottleneck in these traditional routes is the necessity for anhydrous and oxygen-free conditions, which demands specialized equipment such as gloveboxes or Schlenk lines, thereby inflating capital expenditure and operational complexity. Moreover, many existing protocols utilize stoichiometric amounts of expensive coupling reagents or transition metal catalysts that leave behind toxic residues, necessitating rigorous and costly purification steps to meet pharmaceutical purity standards. The reliance on specific, often unstable C1 synthons further complicates the logistics, as these reagents may have short shelf lives or require cold chain storage, introducing fragility into the supply chain.
The Novel Approach
The methodology described in CN114920707B elegantly circumvents these obstacles by repurposing DMF, a ubiquitous solvent, into a reactive synthon. This dual-functionality approach means that the solvent itself participates in the bond-forming events, effectively reducing the number of distinct reagents required and simplifying the reaction mixture. The use of molecular iodine as a promoter is particularly advantageous; it is inexpensive, easy to handle, and effective at activating the DMF molecule for cyclization without the need for precious metals. Crucially, the reaction tolerates air and moisture to a significant extent, operating successfully at temperatures between 110°C and 130°C without the need for inert gas blanketing. This operational simplicity translates directly into lower processing costs and reduced safety risks. The versatility of this approach is evidenced by its compatibility with a wide range of substituents on the aromatic ring, enabling the rapid generation of diverse libraries of triazole derivatives for drug discovery programs without the need to re-optimize conditions for each new substrate.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for process optimization and impurity control. The reaction proceeds through a sophisticated tandem sequence where molecular iodine plays a pivotal role in activating the DMF molecule. Mechanistically, the pathway can diverge based on which part of the DMF molecule acts as the carbon source. In one plausible pathway, the formyl group of DMF undergoes condensation with the trifluoroethyliminohydrazide to form a hydrazone intermediate. This is followed by an intramolecular cyclization that eliminates dimethylamine, yielding the desired 3-trifluoromethyl-1,2,4-triazole core. Alternatively, the N-methyl group of DMF can serve as the carbon source. In this scenario, iodine reacts with DMF to generate an reactive amine salt species, which then undergoes nucleophilic addition with the hydrazide. Subsequent elimination of N-methylformamide generates an azadiene intermediate, which then undergoes intramolecular nucleophilic attack and oxidative aromatization to finalize the triazole ring system.
This mechanistic flexibility highlights the robustness of the chemistry, as it can accommodate slight variations in reaction conditions while still driving towards the thermodynamic product. For quality control teams, understanding these pathways is essential for identifying potential byproducts, such as unreacted hydrazides or over-oxidized species, although the patent data indicates high selectivity. The oxidative aromatization step, facilitated by the iodine/air system, ensures the formation of the stable aromatic triazole ring, which is a key driver for the high yields observed across different substrates. The ability of the system to function under air suggests that oxygen may also play a role as a terminal oxidant in the catalytic cycle, regenerating the active iodine species and sustaining the reaction turnover. This intricate interplay between the solvent, promoter, and atmosphere underscores the sophistication of what appears to be a simple mixing procedure, offering a reliable platform for the synthesis of complex heterocycles.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory scale-up and pilot plant production. The protocol involves combining the trifluoroethyliminohydrazide precursor with molecular iodine in DMF, followed by heating. The detailed standardized synthesis steps, including precise stoichiometry and workup procedures, are outlined below to ensure reproducibility and safety.
- Charge a reaction vessel with molecular iodine (1.5 equiv), trifluoroethyliminohydrazide substrate, and DMF solvent.
- Heat the mixture to 110-130°C under air atmosphere and stir for 10-15 hours to facilitate the tandem cyclization.
- Upon completion, perform standard workup including filtration, silica gel treatment, and column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this DMF-based synthesis route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The primary driver for cost reduction is the elimination of exotic reagents; by using DMF as both solvent and reactant, the bill of materials is significantly simplified. This consolidation reduces the number of SKUs that need to be sourced, stored, and managed, leading to substantial administrative and inventory holding cost savings. Furthermore, the removal of the requirement for anhydrous and anaerobic conditions means that standard stainless steel reactors can be used without the need for specialized nitrogen purging systems or dry rooms, drastically lowering the capital intensity of the manufacturing process. The use of molecular iodine, a commodity chemical, instead of expensive transition metal catalysts like palladium or rhodium, further drives down the raw material cost per kilogram of product.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the high atom economy derived from using the solvent as a reagent. Traditional methods often generate significant waste from stoichiometric coupling agents, whereas this route produces volatile byproducts like dimethylamine or N-methylformamide which are easier to separate. The avoidance of precious metal catalysts eliminates the need for costly metal scavenging steps and the associated validation testing for residual metals, which is a major expense in API production. Additionally, the energy profile is favorable; running the reaction at 110-130°C is achievable with standard heating media like steam or thermal oil, avoiding the need for cryogenic cooling or extreme high-pressure equipment.
