Advanced Catalytic Oxidation Route for High-Purity Zolpidem Manufacturing
Introduction to Patent CN114591320A
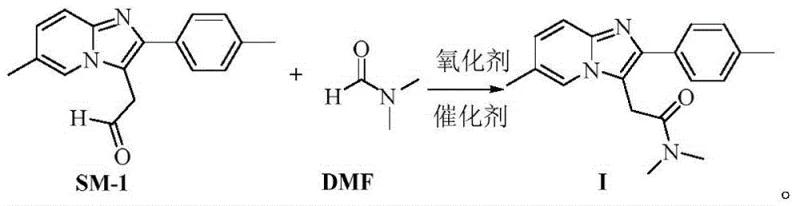
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for the synthesis of critical central nervous system (CNS) agents. Patent CN114591320A, published in June 2022, introduces a groundbreaking preparation method for Zolpidem, a widely prescribed non-benzodiazepine hypnotic agent marketed under trade names such as Stilnox®. This intellectual property details a novel catalytic oxidative amidation strategy that fundamentally shifts the synthetic paradigm away from hazardous traditional reagents. By utilizing 2-(6-methyl-2-(p-tolyl)imidazo[1,2-a]pyridin-3-yl)acetaldehyde as the key starting material and leveraging N,N-dimethylformamide (DMF) as a dual-purpose solvent and amine source, the inventors have achieved a process that is not only operationally safer but also chemically superior. The significance of this patent lies in its ability to bypass the use of transition metal catalysts and toxic alkylating agents, thereby addressing major pain points in modern pharmaceutical intermediates manufacturing regarding regulatory compliance and environmental sustainability.
The core innovation described in this patent involves a direct oxidative coupling reaction that constructs the acetamide side chain in a single pot. Unlike legacy processes that require multiple isolation steps and the handling of unstable intermediates, this method streamlines the synthesis into a concise sequence. The reaction employs organic peroxides as oxidants and tetrabutylammonium iodide as a phase-transfer catalyst, creating a robust system that tolerates a variety of solvent conditions. For global specialty chemical buyers and production managers, this represents a tangible opportunity to optimize their supply chains. The method's compatibility with standard industrial equipment and its reliance on commodity chemicals suggest a high degree of feasibility for immediate technology transfer and commercial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Zolpidem has been plagued by significant safety and efficiency challenges that hinder scalable commercial scale-up of complex pharmaceutical intermediates. Traditional routes, such as those described in earlier literature involving p-methyl-beta-nitrostyrene, often necessitate the use of phosphorus pentachloride and pyridine, reagents known for their corrosivity and irritating nature. More critically, other established pathways rely on Mannich reactions involving genotoxic formaldehyde and subsequent nucleophilic substitutions using sodium cyanide and iodomethane. The handling of sodium cyanide poses severe occupational health risks and requires stringent waste management protocols to prevent environmental contamination. Furthermore, the use of carbonyldiimidazole (CDI) for the final amidation step in some processes introduces high costs and allergenic risks, while the decomposition products of CDI can contaminate the final API, complicating purification efforts.
Another prevalent conventional approach involves the use of glyoxylate esters and noble metal catalysts for reduction steps. These methods suffer from the requirement of expensive precious metals that must be rigorously removed to meet residual metal specifications in the final drug product. The operational complexity is further exacerbated by the need for dry hydrogen chloride gas introduction under heating conditions, a procedure that demands specialized corrosion-resistant reactors and poses significant safety hazards to plant personnel. Additionally, the reliance on flammable dimethylamine gas, which has a low boiling point of 7°C, creates logistical nightmares for storage and feeding systems, increasing the risk of leaks and explosions in a manufacturing setting. These cumulative factors result in prolonged production cycles, elevated capital expenditure for safety infrastructure, and inconsistent batch quality.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach disclosed in CN114591320A offers a streamlined and green alternative that directly addresses the aforementioned deficiencies. By employing 2-(6-methyl-2-(p-tolyl)imidazo[1,2-a]pyridin-3-yl)acetaldehyde (SM-1) as the substrate, the process eliminates the need for dangerous cyanide substitutions or high-pressure hydrogenation steps. The use of DMF as the dimethylamine source is particularly ingenious; it serves as a stable, liquid reagent that is easy to handle and dose compared to gaseous dimethylamine. The reaction proceeds via a catalytic oxidative mechanism using readily available organic peroxides such as dicumyl peroxide or tert-butyl peroxide, which are safer and more cost-effective than stoichiometric coupling reagents like CDI.
This new methodology significantly shortens the synthetic route, effectively collapsing multiple transformation steps into a single reaction vessel. The absence of transition metals removes the necessity for expensive scavenging resins or complex filtration units designed to lower metal residues below ppm levels. Moreover, the reaction conditions are mild, typically operating between 85°C and 110°C in solvents like 4-methyl-2-pentanone, which are standard in fine chemical processing. The workup procedure is equally simplified, involving a straightforward quench with aqueous sulfite solutions to neutralize excess oxidant, followed by extraction and recrystallization. This simplicity translates directly into reduced operational expenditure (OPEX) and a smaller environmental footprint, making it an ideal candidate for cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Catalytic Oxidative Amidation
The chemical elegance of this process lies in the oxidative amidation mechanism, where the aldehyde functionality of the starting material is directly converted into the corresponding amide using DMF. In the presence of the tetrabutylammonium iodide (TBAI) catalyst and a peroxide oxidant, the reaction likely proceeds through the generation of an reactive iodine species or a radical intermediate that activates the aldehyde carbonyl. The DMF molecule acts as a nucleophile, attacking the activated carbonyl center to form a hemiaminal-like intermediate, which is subsequently oxidized to the stable amide bond found in Zolpidem. This mechanism avoids the formation of unstable acid chlorides or activated esters that are typical in classical amidation chemistry. The choice of oxidant is critical; peroxides like dicumyl peroxide provide the necessary oxygen atom for the transformation while generating benign byproducts that are easily removed during the aqueous workup phase.
