Advanced Manufacturing of Anastrozole Intermediates via Optimized Bromination and N-Alkylation
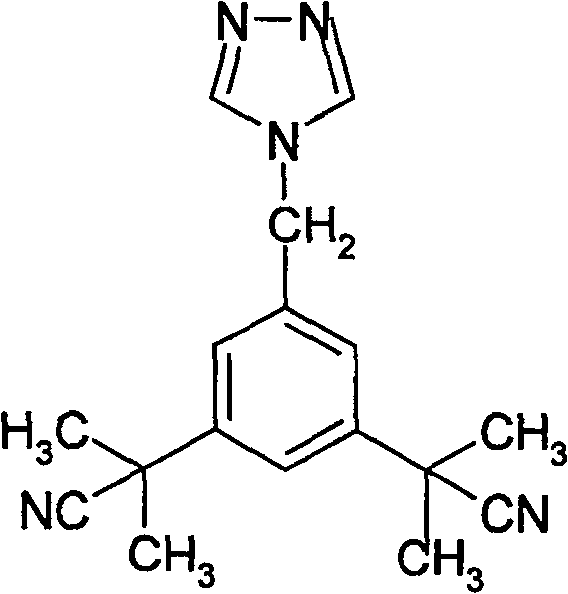
The pharmaceutical industry continuously seeks robust synthetic pathways for critical oncology therapeutics, and the production of Anastrozole (1-[3,5-bis(2,2-dimethyl)ethylcyano]benzyltriazole) stands as a prime example of process optimization. As detailed in patent CN100564365C, a novel preparation method has been developed that fundamentally restructures the synthetic approach to this potent non-steroidal aromatase inhibitor used for treating postmenopausal breast cancer. This technical breakthrough shifts the paradigm from lengthy, low-yield multi-step sequences to a highly efficient two-step protocol starting from the readily accessible precursor 3,5-bis[(2,2-dimethyl)cyanomethyl]toluene. By leveraging advanced phase transfer catalysis and optimized radical bromination, this methodology not only simplifies the operational workflow but also drastically enhances the purity profile of the final active pharmaceutical ingredient (API). For global procurement teams and R&D directors, understanding the nuances of this patented route is essential for securing a reliable API intermediate supplier capable of delivering high-quality materials with consistent batch-to-batch reproducibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for Anastrozole have been plagued by significant inefficiencies that hinder cost-effective mass production. Early methodologies, such as those described in EP0296749 and USP4935437, often relied on starting materials like 3,5-dibromomethyltoluene or methyl 3,5-dimethylbenzoate, necessitating cumbersome sequences involving cyanation, methylation, and reduction steps. Specifically, one prominent prior art route required six distinct reaction steps, utilizing expensive reagents like sodium borohydride and suffering from overall yields typically below 50%. Furthermore, these conventional processes frequently demanded harsh reaction conditions, such as high-temperature reflux in xylene, which promoted the formation of difficult-to-remove side products. Perhaps most critically for industrial application, the purification of the final product in these older methods often relied on column chromatography, a technique that is notoriously difficult to scale and economically prohibitive for tonnage-level manufacturing. Another alternative route developed by academic institutions utilized phase transfer catalysis but failed to achieve sufficient purity, with reported impurity levels exceeding 1%, thereby necessitating multiple recrystallizations that further eroded yield and operational efficiency.
The Novel Approach
In stark contrast to these legacy methods, the innovative process outlined in CN100564365C introduces a streamlined strategy that maximizes atom economy and operational simplicity. The core of this advancement lies in the selection of 3,5-bis[(2,2-dimethyl)cyanomethyl]toluene as the strategic starting point, which allows for the direct installation of the bromomethyl functionality followed by immediate coupling with the triazole ring. This approach condenses the synthesis into just two primary operations: a radical bromination to generate the key intermediate 3,5-bis[(2,2-dimethyl)cyanomethyl]bromotoluene, followed by an N-alkylation with 1,2,4-triazole. The elimination of redundant functional group transformations significantly reduces solvent consumption and waste generation. Moreover, the final isolation procedure abandons column chromatography in favor of a straightforward acid-base workup involving hydrochloric acid dissolution and subsequent basification, which effectively precipitates the product in high purity. This structural simplification of the synthetic tree represents a major leap forward in cost reduction in pharmaceutical manufacturing, enabling producers to achieve superior quality standards without the burden of complex downstream processing.
