Advanced Manufacturing of Trilaciclib: A Novel 5-Step Synthetic Route for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust and scalable pathways for complex oncology therapeutics, and the recent disclosure in patent CN114014864A presents a significant breakthrough in the manufacturing of Trilaciclib, a first-in-class CDK4/6 inhibitor. This novel preparation process addresses critical bottlenecks in the existing supply chain by utilizing a streamlined 5-step synthetic route that begins with readily available commodity chemicals. The strategic design of this pathway focuses on maximizing atom economy and minimizing purification complexity, which are essential factors for reliable API intermediate supplier operations. By leveraging a unique combination of spiro-lactam formation and advanced palladium-catalyzed coupling, this method achieves exceptional yields and purity profiles that meet the rigorous standards required for clinical and commercial drug substance production.
Trilaciclib serves as a vital myeloprotective agent, approved for preventing chemotherapy-induced myelosuppression, making its consistent availability a priority for global health security. The chemical architecture of Trilaciclib, specifically the fused pyrrolo[2,3-d]pyrimidine core linked to a spiro-cyclic lactam, poses significant synthetic challenges regarding regioselectivity and stereochemical integrity. 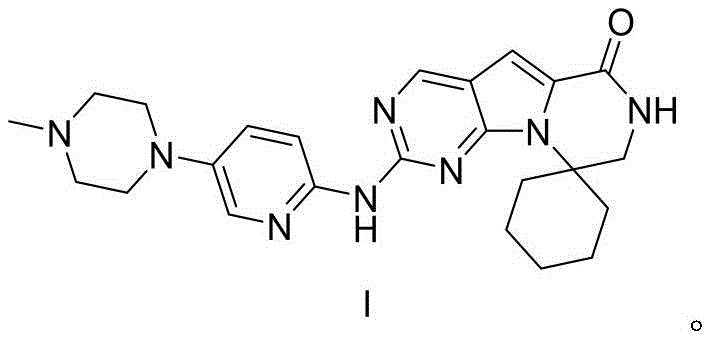 As illustrated in the structural diagram, the molecule requires precise construction of the spiro-center and the subsequent attachment of the piperazine-pyridine side chain. The patented process overcomes historical difficulties in constructing this scaffold by introducing a highly efficient sequence that avoids excessive protecting group manipulations, thereby reducing the overall environmental footprint and production timeline.
As illustrated in the structural diagram, the molecule requires precise construction of the spiro-center and the subsequent attachment of the piperazine-pyridine side chain. The patented process overcomes historical difficulties in constructing this scaffold by introducing a highly efficient sequence that avoids excessive protecting group manipulations, thereby reducing the overall environmental footprint and production timeline.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for complex kinase inhibitors often suffer from low overall yields due to the accumulation of losses across multiple linear steps, particularly when constructing crowded spiro-cyclic systems. Conventional methodologies frequently rely on harsh halogenation conditions or expensive transition metal catalysts that require rigorous removal protocols to meet residual metal specifications, adding substantial cost and time to the downstream processing. Furthermore, older approaches to the pyrimidine core assembly often involve unstable intermediates that necessitate low-temperature storage and specialized handling equipment, creating bottlenecks in batch processing. The reliance on column chromatography for purification in early-stage process development is another major drawback, as this technique is notoriously difficult to scale and generates significant volumes of hazardous solvent waste, conflicting with modern green chemistry principles.
The Novel Approach
In stark contrast, the novel approach detailed in the patent introduces a convergent strategy that simplifies the construction of the key spiro-lactam intermediate through a clever one-pot tandem reaction. Instead of isolating unstable amine intermediates, the process utilizes a dual-base system involving aniline and DMAP to sequentially drive acylation and cyclization, effectively telescoping two reaction steps into a single operational unit. This innovation not only improves the throughput of the manufacturing line but also significantly enhances the safety profile by minimizing the handling of reactive bromoacetyl species. The subsequent coupling of the uracil-derived core with the spiro-lactam fragment is optimized to proceed under mild thermal conditions, preserving the integrity of the sensitive functional groups and ensuring a high-purity profile for the final active pharmaceutical ingredient.
