Advanced Solution-Phase Purification for Scalable Production of Pharmaceutical Intermediates
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for higher purity, lower costs, and more sustainable processes. A pivotal advancement in this domain is detailed in Chinese Patent CN100334067C, which discloses a novel purification method for amide and sulfonamide compounds synthesized via solution-phase combinatorial chemistry. This technology addresses a critical bottleneck in the production of drug candidates: the efficient removal of excess reagents without resorting to expensive and logistically complex solid-phase techniques. By utilizing a small-molecule, water-soluble ionic compound as a nucleophilic scavenger, this method enables the rapid separation of target products through simple liquid-liquid extraction. For R&D directors and procurement managers alike, this represents a significant shift towards more economical and scalable synthetic routes, particularly for the production of high-purity pharmaceutical intermediates where impurity profiles are strictly regulated.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the purification of solution-phase combinatorial libraries has relied heavily on polymer-supported scavengers. While conceptually straightforward, this approach suffers from inherent inefficiencies that hinder large-scale adoption. Polymer resins typically exhibit low loading capacities, necessitating the use of large volumes of solid support to capture excess reagents, which drastically increases material costs and waste generation. Furthermore, the physical nature of solid-phase reactions introduces complications such as swelling effects and diffusion limitations, often leading to incomplete scavenging and inconsistent product quality. From a supply chain perspective, the reliance on specialized functionalized resins creates dependency on niche suppliers and complicates the workflow with additional filtration steps that are difficult to automate or scale continuously. These factors collectively contribute to extended lead times and elevated operational expenditures, making conventional resin-based purification less attractive for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
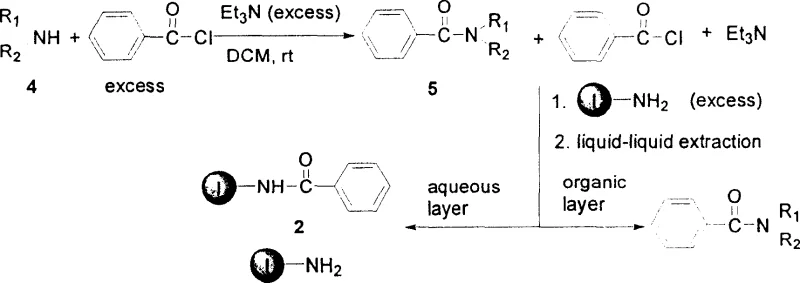
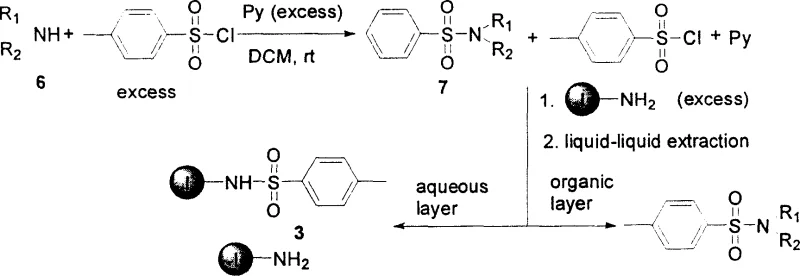
In stark contrast, the methodology outlined in the patent utilizes a water-soluble amino nucleophilic scavenger, specifically N-aminoethylpyridinium bromide, to achieve superior purification efficiency. This small-molecule approach leverages the principles of homogeneous catalysis and phase separation to streamline the workflow. The scavenger reacts rapidly with excess electrophilic reagents such as benzoyl chloride or 4-toluenesulfonyl chloride at room temperature, forming ionic byproducts that are inherently soluble in aqueous media. Consequently, the purification step is reduced to a simple wash with brine, allowing the target organic product to remain in the organic layer while impurities are sequestered in the aqueous phase. This eliminates the need for filtration equipment and expensive resin beds, significantly simplifying the process infrastructure. For a reliable pharmaceutical intermediates supplier, adopting this liquid-phase strategy translates to robust process control and the ability to handle diverse substrate scopes ranging from aromatic amines to aliphatic amines with consistent results.
Mechanistic Insights into Water-Soluble Scavenger Purification
The core of this technological breakthrough lies in the unique chemical properties of the N-aminoethylpyridinium bromide scavenger. Mechanistically, the primary amine group on the scavenger acts as a potent nucleophile, attacking the carbonyl carbon of unreacted acyl chlorides or the sulfur atom of sulfonyl chlorides. This nucleophilic substitution reaction proceeds efficiently under mild conditions, typically requiring only 6 to 12 hours of stirring at room temperature. The resulting adducts retain the quaternary ammonium moiety of the scavenger, rendering them highly polar and water-soluble salts. This polarity difference is the driving force behind the separation; when the reaction mixture is diluted with ethyl acetate and washed with saline solution, the ionic scavenger-adducts partition almost exclusively into the aqueous layer. This precise control over solubility ensures that the organic phase contains primarily the desired amide or sulfonamide product, minimizing the carryover of nitrogen-containing impurities that often plague traditional workups.
Furthermore, the versatility of this mechanism extends to both amide and sulfonamide synthesis, accommodating a wide array of amine substrates including aniline, benzylamine, morpholine, and cyclohexylamine. The reaction stoichiometry is carefully optimized, typically employing a slight excess of the acylating agent (2.0 to 2.5 mmol per 1 mmol of amine) followed by a stoichiometric amount of the scavenger (1.2 to 2.0 mmol) to ensure complete consumption of the electrophile. The robustness of this chemical design allows for high yields, often exceeding 85%, and purities greater than 95% as confirmed by GC/MS analysis. Such high fidelity in impurity control is essential for meeting the stringent specifications required in API manufacturing, ensuring that downstream processing is not compromised by residual reactive species.
