Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Manufacturing
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries continuously seek robust synthetic methodologies to access privileged scaffolds like quinazolinones, which are ubiquitous in bioactive molecules ranging from antifungals to anticancer agents. A significant technological breakthrough in this domain is detailed in patent CN112480015B, which discloses a highly efficient multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. This innovation addresses critical bottlenecks in traditional heterocycle synthesis by leveraging a palladium-catalyzed carbonylation cascade that utilizes cheap nitro compounds and trifluoroethylimidoyl chloride. The introduction of the trifluoromethyl group is particularly strategic, as fluorine atoms dramatically enhance the metabolic stability, lipophilicity, and bioavailability of drug candidates, making this synthetic route invaluable for developing next-generation active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied on synthetic pathways that are fraught with operational complexities and economic inefficiencies. Traditional methods often necessitate the use of high-pressure carbon monoxide gas, which requires specialized autoclaves and rigorous safety protocols due to the extreme toxicity of CO. Furthermore, many established routes depend on pre-activated substrates such as 2-bromoformylaniline or acid anhydrides, which are not only expensive to procure but also generate substantial stoichiometric waste during the activation steps. Other iron or ruthenium-catalyzed variations frequently suffer from narrow substrate scope, failing to tolerate sensitive functional groups, or require harsh reaction conditions that lead to poor yields and difficult purification processes, ultimately hindering their applicability in large-scale commercial manufacturing.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in CN112480015B offers a streamlined, atom-economical solution that transforms simple nitro compounds directly into complex heterocycles. By employing molybdenum hexacarbonyl [Mo(CO)6] as a solid carbon monoxide surrogate, the process completely eliminates the need for handling hazardous CO gas, thereby enhancing workplace safety and reducing infrastructure costs. The reaction proceeds through a tandem sequence where the nitro group is reduced in situ, followed by coupling and cyclization, all within a single pot. This one-pot strategy minimizes solvent usage, reduces work-up time, and significantly improves the overall process mass intensity (PMI), making it an ideal candidate for green chemistry initiatives in fine chemical production.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
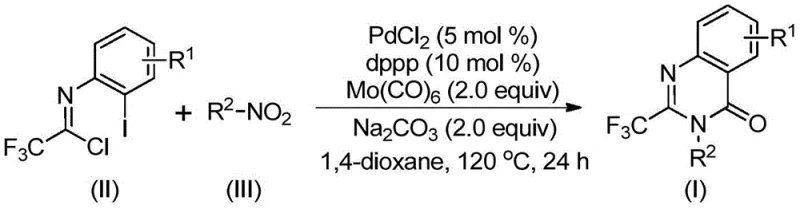
The elegance of this synthesis lies in its intricate yet efficient catalytic cycle, which orchestrates multiple bond-forming events seamlessly. The mechanism is initiated by the reduction of the nitro compound to the corresponding amine by Mo(CO)6, which simultaneously releases carbon monoxide under thermal conditions. This generated amine then undergoes a base-promoted nucleophilic attack on the trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate. Subsequently, the palladium catalyst, coordinated with the dppp ligand, inserts into the carbon-iodine bond of the imidoyl chloride moiety, generating a reactive organopalladium species. The released carbon monoxide then inserts into the carbon-palladium bond to form an acyl-palladium intermediate, setting the stage for the final ring closure.
Following the carbonyl insertion, the nitrogen atom of the amidine moiety coordinates with the palladium center, facilitating the formation of a seven-membered palladacycle intermediate. This crucial step is driven by the presence of the base, sodium carbonate, which neutralizes the acidic byproducts and promotes the cyclization. The catalytic cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted quinazolinone product and regenerates the active Pd(0) catalyst. This mechanistic pathway ensures high regioselectivity and minimizes the formation of side products, resulting in the clean impurity profiles required for pharmaceutical grade intermediates.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting is straightforward, relying on standard Schlenk line techniques or sealed pressure vessels capable of withstanding moderate temperatures. The protocol involves charging the reaction vessel with the palladium catalyst system, the solid CO source, and the organic substrates in an aprotic solvent such as 1,4-dioxane. The reaction is typically heated to 120°C for a period ranging from 16 to 30 hours, allowing sufficient time for the slow release of CO and the subsequent cascade reactions to reach completion. Detailed standardized operating procedures for scaling this reaction from gram to kilogram scale are outlined below.
