Advanced Iridium-Catalyzed C-H Activation for Scalable Sulfonamide Manufacturing
Advanced Iridium-Catalyzed C-H Activation for Scalable Sulfonamide Manufacturing
The landscape of fine chemical synthesis is undergoing a significant transformation driven by the need for more sustainable and efficient methodologies, particularly in the production of high-value scaffolds like sulfonamides. Patent CN107739322B introduces a groundbreaking approach that leverages transition metal catalysis to streamline the construction of these vital molecular architectures. This technology represents a paradigm shift from classical stoichiometric methods to catalytic C-H functionalization, offering a robust pathway for generating diverse sulfonamide libraries. For R&D directors and procurement specialists alike, understanding the nuances of this iridium-catalyzed protocol is essential for optimizing supply chains and reducing the environmental footprint of pharmaceutical and agrochemical intermediate manufacturing.
At the core of this innovation is the direct coupling of oxime compounds with phenylsulfonamides, facilitated by a pentamethylcyclopentadienyl iridium dichloride dimer catalyst system. Unlike traditional routes that often require pre-functionalized starting materials or harsh activating agents, this method utilizes the inherent reactivity of the C-H bond adjacent to the oxime directing group. The process operates under remarkably mild thermal conditions, typically around 60°C, which preserves sensitive functional groups and minimizes energy consumption. This technical advancement not only enhances the synthetic efficiency but also aligns with the growing global demand for greener chemistry solutions in the production of complex organic molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfonamide derivatives has relied heavily on the reaction between amines and sulfonyl chlorides, a process fraught with significant operational and safety challenges. Sulfonyl chlorides are notoriously moisture-sensitive and often require stringent anhydrous conditions, complicating storage and handling protocols in large-scale facilities. Furthermore, the generation of these activated species frequently involves the use of hazardous chlorinating agents such as thionyl chloride or phosphorus pentachloride, which produce corrosive byproducts and pose substantial environmental disposal issues. Alternative methods involving sulfonic acids often necessitate toxic activation reagents like trichloroacetonitrile or require expensive and difficult-to-source aryl triflates, limiting their practical applicability in cost-sensitive manufacturing environments.
Beyond safety concerns, conventional multi-step syntheses often suffer from poor atom economy and low overall yields due to the necessity of isolating unstable intermediates. The requirement for strong bases or extreme temperatures in some legacy protocols can lead to significant decomposition of sensitive substrates, resulting in complex impurity profiles that are difficult to purge. For supply chain managers, these inefficiencies translate into longer lead times, higher raw material costs, and increased waste treatment expenses. The inability to tolerate a broad range of functional groups without protection-deprotection sequences further restricts the structural diversity accessible through these traditional pathways, hindering rapid drug discovery and development cycles.
The Novel Approach
The methodology disclosed in CN107739322B circumvents these historical bottlenecks by employing a direct C-H amination strategy that merges an oxime derivative and a sulfonamide in a single reaction vessel. This one-pot protocol eliminates the need for isolating reactive intermediates, thereby drastically simplifying the operational workflow and reducing the potential for material loss during transfer steps. The use of a well-defined iridium catalyst, specifically [Cp*IrCl2]2, in conjunction with a silver oxidant like silver acetate, enables the selective activation of inert C-H bonds under neutral to mild conditions. This approach not only improves the overall yield, with reported efficiencies reaching upwards of 97%, but also significantly enhances the purity of the final product by minimizing side reactions associated with harsh reagents.
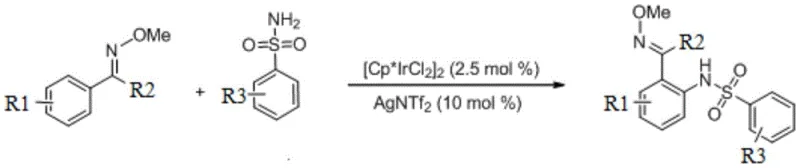
Furthermore, this novel synthetic route demonstrates exceptional substrate scope, accommodating a wide variety of electronic and steric environments on both the oxime and sulfonamide components. Substituents such as halogens, trifluoromethyl groups, and alkoxy moieties are well-tolerated, allowing for the rapid generation of diverse chemical libraries essential for structure-activity relationship studies. The reliance on commercially available and stable starting materials, combined with the use of dichloromethane as a standard organic solvent, ensures that this technology can be seamlessly integrated into existing manufacturing infrastructure. For procurement teams, this translates to a more reliable supply of high-quality intermediates with reduced dependency on specialized or hazardous reagents.
Mechanistic Insights into Ir-Catalyzed C-H Activation
The mechanistic underpinning of this transformation involves a sophisticated catalytic cycle initiated by the coordination of the oxime nitrogen to the iridium center, which serves as a directing group to facilitate proximal C-H bond cleavage. This cyclometalation step generates a stable five-membered iridacycle intermediate, positioning the metal center for subsequent nucleophilic attack or oxidative addition processes. The presence of the silver additive, AgNTf2, plays a critical role in abstracting the chloride ligands from the iridium precursor, generating a cationic active species that is more electrophilic and capable of engaging the sulfonamide nucleophile. This ligand exchange is crucial for maintaining the catalytic turnover and ensuring high reaction rates even at moderate temperatures.
