Advanced Palladium-Catalyzed Synthesis of N-Acyl Indoles for Scalable Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of N-Acyl Indoles for Scalable Pharmaceutical Manufacturing
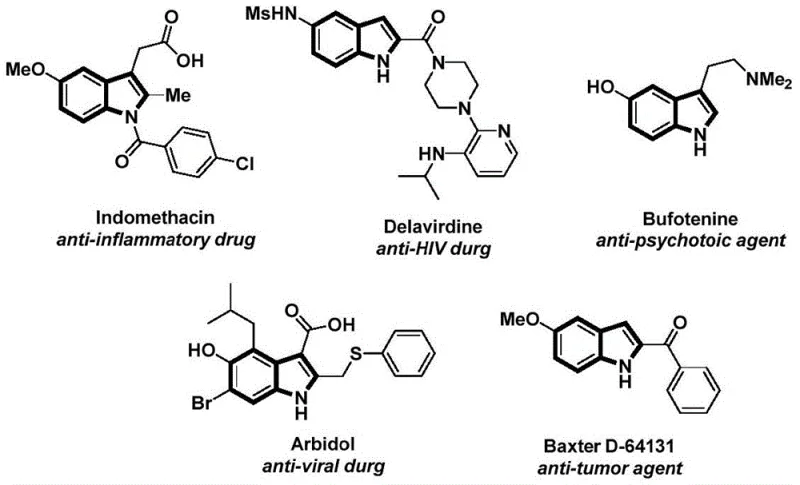
The structural motif of the indole ring serves as a foundational backbone in medicinal chemistry, appearing in a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to potent anti-tumor compounds. As depicted in the visual overview of indole-based pharmaceuticals, the versatility of this heterocyclic system is unmatched in modern drug discovery. However, the efficient construction of N-acyl indole derivatives has historically presented significant synthetic challenges, often requiring harsh conditions or multi-step sequences that hinder rapid scale-up. A groundbreaking development detailed in patent CN112898192B introduces a streamlined, palladium-catalyzed carbonylation cyclization strategy that addresses these bottlenecks. This innovative methodology leverages readily available 2-alkynyl anilines and aryl iodides to construct the N-acyl indole core in a highly efficient manner. By integrating a solid carbon monoxide surrogate, the process not only enhances safety profiles but also simplifies the operational complexity typically associated with carbonylation reactions. For global procurement teams and R&D directors seeking reliable pharmaceutical intermediate suppliers, this technology represents a pivotal shift towards more sustainable and cost-effective manufacturing pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for accessing N-acyl indole scaffolds frequently rely on direct acylation of indoles or complex cyclization protocols that suffer from poor atom economy and limited functional group tolerance. Many established methods necessitate the use of gaseous carbon monoxide under high pressure, which imposes severe safety constraints and requires specialized high-pressure reactor infrastructure that is not universally available in standard pilot plants. Furthermore, conventional approaches often struggle with regioselectivity issues, leading to difficult-to-separate impurity profiles that compromise the purity specifications required for active pharmaceutical ingredients (APIs). The reliance on stoichiometric amounts of toxic reagents or expensive transition metal complexes without efficient recycling mechanisms further exacerbates the environmental footprint and production costs. These factors collectively create substantial barriers for supply chain heads aiming to reduce lead time for high-purity pharmaceutical intermediates, as the purification burden often outweighs the synthesis speed. Consequently, there is an urgent industry demand for methodologies that can bypass these inherent limitations while maintaining rigorous quality standards.
The Novel Approach
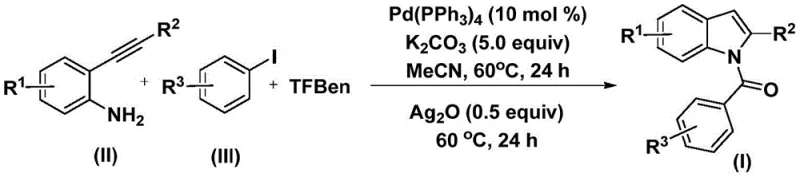
The patented methodology described in CN112898192B offers a transformative solution by employing a tandem carbonylation-cyclization sequence catalyzed by palladium under remarkably mild conditions. Instead of handling dangerous CO gas, the process utilizes 1,3,5-tricarboxylic acid phenol ester (TFBen) as a safe, solid carbon monoxide substitute that releases CO in situ. This strategic substitution allows the reaction to proceed in standard glassware or stainless steel reactors without the need for autoclaves, drastically simplifying the engineering requirements for commercial scale-up of complex pharmaceutical intermediates. The reaction operates at a moderate temperature of 60°C in acetonitrile, demonstrating exceptional energy efficiency compared to high-temperature alternatives. Moreover, the one-pot nature of the synthesis minimizes intermediate isolation steps, thereby reducing solvent consumption and overall processing time. This approach not only accelerates the timeline from bench to kilogram scale but also ensures a cleaner reaction profile, making it an ideal candidate for cost reduction in API manufacturing where operational simplicity translates directly to margin improvement.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation is a sophisticated orchestration of organometallic steps that ensures high fidelity in bond formation. The cycle initiates with the oxidative addition of the palladium(0) catalyst into the carbon-iodine bond of the aryl iodide substrate, generating a reactive aryl-palladium(II) intermediate. Subsequently, carbon monoxide, generated thermally from the TFBen precursor, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the amino group of the 2-alkynyl aniline, followed by reductive elimination to yield an amide precursor. The final critical step involves the silver oxide-mediated intramolecular cyclization, which activates the alkyne moiety and facilitates the closure of the five-membered pyrrole ring to furnish the target N-acyl indole. Understanding this mechanism is crucial for R&D directors focusing on purity and impurity profiles, as it highlights the specific roles of each reagent in driving the reaction to completion without forming significant side products.