Mechanistic Insights into Pd-Catalyzed Amination and Spiro-Lactam Formation
The cornerstone of this synthetic success lies in the mechanistic elegance of the initial spiro-lactam formation and the final C-N bond construction. In the first step, the reaction between 1-(aminomethyl)cyclohexanamine and bromoacetyl bromide is meticulously controlled using a specific molar ratio of bases to direct the reaction pathway. The initial use of aniline acts as a mild acid scavenger to facilitate the formation of the amide bond without triggering premature cyclization, while the subsequent addition of the stronger nucleophilic catalyst DMAP promotes the intramolecular displacement to close the lactam ring. This sequential addition prevents the formation of polymeric byproducts and ensures that the Boc-protection occurs selectively on the exocyclic nitrogen, setting the stage for the subsequent coupling reactions with high fidelity.
The final step involves a sophisticated Buchwald-Hartwig amination, where the chloro-pyrimidine intermediate reacts with the piperazine-pyridine amine fragment. The patent specifies the use of Pd2(dba)3 as the palladium source combined with BINAP as the ligand, a combination known for facilitating difficult couplings on electron-deficient heterocycles. The presence of potassium tert-butoxide serves to generate the reactive amido-species in situ, which then undergoes oxidative addition and reductive elimination cycles with the palladium catalyst to forge the C-N bond. 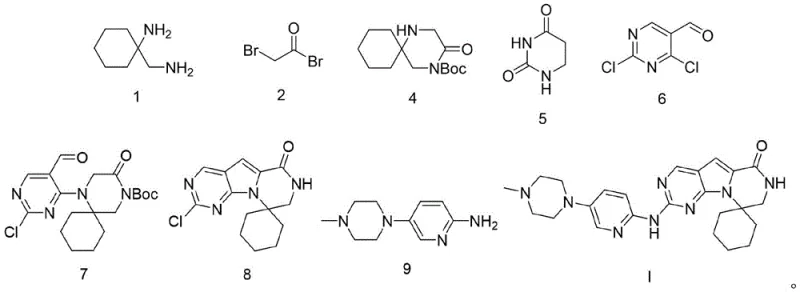 Optimization of the ligand-to-metal ratio is critical here, as the patent data indicates that deviations can lead to significant drops in yield and purity. This mechanistic precision allows the reaction to proceed to completion with minimal formation of dehalogenated side products, resulting in a crude product quality that often requires only simple pulping for purification.
Optimization of the ligand-to-metal ratio is critical here, as the patent data indicates that deviations can lead to significant drops in yield and purity. This mechanistic precision allows the reaction to proceed to completion with minimal formation of dehalogenated side products, resulting in a crude product quality that often requires only simple pulping for purification.
How to Synthesize Trilaciclib Efficiently
The synthesis of Trilaciclib via this patented route offers a clear roadmap for process chemists aiming to establish a robust manufacturing protocol. The procedure is divided into five distinct chemical transformations, beginning with the preparation of the Boc-protected spiro-lactam and concluding with the final palladium-catalyzed coupling. Each step has been optimized for specific temperature ranges and stoichiometric ratios to maximize conversion while minimizing impurity generation. For R&D teams looking to implement this technology, it is crucial to adhere to the specified solvent systems, such as the dichloromethane/n-heptane mixture for pulping, which facilitates the isolation of high-purity solids without the need for energy-intensive distillation. Detailed standard operating procedures for each transformation, including precise addition rates and quenching protocols, are essential for reproducibility.
- Synthesize Boc-protected spiro-lactam (Compound 4) from 1-(aminomethyl)cyclohexanamine and bromoacetyl bromide using a dual-base system.
- Prepare chlorinated formyl-uracil (Compound 6) via reaction with POCl3 and H3BO3.
