Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Pharmaceutical Intermediates Production
Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Pharmaceutical Intermediates Production
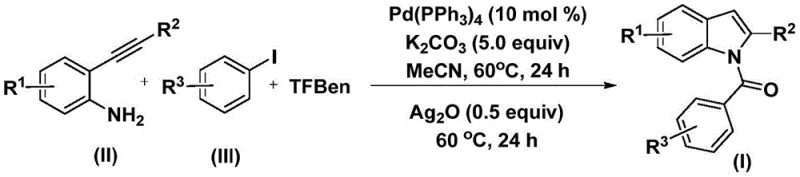
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds, particularly indoles, which serve as critical backbones in a vast array of bioactive molecules ranging from anti-inflammatory agents to anti-tumor drugs. Patent CN112898192B introduces a significant technological advancement in this domain by disclosing a highly efficient preparation method for N-acyl indole compounds. This innovation leverages a palladium-catalyzed carbonylation cyclization strategy that utilizes 2-alkynyl anilines and aryl iodides as primary starting materials. Unlike traditional methods that often rely on dangerous gaseous carbon monoxide, this novel approach employs a solid CO surrogate, phenyl 1,3,5-tricarboxylate (TFBen), thereby drastically improving operational safety and ease of handling. The significance of this development cannot be overstated for manufacturers aiming to produce high-purity pharmaceutical intermediates, as it combines mild reaction conditions with exceptional substrate tolerance.
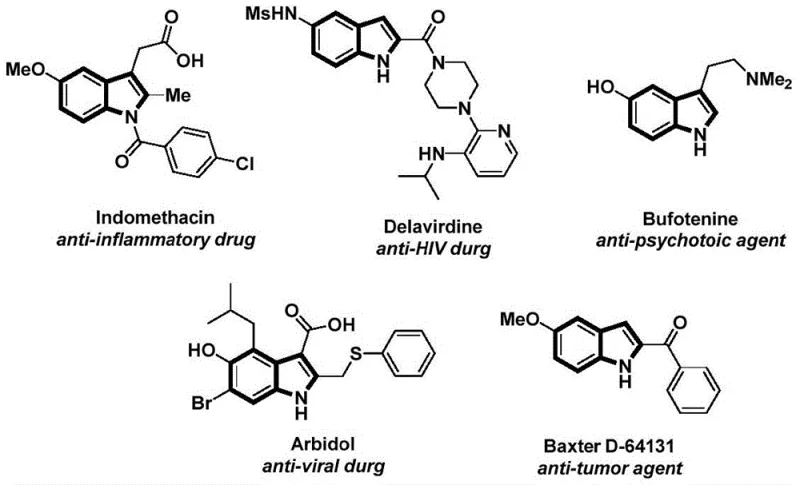
Indole derivatives are ubiquitous in medicinal chemistry, evidenced by their presence in blockbuster drugs such as Indomethacin, an anti-inflammatory agent, and Delavirdine, an anti-HIV drug. The structural diversity required for these applications demands synthetic methodologies that are not only efficient but also versatile enough to accommodate various functional groups without extensive protection-deprotection sequences. The technology described in CN112898192B addresses this need by providing a one-pot synthesis route that constructs the N-acyl indole core directly. This capability is essential for a reliable pharmaceutical intermediate supplier, as it allows for the rapid generation of compound libraries for drug discovery and the streamlined production of key intermediates for commercial API manufacturing. The ability to access these complex structures through a simplified workflow represents a paradigm shift in how fine chemical intermediates are produced.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indoles via carbonylation has faced significant hurdles that limited its widespread industrial application. Conventional carbonylation reactions typically require the use of carbon monoxide gas, which poses severe safety risks due to its high toxicity and flammability, necessitating specialized high-pressure equipment and rigorous safety protocols that increase capital expenditure. Furthermore, traditional methods often suffer from poor atom economy and require harsh reaction conditions, such as elevated temperatures and pressures, which can lead to the decomposition of sensitive functional groups present on the substrate. These limitations result in lower overall yields, complex