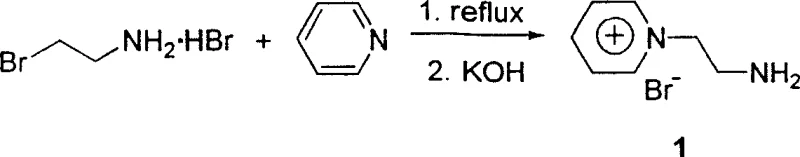
The preparation of the scavenger itself is another testament to the process efficiency. Synthesized from readily available commodity chemicals like pyridine and 2-bromoethylamine hydrochloride, the scavenger can be produced in high yield (approximately 90%) through a simple reflux and neutralization sequence. This ease of preparation ensures a stable and cost-effective supply of the key purification reagent. The structural integrity of the scavenger, characterized by distinct NMR signals and a melting point range of 227.9 to 229.6°C, guarantees batch-to-batch consistency. By integrating the synthesis of the scavenger into the broader manufacturing workflow, producers can further reduce dependency on external vendors and mitigate supply chain risks associated with specialized reagents.
How to Synthesize High-Purity Amide Intermediates Efficiently
Implementing this purification strategy requires adherence to specific operational parameters to maximize yield and purity. The process begins with the dissolution of the amine substrate in dichloromethane, followed by the controlled addition of the acylating agent and a base such as triethylamine or pyridine. Reaction progress is monitored via gas chromatography to ensure complete conversion before the scavenger is introduced. Once the scavenging step is complete, the workup involves dilution with ethyl acetate and multiple washes with brine to extract the ionic byproducts. The organic layer is then passed through a short silica gel column to remove any trace polar impurities before drying and concentration. For detailed standardized operating procedures and specific stoichiometric ratios for various substrates, please refer to the comprehensive guide below.
- Dissolve the amine compound in dichloromethane and react with excess benzoyl chloride or 4-toluenesulfonyl chloride in the presence of a base like triethylamine or pyridine at room temperature.
- Add the water-soluble nucleophilic scavenger, N-aminoethylpyridinium bromide, to the reaction mixture to consume any remaining electrophilic reagents.
- Dilute the mixture with ethyl acetate and perform liquid-liquid extraction using brine to remove the water-soluble scavenger and its byproducts, then dry and concentrate the organic phase.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial standpoint, the adoption of this water-soluble scavenger technology offers profound benefits for cost reduction in API manufacturing and overall supply chain resilience. By replacing expensive polymer-supported resins with a synthetically accessible small molecule, the direct material costs associated with purification are drastically reduced. The elimination of solid-phase filtration steps also reduces solvent consumption and waste disposal costs, contributing to a more sustainable and economically viable process. Moreover, the simplicity of the liquid-liquid extraction workup allows for faster turnaround times between batches, enhancing production throughput without the need for significant capital investment in new equipment. This operational agility is crucial for maintaining competitive pricing and meeting tight delivery schedules in the fast-paced pharmaceutical market.
- Cost Reduction in Manufacturing: The transition from polymer resins to water-soluble scavengers removes the premium pricing associated with functionalized solid supports. Since the scavenger is synthesized from bulk chemicals, the cost per mole of purification reagent is significantly lower. Additionally, the process avoids the loss of product often associated with adsorption onto resin matrices, thereby improving overall mass balance and effective yield. This economic efficiency allows manufacturers to offer high-purity amide intermediates at more competitive price points while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like pyridine and bromoethylamine for scavenger production mitigates the risk of supply disruptions common with specialized proprietary resins. The ability to produce the scavenger in-house or source it from multiple general chemical suppliers ensures a continuous flow of materials. This redundancy is vital for reducing lead time for high-purity sulfonamides and other critical intermediates, ensuring that production schedules are not derailed by vendor shortages or logistics bottlenecks.
- Scalability and Environmental Compliance: The liquid-phase nature of this purification method aligns perfectly with standard industrial chemical engineering practices. Scaling up from gram to kilogram or ton quantities involves simply increasing reactor volumes and extraction stages, without the complexities of handling large masses of swollen resin. Furthermore, the aqueous waste stream containing the ionic scavenger byproducts is easier to treat and manage compared to solid chemical waste, facilitating compliance with increasingly stringent environmental regulations and supporting green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using water-soluble scavengers over polymer-supported resins?
A: Water-soluble scavengers offer higher reactivity and lower costs compared to polymer resins. They allow for purification via simple liquid-liquid extraction rather than filtration, eliminating issues related to resin swelling, low loading capacity, and solid-phase reaction effects.
Q: How does this method improve the purity of pharmaceutical intermediates?
A: The method effectively removes excess acylating agents and the scavenger itself through aqueous washing. The resulting ionic byproducts partition into the water layer, leaving the target amide or sulfonamide in the organic phase with high purity, often exceeding 95%.
Q: Is this purification method suitable for large-scale manufacturing?
A: Yes, the process is highly scalable because it relies on standard liquid-liquid extraction techniques common in industrial chemical production, avoiding the logistical challenges and high costs associated with handling large volumes of solid polymer resins.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced purification technologies like the one described in CN100334067C for enhancing the quality and efficiency of pharmaceutical production. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that in the pharmaceutical industry, consistency is key, and our infrastructure is designed to deliver high-purity amide intermediates that meet the exacting standards of global regulatory bodies.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating how this purification strategy can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline with reliable, high-quality chemical solutions.
