Advanced Synthesis of Bioactive Indolizine Diarylmethane Derivatives for Pharmaceutical Applications
Advanced Synthesis of Bioactive Indolizine Diarylmethane Derivatives for Pharmaceutical Applications
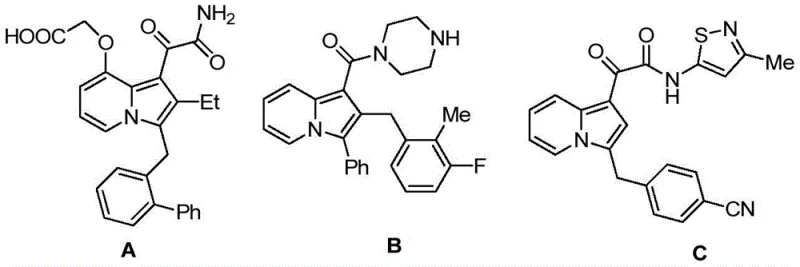
The pharmaceutical landscape is constantly evolving, driven by the need for novel scaffolds that offer enhanced biological profiles and streamlined synthetic accessibility. Patent CN114163436A introduces a significant breakthrough in this domain by disclosing a series of diarylmethane derivatives containing an indolizine core. These molecules are not merely structural curiosities; they represent a class of compounds with potent dual biological activities, specifically targeting antibacterial and anticancer pathways. Unlike previous iterations of indolizine-containing molecules which often suffered from limited activity spectra, such as the singular secreted phospholipase sPLA2 inhibitory activity seen in earlier compounds, these new derivatives offer a broader therapeutic potential. The structural versatility allows for extensive modification at multiple positions, enabling medicinal chemists to fine-tune pharmacokinetic properties while maintaining high potency. This technological advancement provides a robust foundation for developing next-generation therapeutic agents, positioning these intermediates as critical assets for R&D teams focused on oncology and infectious disease portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of complex heterocyclic systems like indolizine-fused diarylmethanes has been a formidable challenge in organic synthesis. Conventional routes often rely on multi-step sequences that involve harsh reaction conditions, expensive reagents, and tedious purification protocols. Many existing methods struggle with regioselectivity issues, leading to complex mixtures of isomers that are difficult to separate, thereby drastically reducing the overall yield and purity of the final product. Furthermore, the reliance on pre-functionalized starting materials that are not commercially available adds significant lead time and cost to the development process. The environmental footprint of these older methodologies is also a concern, as they frequently generate substantial amounts of hazardous waste due to low atom economy and the use of stoichiometric amounts of toxic heavy metals. For procurement and supply chain managers, these inefficiencies translate into volatile pricing, unreliable delivery schedules, and increased regulatory burden regarding waste disposal and solvent recovery.
The Novel Approach
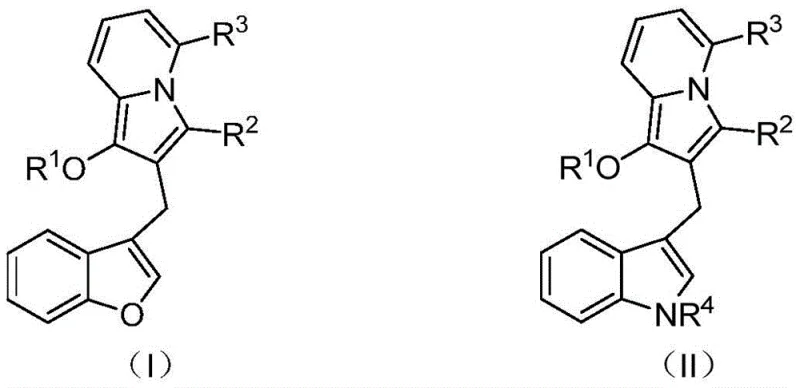
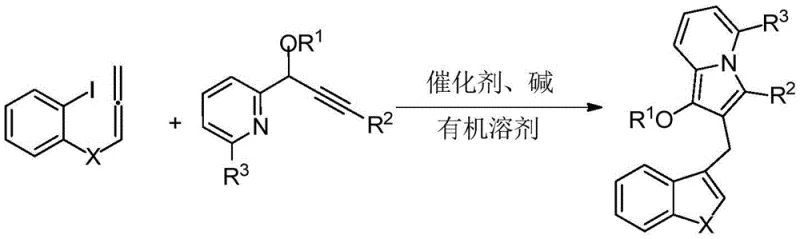
In stark contrast, the methodology described in patent CN114163436A offers a paradigm shift through a highly efficient, one-pot palladium-catalyzed cascade cyclization. This novel approach leverages the reactivity of 2-iodophenyl allene compounds and propargyl pyridine derivatives to construct the indolizine core and the diarylmethane backbone simultaneously. The reaction proceeds under relatively mild conditions, typically between 60-90°C, using common organic solvents like N,N-dimethylformamide. This telescoped strategy eliminates the need for isolating unstable intermediates, significantly simplifying the operational workflow. The structural diversity achievable through this method is impressive, as evidenced by the general formulas (I) and (II), where various acyl groups, substituted phenyl rings, and alkyl chains can be introduced with high fidelity. This flexibility allows for the rapid generation of analog libraries, accelerating the hit-to-lead optimization process for drug discovery teams seeking reliable pharmaceutical intermediate suppliers.
