Optimizing Anastrozole Production: A Technical Analysis of the AIBN-Catalyzed Route
Optimizing Anastrozole Production: A Technical Analysis of the AIBN-Catalyzed Route
The global demand for high-quality oncology therapeutics continues to drive innovation in the synthesis of critical Active Pharmaceutical Ingredients (APIs). Among these, Anastrozole stands out as a pivotal third-generation non-steroidal aromatase inhibitor, essential for treating advanced breast cancer in postmenopausal patients. Recent advancements in synthetic methodology, specifically detailed in patent CN103554041A, have introduced a transformative approach to manufacturing this vital compound. This technical insight report analyzes the shift from traditional radical bromination methods to a novel AIBN-initiated protocol, highlighting its profound implications for purity, yield, and operational safety. By leveraging this optimized pathway, manufacturers can achieve superior impurity profiles and streamline downstream processing, addressing the rigorous quality standards demanded by modern regulatory bodies.
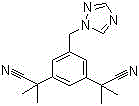
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
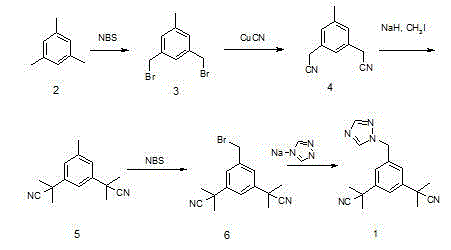
Historically, the industrial synthesis of Anastrozole has relied on a bromination-condensation sequence that, while functional, suffers from significant inefficiencies that impact both cost and quality. The conventional route typically employs benzoyl peroxide (BPO) as the radical initiator for the bromination of the dinitrile precursor. However, this method often struggles with incomplete conversion and the concurrent formation of undesirable dibrominated by-products. These side reactions complicate the purification landscape, frequently resulting in intermediate purities hovering around 75%, which necessitates aggressive and costly purification measures. Furthermore, the subsequent condensation step with 1,2,4-triazole often requires column chromatography to isolate the final API, a technique that is notoriously difficult to scale and economically burdensome for multi-kilogram production runs.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN103554041A introduces a refined strategy that fundamentally alters the reaction kinetics and workup procedures. By substituting benzoyl peroxide with azobisisobutyronitrile (AIBN) as the radical initiator, the process achieves a marked improvement in selectivity, driving the conversion of the starting material, alpha,alpha,alpha',alpha',5-pentamethyl-1,3-diacetonitrile benzene, to the monobrominated intermediate with exceptional fidelity. This modification alone elevates the intermediate purity to over 90%, drastically reducing the burden on downstream purification. Additionally, the integration of phase transfer catalysis (PTC) in the condensation step allows for efficient nucleophilic substitution under mild alkaline conditions, facilitating a simple recrystallization workflow that completely bypasses the need for column chromatography.
Mechanistic Insights into AIBN-Initiated Radical Bromination
The core of this technological advancement lies in the mechanistic superiority of AIBN over traditional peroxide initiators in radical halogenation reactions. AIBN decomposes thermally to generate nitrogen gas and stable cyanoisopropyl radicals, which abstract hydrogen atoms from the benzylic positions of the substrate with high specificity. This controlled radical generation minimizes the occurrence of runaway reactions and excessive bromination, which are common pitfalls when using BPO. The result is a cleaner reaction profile where the desired 3,5-bis[(2,2-dimethyl)cyanomethyl]-bromomethylbenzene is formed predominantly, suppressing the formation of dibromo impurities that are difficult to separate due to their similar physicochemical properties. This selectivity is crucial for maintaining the integrity of the nitrile groups, which are sensitive to harsh radical conditions.
Furthermore, the implementation of phase transfer catalysis in the second step represents a masterclass in process intensification. In this biphasic system, the quaternary ammonium salt, such as tetrabutylammonium bromide (TBAB), acts as a molecular shuttle, transporting the triazolate anion from the aqueous phase into the organic phase where the electrophilic bromide resides. This proximity effect significantly accelerates the SN2 substitution rate without requiring extreme temperatures or anhydrous conditions that could degrade the sensitive nitrile functionalities. The outcome is a robust reaction that tolerates minor variations in water content, making it inherently safer and more reproducible on a commercial scale compared to strictly anhydrous protocols.