Oxidative regeneration of the active catalyst is achieved through the action of silver acetate, which serves as the terminal oxidant in the cycle. This reagent facilitates the reductive elimination step that forms the new C-N bond, releasing the desired sulfonamide product and restoring the iridium catalyst to its active oxidation state. The choice of silver acetate over other oxidants is pivotal, as comparative data within the patent indicates that alternatives like PhI(OAc)2 or copper salts result in negligible yields or complete reaction failure. This specificity underscores the delicate balance of redox potentials required to drive the C-H activation forward without degrading the sensitive oxime functionality or the sulfonamide backbone.
How to Synthesize Sulfonamide Derivatives Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and minimize impurity formation. The standard protocol involves charging a reaction vessel with the iridium catalyst, silver additives, and substrates in dichloromethane, followed by heating to 60°C for a duration of 24 hours. This extended reaction time ensures complete conversion of the starting materials, particularly for sterically hindered substrates, while the moderate temperature prevents thermal degradation. Following the reaction, the crude mixture is subjected to standard purification techniques, typically column chromatography using silica gel, to isolate the pure sulfonamide product. Detailed standardized synthesis steps are provided in the guide below.
- Combine the iridium catalyst [Cp*IrCl2]2, silver additive AgNTf2, oxidant silver acetate, oxime substrate, and phenylsulfonamide in a reaction vessel with dichloromethane solvent.
- Heat the reaction mixture to 60°C and maintain stirring for 24 hours to ensure complete conversion via C-H activation.
- Upon completion, purify the crude sulfonamide product using column chromatography with silica gel and an ethyl acetate/petroleum ether eluent system.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iridium-catalyzed methodology offers substantial strategic benefits for organizations managing the supply of fine chemical intermediates. The elimination of hazardous chlorinating agents and the reduction of synthetic steps directly correlate to a significant decrease in operational risk and regulatory compliance burdens. By transitioning to a one-pot process, manufacturers can reduce the physical footprint required for production, lower energy consumption associated with heating and cooling cycles, and minimize the volume of chemical waste generated. These factors collectively contribute to a more sustainable and cost-effective manufacturing model that aligns with modern environmental, social, and governance (ESG) goals.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis eliminates the need for expensive and toxic activating reagents such as thionyl chloride or aryl triflates, which are often cost-prohibitive at scale. By utilizing readily available sulfonamides and oximes as direct coupling partners, the raw material costs are significantly optimized. Furthermore, the high atom economy of the C-H activation process ensures that a greater proportion of the input mass is converted into the final product, reducing the cost per kilogram of the active pharmaceutical ingredient or agrochemical intermediate.
- Enhanced Supply Chain Reliability: The stability of the starting materials and the robustness of the catalytic system mitigate the risks associated with supply chain disruptions common with sensitive reagents. Since the reaction does not require strictly anhydrous conditions or cryogenic temperatures, it can be performed in a wider range of manufacturing facilities, increasing the pool of potential contract manufacturing organizations. This flexibility ensures consistent delivery schedules and reduces the likelihood of production delays caused by reagent instability or specialized equipment requirements.
- Scalability and Environmental Compliance: The use of dichloromethane, a common industrial solvent, alongside the absence of heavy metal waste streams typical of stoichiometric chromium or manganese oxidations, simplifies waste management protocols. The high selectivity of the iridium catalyst minimizes the formation of byproducts, reducing the load on downstream purification units and lowering the environmental impact of the effluent. This green chemistry profile facilitates easier regulatory approval and supports long-term sustainability initiatives within the chemical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfonamide synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential adopters. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement officers assessing vendor qualifications.
Q: What are the key advantages of this Ir-catalyzed method over traditional sulfonyl chloride routes?
A: This method avoids the use of moisture-sensitive and toxic sulfonyl chlorides, operating instead under mild conditions with readily available sulfonamides and oximes, significantly improving safety and atom economy.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the one-pot nature of the reaction eliminates intermediate isolation steps, reducing processing time and waste, which makes it highly amenable to commercial scale-up for pharmaceutical and agrochemical applications.
Q: What is the typical yield and purity profile for these sulfonamide derivatives?
A: The patent reports yields exceeding 97% for optimized substrates, with products easily purified by standard column chromatography, ensuring high-purity specifications required for fine chemical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonamide Compounds Supplier
As the global demand for high-performance sulfonamide derivatives continues to rise across the pharmaceutical and agrochemical sectors, partnering with an experienced CDMO is critical for successful commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this Ir-catalyzed route can be effectively translated to industrial reality. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch meets the exacting standards required for downstream drug formulation and agricultural application.
We invite industry leaders to collaborate with us to leverage this advanced synthetic technology for their specific pipeline needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume and quality requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in transition metal catalysis can accelerate your time to market while optimizing your overall production costs.
