Advanced Synthesis of Benzodioxane-Thiazole Hybrids for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of Benzodioxane-Thiazole Hybrids for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry is constantly seeking novel scaffolds that offer improved bioavailability and targeted therapeutic effects, and the technology disclosed in patent CN103724340A represents a significant advancement in this domain. This patent details the preparation of a unique class of thiazole derivatives containing a benzo-oxygen-containing heterocyclic structure, specifically merging the pharmacologically privileged 1,4-benzodioxane motif with the versatile thiazole ring. The strategic combination of these two distinct heterocyclic systems creates a molecular architecture that leverages the known anti-inflammatory and antiviral properties of benzodioxanes while incorporating the broad-spectrum biological activity associated with thiazoles. For R&D directors and procurement specialists, understanding the synthetic accessibility and structural versatility of these compounds is crucial for developing next-generation active pharmaceutical ingredients (APIs). The general structure of these derivatives, characterized by variable alkylene bridges and diverse aromatic substituents, provides a robust platform for medicinal chemistry optimization.
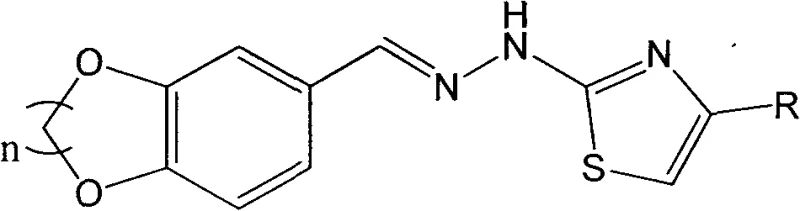
From a commercial perspective, the ability to access such complex heterocyclic systems through a streamlined, scalable process is a major value driver. The patent outlines a methodology that avoids the use of exotic reagents or hazardous conditions, relying instead on fundamental organic transformations that are well-suited for industrial scale-up. This approach not only ensures a reliable supply chain for high-purity pharmaceutical intermediates but also significantly reduces the environmental footprint associated with traditional multi-step syntheses. By focusing on reactions that yield solid products amenable to simple filtration and recrystallization, the process minimizes solvent waste and energy consumption. Consequently, this technology offers a compelling solution for cost reduction in API manufacturing, addressing the critical need for efficient, sustainable production methods in the competitive global pharmaceutical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing complex heterocyclic hybrids often suffer from significant drawbacks that hinder their commercial viability and practical application in drug discovery. Many conventional routes rely heavily on transition metal catalysis, which introduces challenges related to residual metal contamination, a critical quality attribute for pharmaceutical intermediates that requires expensive and time-consuming purification steps to meet regulatory standards. Furthermore, older synthetic strategies frequently involve harsh reaction conditions, such as extreme temperatures or strong acidic environments, which can lead to poor selectivity, decomposition of sensitive functional groups, and lower overall yields. The reliance on chromatographic purification for product isolation in many legacy processes further exacerbates cost issues and limits scalability, making it difficult to transition from laboratory benchtop quantities to kilogram or ton-scale production required for clinical trials and commercial launch.
The Novel Approach
In contrast, the novel approach described in the patent utilizes a concise three-step sequence that circumvents these traditional bottlenecks through the use of mild, base-catalyzed conditions and readily available starting materials. The strategy employs simple inorganic bases like potassium carbonate or sodium hydroxide, effectively eliminating the risk of heavy metal contamination and simplifying the downstream processing workflow. A key innovation lies in the physical state of the intermediates and final products; the reactions are designed such that the desired compounds precipitate as solids, allowing for isolation via straightforward suction filtration rather than complex extraction or column chromatography. This operational simplicity translates directly into enhanced process robustness and reduced manufacturing costs, providing a distinct competitive advantage for suppliers aiming to deliver high-purity OLED material or pharmaceutical precursors with consistent quality and shorter lead times.
Mechanistic Insights into Base-Catalyzed Heterocyclic Construction
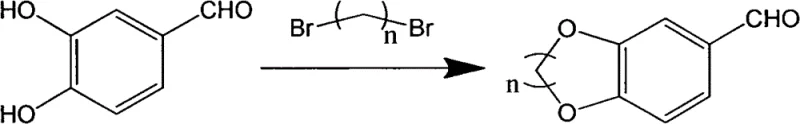
The synthetic pathway is underpinned by a series of well-defined mechanistic steps that ensure high regioselectivity and conversion efficiency. The initial phase involves the nucleophilic substitution of protocatechualdehyde with a dibromoalkane, facilitated by a base catalyst in a polar aprotic solvent such as acetonitrile or acetone. This step constructs the critical benzodioxane ring system, where the phenolic hydroxyl groups attack the terminal carbons of the dibromoalkane, closing the ring to form the aldehyde intermediate. The choice of base and solvent is optimized to balance reaction rate and solubility, ensuring complete conversion within a reasonable timeframe of approximately 5 hours at moderate temperatures around 80°C. This controlled environment prevents side reactions and ensures the formation of the desired cyclic ether structure with high fidelity.