purification processes to remove metal impurities, and a narrower scope of compatible substrates, ultimately driving up the cost of goods sold and extending the lead time for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the method disclosed in patent CN112898192B offers a transformative solution by utilizing phenyl 1,3,5-tricarboxylate (TFBen) as a safe and convenient solid source of carbon monoxide. This innovation eliminates the need for handling hazardous CO gas, allowing the reaction to proceed under atmospheric pressure in standard laboratory glassware or reactors. The process operates at a mild temperature of 60°C in acetonitrile, conditions that are energetically favorable and compatible with a wide range of thermally sensitive functionalities. By integrating the carbonylation and cyclization steps into a seamless two-stage sequence within a single pot, this novel approach significantly reduces waste generation and simplifies the downstream processing workflow. This efficiency makes it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing, providing a competitive edge in the global supply chain.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation is a sophisticated orchestration of organometallic steps that ensures high selectivity and yield. The reaction initiates with the oxidative addition of the palladium(0) catalyst into the carbon-iodine bond of the aryl iodide substrate, generating a reactive aryl-palladium(II) intermediate. Subsequently, carbon monoxide, which is released in situ from the thermal decomposition of the TFBen surrogate, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the amino group of the 2-alkynyl aniline, followed by reductive elimination to yield an amide precursor. The final and crucial step involves the addition of silver oxide (Ag2O), which acts as an oxidant and promoter to facilitate the intramolecular cyclization of the alkyne moiety, closing the indole ring and regenerating the active palladium catalyst for the next cycle.
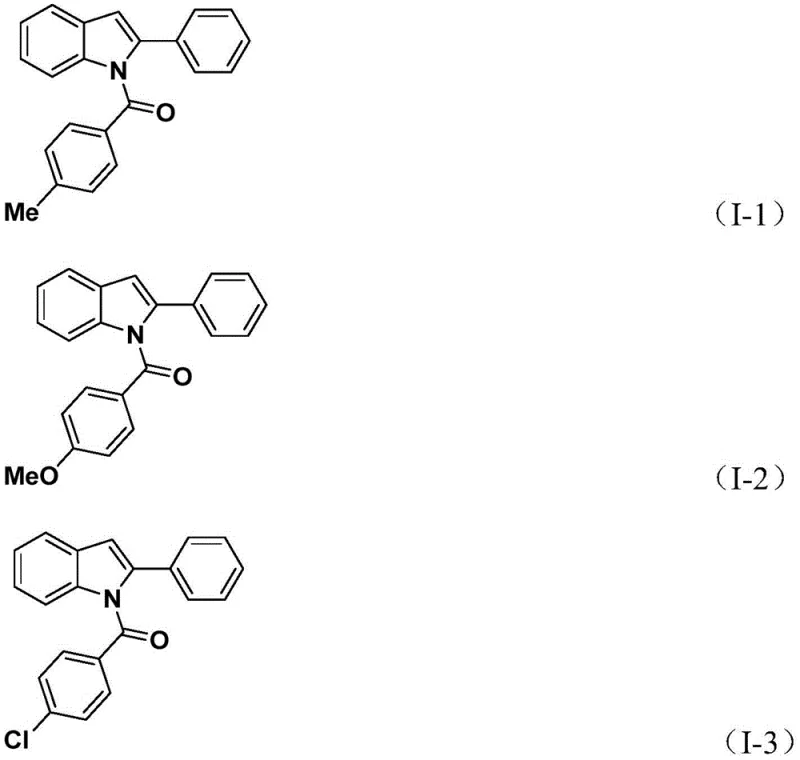
Understanding the role of each reagent is vital for optimizing the impurity profile of the final product. The use of potassium carbonate as a base ensures the deprotonation of the amine nucleophile without promoting side reactions such as hydrolysis of the ester surrogate. Moreover, the specific choice of tetrakis(triphenylphosphine)palladium as the catalyst provides the necessary steric and electronic environment to stabilize the intermediates throughout the catalytic cycle. The broad substrate compatibility observed in the patent data, where substituents like fluorine, chlorine, bromine, and methoxy groups are well-tolerated, suggests that the electronic properties of the substrates do not significantly hinder the oxidative addition or CO insertion steps. This robustness is critical for R&D directors focusing on purity and杂质谱 (impurity profiles), as it minimizes the formation of by-products that are difficult to separate.