- Charge a reaction vessel with palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and the specific nitro compound in an organic solvent like dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for a duration of 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere chemical yield. The primary driver for cost optimization is the substitution of expensive, pre-functionalized starting materials with commodity chemicals. Nitro compounds are among the most abundant and inexpensive building blocks in the organic chemical industry, available in vast quantities from bulk suppliers. By utilizing these low-cost inputs, manufacturers can drastically reduce the bill of materials (BOM) for the final API intermediate, creating a significant margin advantage in competitive bidding scenarios for generic drug production.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide gas cylinders removes the need for costly safety infrastructure, regular inspections, and specialized gas handling personnel, leading to substantial overhead savings. Additionally, the use of Mo(CO)6 as a stoichiometric CO source allows for precise control over the carbonylation step without the losses associated with gas sparging, ensuring that every mole of reagent contributes effectively to product formation. The high atom economy of the one-pot process further reduces waste disposal costs, as fewer byproducts and solvent volumes are generated compared to multi-step linear syntheses.
- Enhanced Supply Chain Reliability: Relying on widely available nitro compounds and commercially off-the-shelf catalysts like PdCl2 and dppp mitigates the risk of supply disruptions often associated with custom-synthesized precursors. The robustness of the reaction conditions, which tolerate a wide array of functional groups including halogens and trifluoromethyl groups, means that supply chains are less vulnerable to quality fluctuations in raw materials. This flexibility allows procurement teams to source materials from multiple vendors without compromising the integrity of the final synthesis, ensuring continuous production flow.
- Scalability and Environmental Compliance: The transition from batch to continuous flow or larger batch sizes is facilitated by the homogeneous nature of the reaction mixture and the absence of gaseous reagents that require complex mass transfer engineering. The process operates in common organic solvents like dioxane, which are well-understood in terms of recovery and recycling, supporting sustainability goals. Furthermore, the high yields reported in the patent data, reaching up to 96% for certain substrates, minimize the environmental footprint per kilogram of product, aligning with increasingly stringent global regulations on pharmaceutical manufacturing emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for R&D teams evaluating this route for process development. Understanding these nuances is essential for assessing the feasibility of integrating this chemistry into existing manufacturing pipelines.
Q: What are the key advantages of using nitro compounds in this synthesis?
A: Nitro compounds serve as inexpensive and readily available starting materials that replace costly pre-activated substrates. In this patented process, they undergo in-situ reduction, streamlining the supply chain and reducing raw material costs significantly compared to traditional amine-based routes.
Q: How does this method improve safety regarding carbon monoxide usage?
A: Unlike conventional methods requiring high-pressure carbon monoxide gas cylinders which pose severe safety risks, this protocol utilizes solid Mo(CO)6 as a safe CO surrogate. This eliminates the need for specialized high-pressure equipment and mitigates the hazards associated with handling toxic CO gas.
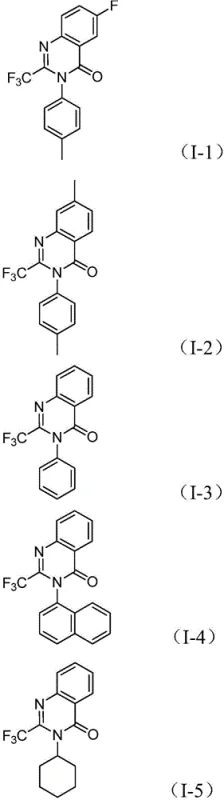
Q: What is the substrate scope for the R2 group in the quinazolinone structure?
A: The method demonstrates excellent functional group tolerance, accommodating a wide range of R2 groups including substituted phenyl rings (with methyl, methoxy, halogen, or trifluoromethyl groups), naphthyl groups, cyclohexyl, and alkyl chains, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development timelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory results translate seamlessly into industrial reality. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay.
We invite potential partners to engage with our technical procurement team to discuss how this innovative palladium-catalyzed route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this efficient one-pot process. We encourage you to contact us today to obtain specific COA data for our catalog compounds and to receive comprehensive route feasibility assessments for your custom synthesis requirements.