- Couple Compound 6 and Compound 4 to form intermediate Compound 7, followed by cyclization with tBuOK to yield Compound 8.
- Perform final Buchwald-Hartwig amination between Compound 8 and the piperazine-pyridine amine (Compound 9) using Pd2(dba)3/BINAP catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the economic implications of this new synthetic route are profound, offering a pathway to substantial cost reduction in API intermediate manufacturing. The primary driver of cost efficiency is the selection of starting materials; uracil and bromoacetyl bromide are commodity chemicals available in bulk quantities at stable prices, insulating the production cost from the volatility associated with exotic custom synthons. Furthermore, the high yield observed in the uracil chlorination step, reported to be over 96%, ensures that raw material utilization is maximized, directly translating to lower cost of goods sold (COGS). The elimination of complex purification steps in favor of simple filtration and pulping further reduces the operational expenditure related to solvent recovery and waste disposal.
- Cost Reduction in Manufacturing: The implementation of a one-pot synthesis for the spiro-lactam intermediate eliminates the need for isolating and purifying reactive intermediates, which significantly reduces solvent consumption and labor hours. By telescoping the acylation, cyclization, and protection steps, the process minimizes the number of unit operations required, leading to a drastic simplification of the production schedule. This efficiency gain allows for higher batch throughput within existing facility footprints, effectively increasing capacity without capital investment in new reactors. Additionally, the avoidance of column chromatography in the final steps removes a major cost center typically associated with silica gel consumption and large solvent volumes.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved as the route depends on widely available building blocks rather than specialized reagents that may have single-source suppliers. The robustness of the reaction conditions, which tolerate moderate temperature fluctuations and standard atmospheric controls, reduces the risk of batch failures due to minor process deviations. This resilience ensures a consistent supply of high-purity intermediates, mitigating the risk of production delays that can impact downstream formulation timelines. The ability to store key intermediates as stable solids further enhances inventory management flexibility, allowing manufacturers to build strategic stockpiles against market demand surges.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing solvents and reagents that are compatible with standard stainless steel reactor trains used in the fine chemical industry. The shift towards pulping purification aligns with increasingly stringent environmental regulations by reducing the volume of hazardous waste generated per kilogram of product. Lower solvent intensity not only reduces the carbon footprint of the manufacturing process but also simplifies the permitting process for new production lines. The mild reaction temperatures employed in the coupling steps reduce energy consumption for heating and cooling, contributing to a more sustainable and economically viable long-term production strategy.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Trilaciclib synthesis process. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing route.
Q: What are the key cost-saving advantages of this Trilaciclib synthesis route?
A: The process utilizes commercially available, low-cost starting materials such as uracil and bromoacetyl bromide. Additionally, the one-pot synthesis of the spiro-lactam intermediate reduces operational steps and solvent usage, significantly lowering manufacturing costs compared to multi-step protection strategies.
Q: How does this method ensure high purity for pharmaceutical applications?
A: The final step employs a optimized Pd-catalyzed amination using specific ligands (BINAP) and bases (tBuOK), which minimizes byproduct formation. The patent reports achieving HPLC purity of up to 99.8% through simplified purification methods like pulping rather than extensive column chromatography.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the route is designed for industrial operability. It features mild reaction conditions, avoids hazardous reagents where possible, and relies on robust purification techniques like crystallization and pulping, which are easily scalable from kilogram to metric ton quantities without complex equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trilaciclib Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of Trilaciclib in the evolving landscape of oncology support care, and we are positioned to be your trusted partner in bringing this molecule to market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest international regulatory standards. Our commitment to quality assurance means that we can consistently deliver high-purity Trilaciclib intermediates that facilitate smooth downstream processing for our global clients.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can be tailored to your specific supply chain requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the total landed cost benefits of adopting this novel process. We encourage potential partners to request specific COA data and route feasibility assessments to validate the performance metrics against your internal benchmarks. Let us collaborate to secure a resilient and cost-effective supply of this critical pharmaceutical intermediate.