How to Synthesize Anastrozole Efficiently
Implementing this optimized synthesis route requires precise control over reaction parameters to maximize the benefits of the AIBN initiation and PTC systems. The process begins with the careful addition of N-bromosuccinimide (NBS) to a refluxing solution of the dinitrile precursor and AIBN in a suitable solvent like acetonitrile or carbon tetrachloride. Maintaining the temperature between 75-80°C is critical to ensure steady radical flux without thermal degradation. Following the isolation of the high-purity brominated intermediate, the subsequent condensation is performed by mixing the intermediate with 1,2,4-triazole, aqueous potassium hydroxide, and the phase transfer catalyst in a solvent such as toluene or dichloroethane. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below to guide process development teams.
- Perform radical bromination of alpha,alpha,alpha',alpha',5-pentamethyl-1,3-diacetonitrile benzene using NBS and AIBN initiator at 75-80°C to achieve high conversion.
- Execute phase transfer catalyzed condensation of the brominated intermediate with 1,2,4-triazole using TBAB and aqueous KOH in an organic solvent.
- Purify the crude product via simple isopropanol recrystallization to obtain high-purity Anastrozole without column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this AIBN-driven synthesis protocol offers substantial strategic advantages beyond mere technical elegance. The elimination of column chromatography is perhaps the most significant economic driver, as this unit operation is a major bottleneck in pharmaceutical manufacturing, consuming vast amounts of silica gel, solvents, and labor hours. By replacing this with a simple crystallization step, the overall processing time is drastically reduced, leading to faster batch turnover and improved asset utilization. Moreover, the higher purity of the intermediate reduces the load on final purification, minimizing solvent consumption and waste generation, which directly translates to lower operational expenditures and a reduced environmental footprint.
- Cost Reduction in Manufacturing: The shift to AIBN initiation and PTC condensation eliminates the need for expensive and time-consuming column chromatography, which is a primary cost driver in the conventional route. This simplification of the purification train significantly lowers solvent usage and waste disposal costs, while the higher yields reduce the cost of goods sold per kilogram of active ingredient. The ability to use simpler equipment and fewer processing steps further decreases capital expenditure requirements for new production lines dedicated to this API.
- Enhanced Supply Chain Reliability: The robustness of the AIBN-initiated reaction ensures consistent batch-to-batch quality, reducing the risk of production failures or off-spec material that can disrupt supply schedules. The use of readily available and stable reagents like AIBN and TBAB mitigates the risk of raw material shortages associated with more specialized or hazardous initiators. This reliability allows for more accurate forecasting and inventory management, ensuring a steady flow of high-purity Anastrozole to meet market demand without unexpected interruptions.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of hazardous purification steps make this process inherently safer and easier to scale from pilot plant to commercial production volumes. The reduction in solvent intensity and waste generation aligns with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. This sustainability profile not only reduces regulatory risk but also enhances the brand value of the final pharmaceutical product in markets that prioritize eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the operational benefits. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for large-scale production.
Q: Why is AIBN preferred over Benzoyl Peroxide for Anastrozole bromination?
A: AIBN provides a more controlled radical initiation compared to Benzoyl Peroxide, significantly reducing the formation of dibrominated by-products and increasing intermediate purity from approximately 75% to over 90%.
Q: How does Phase Transfer Catalysis improve the condensation step?
A: Using Phase Transfer Catalysts like TBAB allows the reaction to proceed efficiently in a biphasic system, eliminating the need for complex column chromatography purification and enabling simple recrystallization instead.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the mild reaction conditions (75-80°C), absence of hazardous purification steps, and high yields make this method highly scalable and compliant with industrial safety and environmental standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anastrozole Supplier
The technical potential of the AIBN-catalyzed synthesis route for Anastrozole represents a significant leap forward in the manufacturing of this critical oncology drug. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovative processes to life. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of Anastrozole we produce adheres to the highest international standards. We are committed to translating complex laboratory breakthroughs into reliable, industrial-scale realities that support the global healthcare supply chain.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this optimized route can enhance your supply security and cost efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in the production of high-value pharmaceutical intermediates and APIs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
