Advanced Metal-Free Synthesis of 3-Quinolinyl-5-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-rich heterocycles, particularly the 1,2,4-triazole scaffold, which serves as a critical pharmacophore in numerous bioactive molecules. Patent CN113307790B introduces a groundbreaking preparation method for 3-quinolinyl-5-trifluoromethyl substituted 1,2,4-triazole compounds that addresses long-standing inefficiencies in synthetic organic chemistry. This innovation leverages a metal-free oxidative cyclization strategy utilizing tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP) to forge the triazole ring directly from readily available 2-methylquinoline derivatives. By eliminating the need for expensive transition metal catalysts and严苛 reaction conditions, this technology offers a streamlined pathway for producing high-value intermediates used in drug discovery and functional material science.
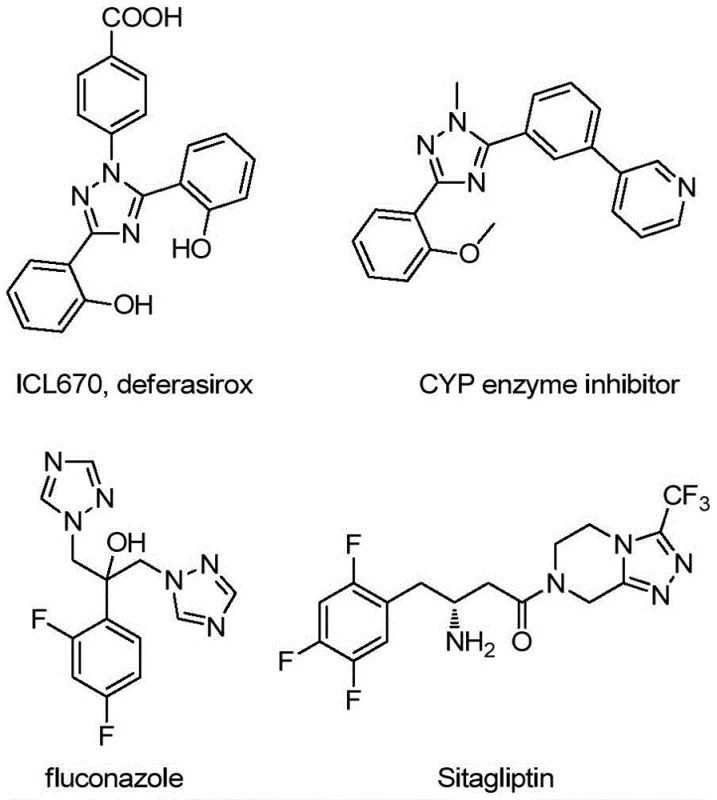
The significance of this structural motif cannot be overstated, as 1,2,4-triazoles are ubiquitous in medicinal chemistry, appearing in antifungal agents, enzyme inhibitors, and anticancer drugs. The ability to efficiently append a quinoline moiety alongside a trifluoromethyl group enhances the lipophilicity and metabolic stability of the resulting molecules, making them prime candidates for lead optimization. For R&D directors and process chemists, the availability of a reliable synthesis route for these complex heterocycles is essential for accelerating pipeline development. This patent provides not just a theoretical framework but a practical, experimentally validated protocol that has demonstrated exceptional substrate scope and functional group tolerance across fifteen distinct examples.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by operational complexity and poor atom economy. Traditional literature methods typically rely on quinoline-2-carboxylic acid as the starting material, necessitating a tedious five-step synthetic sequence to arrive at the desired triazole core. This multi-step approach suffers from a dismal cumulative yield of approximately 17%, rendering it economically unviable for large-scale applications. Furthermore, these conventional routes often demand severe reaction conditions, including the use of hazardous reagents and strict environmental controls, which escalate both the safety risks and the operational costs associated with manufacturing. The reliance on such inefficient pathways creates a bottleneck in the supply chain, limiting the availability of these critical intermediates for downstream drug development programs.
The Novel Approach
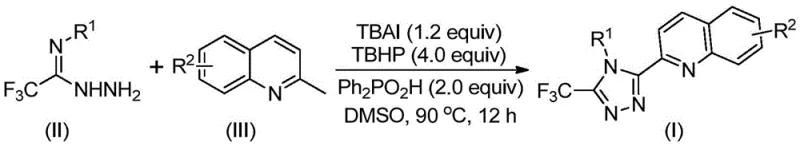
In stark contrast, the methodology disclosed in CN113307790B represents a paradigm shift towards green and efficient synthesis. By utilizing 2-methylquinoline and trifluoroethylimide hydrazide as starting materials, the process condenses what was once a five-step ordeal into a single, direct oxidative cyclization event. The reaction is promoted by a catalytic system comprising TBAI and TBHP, with diphenylphosphoric acid serving as a key additive to facilitate the transformation. This novel approach operates under mild thermal conditions (80-100°C) in common solvents like DMSO, completely bypassing the need for anhydrous or oxygen-free environments. The result is a dramatic improvement in efficiency, with isolated yields reaching as high as 97% for certain substrates, thereby offering a cost-effective and scalable solution for the production of high-purity pharmaceutical intermediates.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The mechanistic elegance of this transformation lies in its ability to generate reactive intermediates in situ without the aid of precious metals. The reaction initiates with the oxidation of the methyl group on the 2-methylquinoline substrate to a reactive aldehyde species, mediated by the TBAI/TBHP system. This generated aldehyde then undergoes a condensation reaction with the trifluoroethylimide hydrazide to form a dehydrated hydrazone intermediate. Subsequent oxidative iodination activates the hydrazone, triggering an intramolecular electrophilic substitution that closes the triazole ring. Finally, an aromatization step yields the stable 3-quinolinyl-5-trifluoromethyl-1,2,4-triazole product. This cascade sequence is highly efficient, minimizing the formation of byproducts and ensuring that the majority of the starting material is converted into the desired target molecule.