Control over the impurity spectrum is inherently built into this catalytic cycle due to the high chemoselectivity of the palladium system. The use of potassium carbonate as a mild base ensures that sensitive functional groups on the aromatic rings remain intact throughout the process, preventing degradation or unwanted side reactions that often plague harsher acidic or basic conditions. The specific choice of tetrakis(triphenylphosphine)palladium as the catalyst provides a stable yet active species that maintains turnover numbers sufficient for practical synthesis without requiring excessive loading. Furthermore, the sequential addition of silver oxide allows for temporal control over the cyclization event, ensuring that the amide formation is complete before the ring-closing step commences. This staged approach minimizes the formation of oligomeric byproducts or incomplete cyclization intermediates, resulting in a crude product mixture that is significantly easier to purify. For quality assurance teams, this mechanistic robustness translates to consistent batch-to-batch reproducibility and adherence to stringent purity specifications required for regulatory filings.
How to Synthesize N-Acyl Indole Efficiently
The operational protocol for this synthesis is designed to be accessible for both laboratory research and industrial production environments. The procedure begins by charging a reaction vessel with the palladium catalyst, potassium carbonate, the solid CO source TFBen, the 2-alkynyl aniline, and the aryl iodide in acetonitrile. The mixture is stirred and heated to 60°C for 24 hours to allow the carbonylation and amidation steps to reach full conversion. Following this initial phase, silver oxide is introduced to the reaction mixture, and heating is continued at the same temperature for an additional 24 hours to drive the cyclization to completion. Detailed standardized synthesis steps see the guide below.
- Combine palladium catalyst, potassium carbonate, 1,3,5-tricarboxylic acid phenol ester (TFBen), 2-alkynyl aniline, and aryl iodide in an organic solvent such as acetonitrile.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation.
- Add silver oxide to the mixture and continue heating at 60°C for an additional 24 hours to induce cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the N-acyl indole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process delivers compelling value propositions that directly address the pain points of modern chemical supply chains. The elimination of high-pressure gas infrastructure reduces capital expenditure requirements for manufacturing facilities, allowing for more flexible production scheduling and lower overhead costs. The use of commercially available starting materials, such as simple aryl iodides and substituted anilines, ensures a robust and resilient supply chain that is less susceptible to raw material shortages. Additionally, the mild reaction conditions contribute to enhanced process safety, lowering insurance premiums and regulatory compliance burdens associated with hazardous operations. These factors combine to create a manufacturing route that is not only technically superior but also economically advantageous for long-term procurement strategies.
- Cost Reduction in Manufacturing: The replacement of gaseous carbon monoxide with a solid surrogate eliminates the need for specialized gas handling equipment and safety monitoring systems, leading to substantial operational cost savings. By consolidating the synthesis into a one-pot procedure, the process reduces solvent usage, labor hours, and waste disposal costs associated with multiple isolation steps. The high reaction efficiency observed across various substrates means that less starting material is wasted on side products, improving the overall material balance and yield per batch. These cumulative efficiencies result in a significantly lower cost of goods sold (COGS), enabling competitive pricing for downstream API manufacturers without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable solid reagents like TFBen and common aryl halides mitigates the risks associated with the logistics of hazardous gases. This stability allows for larger inventory buffers and more predictable lead times, ensuring continuity of supply even during market fluctuations. The broad substrate compatibility means that a single manufacturing platform can be adapted to produce a wide library of N-acyl indole derivatives, providing flexibility to respond quickly to changing customer demands. For supply chain heads, this versatility translates to a more agile and responsive sourcing strategy that can accommodate diverse project pipelines with minimal retooling.
- Scalability and Environmental Compliance: The moderate temperature profile and ambient pressure operation make this process inherently scalable from gram to ton quantities without encountering the heat transfer or safety limitations of exothermic high-pressure reactions. The use of acetonitrile, a common and recyclable solvent, aligns with green chemistry principles and simplifies waste stream management. Reduced generation of hazardous byproducts lowers the environmental impact and simplifies the permitting process for new production lines. This alignment with sustainability goals is increasingly critical for multinational corporations aiming to meet their corporate social responsibility targets while maintaining efficient production capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and scope defined in the patent literature to provide clarity for potential adopters. Understanding these nuances is essential for evaluating the feasibility of this route for specific target molecules.
Q: What carbon monoxide source is used in this novel synthesis method?
A: The process utilizes 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide substitute, eliminating the need for hazardous high-pressure CO gas cylinders.
Q: What are the optimal reaction conditions for maximizing yield?
A: The patent specifies using tetrakis(triphenylphosphine)palladium as the catalyst in acetonitrile solvent at 60°C, with a two-stage heating process totaling 48 hours.
Q: Does this method support diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent substrate compatibility, tolerating various substituents such as halogens, alkyl groups, alkoxy groups, and trifluoromethyl groups on the aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into reliable supply streams. We are committed to delivering high-purity N-acyl indole intermediates that meet the rigorous QC labs and stringent purity specifications demanded by the global pharmaceutical industry. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed processes, including efficient metal scavenging and solvent recovery systems.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce overall project costs.