Mechanistic Insights into Pd-Catalyzed Cascade Cyclization
The success of this synthesis hinges on a sophisticated palladium-catalyzed mechanism that orchestrates multiple bond-forming events in a single operation. The reaction initiates with the oxidative addition of the palladium(0) catalyst to the carbon-iodine bond of the 2-iodophenyl allene substrate, generating a reactive aryl-palladium species. This intermediate then undergoes a migratory insertion into the allene moiety, forming a new carbon-carbon bond and setting the stage for the subsequent cyclization. The presence of the propargyl pyridine component is crucial, as it acts as a nucleophile that intercepts the palladium-allyl intermediate, facilitating the formation of the indolizine ring system. The precise control over the catalytic cycle ensures that the reaction proceeds with high regioselectivity, minimizing the formation of byproducts. Understanding this mechanistic pathway is vital for R&D directors, as it highlights the importance of ligand selection and catalyst loading in optimizing the reaction efficiency. The use of tetrakis(triphenylphosphine)palladium, as preferred in the examples, provides the necessary steric and electronic environment to drive the cascade to completion.
Impurity control is another critical aspect addressed by this mechanistic design. The concerted nature of the cascade reaction reduces the likelihood of side reactions that typically plague stepwise syntheses, such as polymerization of the allene or alkyne components. Furthermore, the choice of base, such as potassium carbonate or cesium carbonate, plays a pivotal role in neutralizing acidic byproducts and regenerating the active catalyst species. The reaction conditions are optimized to suppress competing pathways, ensuring that the desired diarylmethane scaffold is formed with high purity. This inherent selectivity translates to simpler downstream processing, as the crude reaction mixtures contain fewer impurities that require removal. For quality control teams, this means more consistent batch-to-batch reproducibility and easier compliance with stringent purity specifications required for pharmaceutical applications. The ability to tune the R groups on the starting materials allows for further optimization of the electronic properties of the substrates, enhancing the overall robustness of the synthetic route.
How to Synthesize Indolizine Diarylmethane Derivatives Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins with the careful weighing and mixing of the 2-iodophenyl allene compound and the propargyl pyridine compound in a molar ratio ranging from 1:1 to 1:2. A palladium catalyst, typically used at a loading of 2-20 mol%, is added along with an inorganic base such as potassium carbonate. The reaction is conducted in an organic solvent like DMF or toluene under an inert nitrogen atmosphere to prevent catalyst deactivation by oxygen. Heating the mixture to 80°C for approximately 6 hours drives the reaction to completion, as monitored by TLC or HPLC. Upon cooling, the reaction mixture is subjected to a standard workup procedure involving aqueous extraction, drying, and solvent removal. The resulting crude product is then purified by column chromatography using a petroleum ether and ethyl acetate gradient to afford the target indolizine derivative in high yield. Detailed standardized synthesis steps are provided in the guide below.
- Mix 2-iodophenyl allene compound, propargyl pyridine compound, palladium catalyst (e.g., Pd(PPh3)4), alkali (e.g., K2CO3), and organic solvent (e.g., DMF) in a reaction vessel.
- Stir the reaction mixture under an inert atmosphere (nitrogen) at a temperature between 60-90°C for 4-12 hours to facilitate the cascade cyclization.