- Enhanced Supply Chain Reliability: Supply chain resilience is improved by the reliance on globally available commodity chemicals. DMF and iodine are produced in massive volumes worldwide, ensuring that supply disruptions are unlikely to impact production schedules. The robustness of the reaction to air and moisture means that raw materials do not require specialized packaging or transport conditions, reducing logistics costs and the risk of degradation during transit. This stability allows for larger batch sizes to be run with confidence, improving throughput and ensuring consistent delivery timelines to downstream customers. The broad substrate tolerance also means that a single manufacturing line can be easily adapted to produce various analogues, providing flexibility to respond to changing market demands without extensive retooling.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method aligns well with green chemistry principles. The reduction in reagent count minimizes the volume of chemical waste generated per unit of product. While DMF itself requires careful handling due to reproductive toxicity, its use as a reactant means it is consumed, potentially reducing the total solvent load compared to processes where it is used solely as a medium in large excess. The absence of heavy metal catalysts simplifies wastewater treatment and effluent management, lowering the cost of environmental compliance. The process is inherently scalable, as the exothermic profile is manageable and the reaction does not rely on sensitive intermediates that might decompose upon scale-up, ensuring a smooth transition from gram-scale R&D to ton-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within patent CN114920707B, providing a factual basis for decision-making.
Q: What is the primary advantage of using DMF in this triazole synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (providing either the formyl or methyl group), which drastically reduces raw material costs and simplifies the reaction setup compared to traditional methods requiring separate C1 synthons.
Q: Does this process require strict anhydrous or oxygen-free conditions?
A: No, one of the key innovations of patent CN114920707B is that the reaction proceeds efficiently under air atmosphere at 110-130°C, eliminating the need for expensive inert gas protection and rigorous drying protocols.
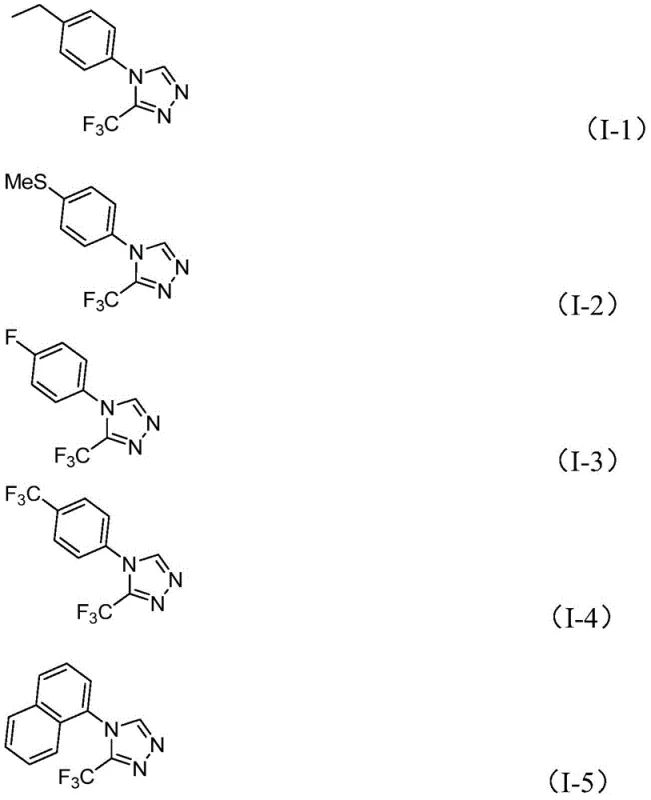
Q: What is the substrate scope for the R group in this synthesis?
A: The method demonstrates broad compatibility with various substituted aryl groups, including those with electron-donating (methyl, methoxy) and electron-withdrawing (fluoro, chloro, trifluoromethyl) substituents, as well as naphthyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN114920707B and is fully equipped to leverage this iodine-promoted DMF cyclization for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory curiosity to industrial reality is seamless. Our state-of-the-art facilities are designed to handle halogenated chemistry and high-temperature reactions safely, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards.
We invite you to collaborate with us to optimize this pathway for your specific API candidates. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to request specific COA data for our triazole intermediates and to discuss route feasibility assessments that can accelerate your development timeline and reduce your overall cost of goods sold.