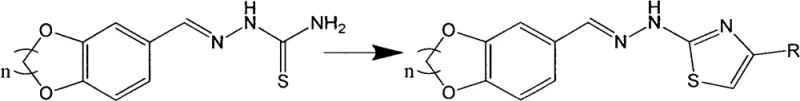
Following the formation of the benzodioxane scaffold, the synthesis proceeds through a condensation reaction with thiosemicarbazide to generate a hydrazone intermediate, which serves as the precursor for the final thiazole ring closure. The subsequent cyclization step involves the reaction of this hydrazone with a substituted bromoacetophenone, a classic Hantzsch-type thiazole synthesis variation. This transformation is driven by the nucleophilic attack of the sulfur atom on the alpha-carbon of the bromoketone, followed by intramolecular cyclization and dehydration to aromatize the thiazole ring. The use of ethanol or isopropanol as the solvent in these later stages facilitates the dissolution of reactants while allowing the less soluble product to crystallize out upon cooling, a feature that is instrumental in achieving high purity without extensive workup procedures.
Impurity control is inherently built into this mechanistic design through the exploitation of solubility differences and the specificity of the base-catalyzed reactions. By maintaining precise stoichiometric ratios, such as a 1:1.05 molar ratio of aldehyde to dibromoalkane, the process minimizes the formation of oligomeric byproducts or unreacted starting materials. The recrystallization steps, utilizing specific solvent mixtures like ethanol and acetone in a 10:1 volume ratio, further refine the product quality by selectively excluding impurities that remain in the mother liquor. This rigorous control over the chemical environment and physical isolation parameters ensures that the final thiazole derivatives meet stringent purity specifications, making them suitable for sensitive biological applications where trace impurities could alter pharmacokinetic profiles or toxicological outcomes.
How to Synthesize Benzodioxane-Thiazole Derivatives Efficiently
The synthesis of these valuable intermediates follows a logical progression that balances chemical efficiency with operational ease, making it an ideal candidate for technology transfer and commercial production. The process begins with the construction of the core heterocyclic framework, followed by functionalization to introduce the thiazole moiety, and concludes with purification to achieve the desired pharmaceutical grade. Each step has been optimized for yield and purity, utilizing standard laboratory equipment and common chemical reagents that are easily sourced from global supply chains. For detailed technical execution, the following guide outlines the standardized synthetic steps derived from the patent examples, providing a clear roadmap for replication and scale-up.
- React protocatechualdehyde with dibromoalkane in organic solvent with base catalyst to form the benzodioxane aldehyde intermediate.
- Condense the aldehyde intermediate with thiosemicarbazide in alcohol solvent to generate the hydrazone derivative.
- Cyclize the hydrazone with substituted bromoacetophenone in refluxing alcohol to yield the final thiazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical novelty, addressing critical pain points related to cost, reliability, and scalability. The elimination of expensive transition metal catalysts and the reliance on commodity chemicals like protocatechualdehyde and dibromoalkanes significantly lowers the raw material cost base, enabling more competitive pricing structures for the final intermediates. Moreover, the robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without compromising product quality, enhances supply chain reliability by reducing the risk of batch failures and production delays. This resilience is particularly valuable in a volatile global market where consistency of supply is paramount for maintaining uninterrupted drug development pipelines.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing inexpensive inorganic bases such as potassium carbonate and sodium hydroxide instead of costly organometallic catalysts, thereby removing the need for specialized metal scavenging steps. The isolation of products via simple filtration and recrystallization drastically reduces solvent consumption and waste disposal costs compared to chromatography-dependent methods. Additionally, the high atom economy of the cyclization reactions ensures that a greater proportion of raw materials are converted into the final product, minimizing waste generation and maximizing resource efficiency throughout the manufacturing lifecycle.
- Enhanced Supply Chain Reliability: The starting materials required for this synthesis, including protocatechualdehyde and various bromoacetophenones, are widely available commodity chemicals with established global supply networks, mitigating the risk of raw material shortages. The simplicity of the reaction setup, which does not require specialized high-pressure equipment or inert atmosphere conditions, allows for flexible production scheduling across multiple manufacturing sites. This flexibility ensures that production capacity can be rapidly scaled up to meet surging demand without the long lead times typically associated with commissioning complex specialized reactors.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, as demonstrated by the successful preparation of multiple derivatives with consistent yields and purity profiles across different substituent patterns. The use of common organic solvents like ethanol and acetone, which are easier to recover and recycle than chlorinated solvents, aligns with modern green chemistry principles and simplifies compliance with increasingly stringent environmental regulations. The generation of solid waste is minimized due to the high conversion rates, and the aqueous washes required for purification produce effluent streams that are easier to treat, supporting sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these benzodioxane-thiazole derivatives, based on the detailed specifications provided in the patent literature. Understanding these aspects is essential for partners looking to integrate these intermediates into their own drug discovery programs or manufacturing processes. The answers reflect the specific advantages of the disclosed methodology, highlighting its suitability for large-scale production and its potential to accelerate the development of new therapeutic agents.
Q: What are the key structural advantages of these thiazole derivatives?
A: These compounds combine the lipophilicity and low toxicity of the 1,4-benzodioxane scaffold with the biological activity of the thiazole ring, enhancing cell penetration and potential pharmacological effects.
Q: What catalysts are used in the synthesis process?
A: The process utilizes inexpensive inorganic bases such as potassium carbonate or sodium hydroxide, avoiding the need for expensive transition metal catalysts.
Q: How is the product isolated to ensure high purity?
A: The products typically precipitate as solids upon cooling or reaction completion, allowing for simple filtration and recrystallization without complex chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the benzodioxane-thiazole scaffold in modern medicinal chemistry and are committed to supporting our partners with high-quality intermediates produced via this advanced methodology. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of thiazole derivatives delivered meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this innovative synthetic route for your next drug development program. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can reduce your overall COGS. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with reliable, cost-effective, and high-purity chemical solutions.