![Novel 2-step synthesis route for Anastrozole starting from 3,5-bis[(2,2-dimethyl)cyanomethyl]toluene](/insights/img/anastrozole-synthesis-pharma-supplier-20260304134444-05.webp)
Mechanistic Insights into Phase Transfer Catalyzed N-Alkylation
The success of this synthetic route hinges on the precise control of the N-alkylation step, where the bromo-intermediate reacts with 1,2,4-triazole under phase transfer catalysis (PTC) conditions. In this critical transformation, the use of solvents such as dichloromethane or ethyl acetate creates a biphasic system where the inorganic base and the organic substrate reside in different phases. The addition of quaternary ammonium salts, specifically tetrabutylammonium bromide, tetrabutylammonium iodide, or tetrapropylammonium bromide, serves as the molecular shuttle that transports the deprotonated triazole anion into the organic phase. This mechanism dramatically accelerates the reaction kinetics, allowing the process to proceed efficiently at mild temperatures ranging from 20°C to 40°C within a timeframe of 6 to 10 hours. Maintaining these lower temperatures is mechanistically vital, as it suppresses the activation energy for unwanted side reactions and prevents the thermal degradation of sensitive functional groups. The choice of solvent is equally critical; unlike dimethylformamide (DMF) used in older protocols which requires extensive removal, dichloromethane and ethyl acetate are easily removed via concentration, facilitating a cleaner isolation of the crude product.
A deeper analysis of the reaction pathway reveals specific challenges regarding impurity formation, particularly the generation of isomeric byproducts. The patent identifies a specific structural impurity, α,α,α’,α’-tetramethyl-5-(1H-1,2,4-triazol-1-ylmethyl)-1,3-benzenediacetonitrile isomer, which can arise if the starting 1,2,4-triazole contains traces of the 1,3,4-isomer or if molecular isomerization occurs during the reaction. To mitigate this, the process mandates the use of 1,2,4-triazole with a purity greater than 98%. By strictly controlling the stoichiometry and maintaining the reaction temperature at the lower end of the specified range, the formation of this isomer is kinetically inhibited. Analytical data confirms that this rigorous control strategy limits the content of this specific isomer to less than 0.1% in the final API, a level of purity that is essential for meeting stringent regulatory specifications for oncology drugs. This focus on impurity profiling demonstrates a sophisticated understanding of reaction dynamics that ensures the commercial scale-up of complex pharmaceutical intermediates remains viable and compliant.
How to Synthesize Anastrozole Efficiently
The implementation of this patented methodology offers a clear roadmap for laboratories and production facilities aiming to optimize their Anastrozole supply chain. The process begins with the radical bromination of the toluene derivative using N-bromosuccinimide (NBS) and a radical initiator like benzoyl peroxide, typically conducted in chloroform or carbon tetrachloride under reflux conditions to ensure complete conversion to the bromo-intermediate. Following the isolation of this intermediate, the subsequent N-alkylation is performed by combining the bromo-species with 1,2,4-triazole and anhydrous potassium carbonate in the presence of the phase transfer catalyst. The reaction mixture is stirred at controlled ambient temperatures until TLC analysis indicates completion, usually within 6 to 10 hours. The detailed standardized synthesis steps, including specific reagent ratios, workup procedures, and purification parameters, are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Perform radical bromination of 3,5-bis[(2,2-dimethyl)cyanomethyl]toluene using N-bromosuccinimide (NBS) and benzoyl peroxide in chloroform or carbon tetrachloride under reflux to obtain the bromo-intermediate.