- Perform post-treatment including extraction with ethyl acetate, drying over anhydrous sodium sulfate, filtration, solvent removal, and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial benefits that directly address the pain points of modern chemical procurement and supply chain management. The shift from multi-step, low-yielding processes to a concise, one-pot cascade reaction represents a significant leap in manufacturing efficiency. This consolidation of steps not only reduces the consumption of raw materials and solvents but also minimizes the labor and energy costs associated with prolonged reaction times and multiple isolation procedures. For procurement managers, this translates into a more favorable cost structure, allowing for competitive pricing without compromising on quality. The use of readily available starting materials further mitigates supply risk, ensuring a stable and continuous flow of intermediates even in volatile market conditions. The robustness of the reaction conditions means that the process is less susceptible to minor variations in parameters, leading to higher reliability in production scheduling.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps and the use of catalytic amounts of palladium significantly lower the overall production costs. By avoiding the need for expensive protecting group strategies and harsh reagents, the process achieves substantial cost savings in raw material procurement. The high atom economy of the cascade reaction ensures that a greater proportion of the starting materials ends up in the final product, reducing waste disposal costs. Additionally, the simplified purification protocol reduces the consumption of chromatography media and solvents, further driving down the cost of goods sold. These efficiencies allow for a more competitive pricing model, making high-purity pharmaceutical intermediates more accessible for large-scale drug manufacturing.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as 2-iodophenyl allenes and propargyl pyridines ensures a resilient supply chain. These precursors are synthesized from bulk petrochemical feedstocks, meaning their availability is not subject to the bottlenecks often seen with exotic or highly specialized reagents. The mild reaction conditions (60-90°C) allow the process to be run in a wide range of manufacturing facilities without requiring specialized high-pressure or cryogenic equipment. This flexibility enables diversified sourcing strategies, reducing the risk of supply disruptions caused by single-source dependencies. Furthermore, the scalability of the process from gram to kilogram scales ensures that supply can be rapidly ramped up to meet increasing demand from clinical trials or commercial launches.
- Scalability and Environmental Compliance: The process is inherently green, aligning with the growing emphasis on sustainable manufacturing practices in the chemical industry. The reduction in solvent usage and waste generation simplifies compliance with environmental regulations, lowering the administrative burden on EHS teams. The ability to run the reaction in common solvents like DMF or toluene facilitates solvent recovery and recycling, contributing to a circular economy approach. The high yields and selectivity minimize the formation of hazardous byproducts, reducing the complexity of waste treatment. This environmental stewardship not only protects the planet but also enhances the corporate reputation of companies adopting this technology, appealing to eco-conscious investors and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these indolizine derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for making informed decisions about integrating these intermediates into your development pipeline. Whether you are concerned about regulatory compliance, technical feasibility, or supply logistics, these insights provide a solid foundation for your strategic planning.
Q: What are the primary biological activities of these indolizine derivatives?
A: According to patent CN114163436A, these compounds exhibit significant antibacterial and anticancer activities, overcoming the single-activity limitation of prior art compounds like sPLA2 inhibitors.
Q: What catalyst system is required for this synthesis?
A: The process utilizes a palladium catalyst system, specifically preferring tetrakis(triphenylphosphine)palladium, combined with inorganic bases like potassium carbonate in polar aprotic solvents.
Q: Is this method scalable for industrial production?
A: Yes, the method uses commercially available raw materials and standard reaction conditions (60-90°C), making it highly suitable for commercial scale-up without requiring extreme pressures or cryogenic temperatures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolizine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in patent CN114163436A for the development of novel therapeutics. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory methodology into robust, commercial-scale manufacturing processes. Our team of experienced chemists is adept at optimizing palladium-catalyzed reactions, ensuring that the transition from benchtop to pilot plant is seamless and efficient. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch meets stringent purity specifications, providing you with the confidence to move forward with your drug development programs.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. By partnering with us, you gain access to our deep reservoir of process knowledge and our commitment to excellence in every aspect of chemical manufacturing. Please contact us today to request specific COA data and route feasibility assessments for these high-value indolizine diarylmethane derivatives. Let us help you accelerate your path to market with reliable, high-quality intermediates that drive innovation in the pharmaceutical industry.