From an impurity control perspective, the absence of heavy metal catalysts is a significant advantage for pharmaceutical manufacturing. Transition metal residues are a major concern in API production, often requiring costly and time-consuming purification steps to meet regulatory limits (e.g., ICH Q3D guidelines). By employing an organocatalytic system based on iodine and peroxide, this method inherently produces a cleaner crude reaction profile. The primary impurities are likely derived from over-oxidation or incomplete cyclization, which are easily managed through standard work-up procedures such as silica gel filtration and column chromatography. This simplified purification workflow not only reduces the consumption of chromatography media but also shortens the overall production cycle time, enhancing the throughput of the manufacturing facility.
How to Synthesize 3-Quinolinyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
The execution of this synthesis is straightforward and amenable to standard laboratory equipment, making it accessible for both process development and production teams. The protocol involves charging a reaction vessel with the requisite amounts of tetrabutylammonium iodide, aqueous tert-butyl peroxide, diphenylphosphoric acid, trifluoroethylimide hydrazide, and the specific 2-methylquinoline derivative in DMSO. The mixture is then heated to a temperature range of 80-100°C and maintained for 8 to 14 hours. Upon completion, the reaction mixture is cooled, filtered, and subjected to standard purification techniques. The detailed standardized synthesis steps for specific derivatives are outlined in the guide below.
- Mix tetrabutylammonium iodide, tert-butyl peroxide, diphenylphosphoric acid, trifluoroethylimide hydrazide, and 2-methylquinoline in DMSO.
- Heat the reaction mixture to 80-100°C and stir for 8-14 hours under air atmosphere without strict anhydrous conditions.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical yield. The shift from a multi-step, low-yield process to a one-pot, high-yield reaction fundamentally alters the cost structure of the intermediate. By drastically reducing the number of unit operations and eliminating the need for exotic catalysts, the overall manufacturing cost is significantly lowered. This cost efficiency allows for more competitive pricing strategies when sourcing these intermediates, providing a buffer against raw material price volatility. Furthermore, the use of commodity chemicals like TBAI and TBHP ensures a stable and reliable supply chain, as these reagents are produced globally in vast quantities and are not subject to the geopolitical constraints often associated with rare earth metals or specialized ligands.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive metal scavenging resins and complex waste treatment protocols required to lower metal content to ppm levels. Additionally, the high atom economy and single-step nature of the reaction reduce solvent consumption and energy usage per kilogram of product. These factors collectively contribute to substantial cost savings in the manufacturing process, allowing for better margin management and resource allocation within the production budget.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically the tolerance to air and moisture, means that production does not require specialized inert atmosphere reactors or rigorous drying of solvents. This operational simplicity reduces the risk of batch failures due to environmental excursions and allows for faster turnaround times between batches. Consequently, lead times for high-purity pharmaceutical intermediates can be reduced, ensuring a continuous flow of materials to downstream formulation teams and mitigating the risk of stockouts.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively on gram scales with potential for expansion to multi-ton production. The avoidance of heavy metals aligns with increasingly stringent environmental regulations regarding industrial effluent and waste disposal. By generating less hazardous waste and utilizing greener reagents, this method supports corporate sustainability goals and simplifies the regulatory approval process for new manufacturing sites, facilitating rapid capacity expansion to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this process for integration into their supply chains.
Q: What are the key advantages of this synthesis method over traditional routes?
A: Unlike traditional methods requiring quinoline-2-carboxylic acid and five steps with only 17% yield, this patent describes a one-pot oxidative cyclization using cheap 2-methylquinoline. It achieves significantly higher yields (up to 97%), avoids toxic heavy metal catalysts, and does not require stringent anhydrous or oxygen-free conditions, simplifying operations.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is highly scalable. The use of inexpensive, commercially available reagents like TBAI and TBHP, combined with the tolerance for air and moisture, eliminates the need for specialized inert atmosphere equipment. This robustness facilitates the transition from gram-scale laboratory synthesis to multi-ton commercial production.
Q: What is the role of diphenylphosphoric acid in the reaction?
A: Diphenylphosphoric acid acts as a crucial additive that promotes the oxidative cyclization efficiency. Its presence, alongside the TBAI/TBHP system, helps drive the conversion of the intermediate hydrazone into the final aromatic triazole ring, ensuring high conversion rates and minimizing side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolinyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN113307790B and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. Our state-of-the-art facilities are designed to handle complex heterocyclic chemistry with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards.
We invite you to collaborate with us to leverage this advanced synthesis method for your specific drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to obtain specific COA data for our pilot batches and to discuss route feasibility assessments that can optimize your supply chain for these valuable 1,2,4-trazole intermediates.