How to Synthesize N-Acyl Indole Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for generating N-acyl indoles with high fidelity. The procedure begins by charging a reaction vessel with the palladium catalyst, potassium carbonate, the solid CO surrogate TFBen, the 2-alkynyl aniline, and the aryl iodide in acetonitrile. The mixture is then heated to 60°C and stirred for 24 hours to allow the initial carbonylation and amidation to reach completion. Following this first stage, silver oxide is introduced to the reaction mixture, and heating is continued at 60°C for an additional 24 hours to drive the cyclization to completion. Upon cooling, the reaction mixture is filtered to remove insoluble salts and metal residues, and the crude product is purified via column chromatography to afford the target N-acyl indole compound in high purity.
- Combine palladium catalyst (Pd(PPh3)4), potassium carbonate, phenyl 1,3,5-tricarboxylate (TFBen), 2-alkynyl aniline, and aryl iodide in acetonitrile solvent.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and amidation steps.
- Add silver oxide (Ag2O) to the mixture and continue heating at 60°C for an additional 24 hours to promote cyclization, followed by filtration and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical efficiency. The substitution of gaseous carbon monoxide with a stable solid surrogate fundamentally alters the risk profile of the manufacturing process, reducing insurance costs and eliminating the need for specialized gas storage infrastructure. This shift translates directly into significant cost savings and enhanced supply chain reliability, as the raw materials required—aryl iodides, alkynyl anilines, and TFBen—are commercially available commodities with stable pricing and consistent quality. Furthermore, the mild reaction conditions reduce energy consumption compared to high-temperature/high-pressure alternatives, contributing to a lower carbon footprint and aligning with modern sustainability goals in chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and hazardous gas handling systems results in substantially reduced capital expenditure and operational overhead. Additionally, the high reaction efficiency and yield reported in the patent examples minimize raw material waste, ensuring that every kilogram of input generates maximum output value. The simplified workup procedure, which avoids complex extraction or distillation steps, further lowers labor and utility costs, making the overall process economically superior to legacy methods.
- Enhanced Supply Chain Reliability: By relying on shelf-stable solid reagents rather than compressed gases, manufacturers can maintain consistent production schedules without the risk of supply interruptions due to gas delivery logistics. The robustness of the reaction across a wide range of substrates means that a single standardized protocol can be used to produce a diverse portfolio of intermediates, simplifying inventory management and reducing the complexity of the supply chain network.
- Scalability and Environmental Compliance: The use of acetonitrile as a solvent and the absence of toxic CO gas make this process inherently safer and easier to scale from gram-scale laboratory synthesis to multi-ton commercial production. The waste stream is primarily composed of benign inorganic salts and spent catalyst, which are easier to treat and dispose of in compliance with environmental regulations, thereby reducing the burden on waste management systems and mitigating regulatory risks.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this patented technology in a commercial setting. These answers are derived directly from the experimental data and specifications provided in patent CN112898192B, ensuring accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production lines or new product development pipelines.
Q: What is the source of carbon monoxide in this synthesis method?
A: The method utilizes phenyl 1,3,5-tricarboxylate (TFBen) as a solid carbon monoxide surrogate, which releases CO in situ under the reaction conditions, avoiding the need for hazardous high-pressure CO gas cylinders.
Q: What are the optimal reaction conditions for high yield?
A: The patent specifies using acetonitrile (MeCN) as the solvent at a temperature of 60°C. The process involves a two-stage heating protocol: 24 hours for the initial coupling followed by the addition of silver oxide and another 24 hours for cyclization.
Q: Does this method tolerate diverse functional groups on the substrates?
A: Yes, the methodology demonstrates excellent substrate compatibility, successfully accommodating various substituents such as methyl, methoxy, halogens (F, Cl, Br), and tert-butyl groups on both the aniline and aryl iodide components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic technologies to meet the evolving demands of the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the methodology described in CN112898192B and is fully equipped to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-quality intermediates. Our state-of-the-art facilities are designed to handle complex organometallic reactions with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to guarantee product integrity.
We invite potential partners to engage with our technical procurement team to discuss how this advanced N-acyl indole synthesis can optimize your specific project requirements. Whether you are in the early stages of drug discovery or preparing for large-scale API manufacturing, we can provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive success in your supply chain.