- React the crude bromo-intermediate with 1,2,4-triazole in dichloromethane or ethyl acetate using a quaternary ammonium salt phase transfer catalyst at mild temperatures (20-40°C).
- Execute a simplified acid-base workup involving dissolution in hydrochloric acid, filtration of insolubles, basification to pH 8-9, and recrystallization to isolate high-purity Anastrozole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and enhanced supply reliability. By condensing the synthesis from six steps down to two, manufacturers can significantly reduce the cumulative loss of material that typically occurs at each stage of a multi-step process. Furthermore, the elimination of column chromatography removes a major bottleneck in production throughput, allowing for faster batch turnover times and more predictable delivery schedules. The use of readily available starting materials ensures that the supply chain is not vulnerable to the volatility of niche reagent markets, thereby securing continuity of supply for long-term contracts. Additionally, the mild reaction conditions reduce the energy load on manufacturing facilities, contributing to a more sustainable and cost-effective production model that aligns with modern environmental, social, and governance (ESG) goals.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive reagents and the simplification of purification. Traditional routes often rely on costly reducing agents and require extensive solvent exchanges, whereas this method utilizes common, lower-cost solvents like dichloromethane and ethyl acetate. The avoidance of high-temperature reflux in the final coupling step also lowers energy consumption, while the high selectivity of the reaction minimizes the loss of valuable starting materials to side products. Consequently, the overall cost of goods sold (COGS) is substantially reduced, providing a competitive pricing advantage for the final API without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals that are widely available from multiple global vendors. The starting material, 3,5-bis[(2,2-dimethyl)cyanomethyl]toluene, is stable and easy to source, mitigating the risk of raw material shortages that can plague specialized synthetic routes. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in utility supply or equipment performance, ensuring consistent output even in large-scale production environments. This reliability is crucial for pharmaceutical companies that require guaranteed volumes to meet clinical and commercial demand without interruption.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this route offers significant advantages for scaling operations. The replacement of highly toxic solvents like carbon tetrachloride with chloroform in the bromination step, and the use of milder conditions overall, reduces the hazardous waste burden on the facility. The simplified workup procedure, which relies on filtration and pH adjustment rather than complex chromatographic separation, generates less solvent waste and requires less specialized equipment for disposal. This ease of waste management facilitates smoother regulatory approvals and supports the commercial scale-up of complex pharmaceutical intermediates in facilities with strict environmental compliance mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Anastrozole using this optimized methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this supply source into their existing procurement strategies and quality assurance frameworks.
Q: How does this novel synthesis route improve upon conventional methods for Anastrozole production?
A: Unlike conventional routes that require 6 steps or complex column chromatography purification, this patented method utilizes a direct 2-step sequence starting from a commercially available precursor. It eliminates the need for expensive reducing agents like sodium borohydride and avoids high-temperature reflux conditions that generate significant impurities, resulting in a total impurity profile of less than 0.31%.
Q: What specific measures are taken to control the formation of isomeric impurities during synthesis?
A: The process strictly controls the purity of the 1,2,4-triazole starting material to greater than 98% to prevent contamination with 1,3,4-triazole isomers. Additionally, the N-alkylation reaction is maintained at mild temperatures between 20°C and 40°C, which kinetically favors the desired product and suppresses thermal isomerization, keeping specific isomeric impurities below 0.1%.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the method is specifically designed for industrial scalability. It replaces toxic solvents like carbon tetrachloride with safer alternatives like chloroform in the first step and utilizes mild reaction conditions (20-40°C) in the second step. The workup procedure relies on simple acid-base extraction and filtration rather than labor-intensive column chromatography, significantly enhancing throughput and operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anastrozole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patented synthesis are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of Anastrozole meets the highest international standards for potency and impurity profiles. Our commitment to quality assurance means that we can consistently deliver high-purity pharmaceutical intermediates that support the development and manufacturing of life-saving oncology therapies.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and reliability in your API sourcing strategy.