From an impurity control perspective, this mechanism offers distinct advantages over traditional alkylation routes. Conventional methods often generate N-methylated byproducts or over-alkylated impurities due to the high reactivity of methylating agents like iodomethane. In the oxidative amidation pathway, the nitrogen source is fixed within the DMF structure, inherently limiting the potential for uncontrolled alkylation side reactions. Furthermore, the absence of cyanide ions prevents the formation of nitrile intermediates that can hydrolyze into carboxylic acid impurities or persist as genotoxic residuals. The use of a phase-transfer catalyst like TBAI ensures homogeneous reaction conditions, minimizing localized hot spots that could lead to thermal degradation of the sensitive imidazopyridine core. Consequently, the crude product obtained from this reaction exhibits a remarkably clean profile, facilitating the achievement of high-purity specifications (>99%) with minimal downstream processing effort.
How to Synthesize Zolpidem Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and temperature control to maximize yield and minimize side products. The patent specifies a molar ratio of SM-1 to DMF ranging from 1:8.0 to 1:20.0, with a preferred ratio of 1:14.0, ensuring that DMF acts effectively as both reactant and solvent medium. The oxidant is typically added in a molar excess (1:4.0 to 1:10.0 relative to SM-1) to drive the reaction to completion. The detailed standardized synthesis steps, including precise addition rates, stirring speeds, and crystallization parameters, are outlined below to guide process engineers in replicating this high-efficiency protocol.
- Combine the acetaldehyde starting material (SM-1), N,N-dimethylformamide (DMF), tetrabutylammonium iodide (TBAI) catalyst, and a peroxide oxidant in an organic solvent such as 4-methyl-2-pentanone.
- Heat the reaction mixture to a temperature range of 85-110°C, preferably 105-110°C, and maintain until the conversion is complete as monitored by HPLC.
- Quench the reaction with a reducing solution like saturated sodium sulfite, separate the organic layer, extract, concentrate, and recrystallize from ethyl acetate to obtain high-purity Zolpidem.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers compelling strategic benefits that extend beyond simple chemical yield. The shift away from controlled substances and highly regulated toxic reagents simplifies the procurement landscape significantly. Sourcing DMF and organic peroxides is far less bureaucratic and risky than managing the supply chain for sodium cyanide or gaseous amines, which often require special permits and dedicated transport logistics. This reduction in regulatory friction translates to a more resilient supply chain that is less susceptible to disruptions caused by environmental inspections or transportation bans on hazardous materials.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the substitution of expensive reagents with commodity chemicals. By eliminating the need for carbonyldiimidazole (CDI), noble metal catalysts, and specialized alkylating agents, the raw material cost per kilogram of Zolpidem is drastically reduced. Additionally, the simplified workup procedure reduces the consumption of solvents and utilities associated with complex purification steps like vacuum distillation or column chromatography. The removal of transition metals also negates the cost of metal scavengers and the analytical testing required to certify low metal content, further lowering the overall cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: The reliance on stable, liquid reagents like DMF and solid peroxides enhances the reliability of the supply chain. Unlike gaseous dimethylamine, which requires pressurized cylinders and careful leak monitoring, DMF can be stored in standard tanks and pumped easily, reducing the risk of supply interruptions due to cylinder shortages or delivery delays. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in raw material quality, ensuring consistent batch-to-batch performance. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior, facilitating easier regulatory approval for new manufacturing sites. The absence of genotoxic reagents simplifies the cleaning validation process and reduces the burden on wastewater treatment facilities. The process generates less hazardous waste, aligning with modern green chemistry principles and corporate sustainability goals. The scalability is proven by the use of standard solvents and moderate temperatures, allowing for seamless transfer from pilot plant to multi-ton commercial production without the need for exotic high-pressure or cryogenic reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative amidation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational feasibility and quality advantages for potential licensees and manufacturing partners.
Q: What are the primary safety advantages of this new Zolpidem synthesis route compared to traditional methods?
A: This novel process eliminates the use of highly toxic and genotoxic reagents such as sodium cyanide, iodomethane, and formaldehyde, which are common in conventional Mannich or cyano-substitution routes. By utilizing N,N-dimethylformamide (DMF) as a safe dimethylamine source and non-transition metal catalysts, the process significantly reduces occupational health hazards and simplifies waste treatment protocols.
Q: How does the use of DMF impact the production cost and scalability?
A: Using DMF as both a solvent and a reactant source drastically lowers raw material costs compared to expensive reagents like CDI or gaseous dimethylamine. Furthermore, the reaction operates under mild conditions without the need for high-pressure equipment or cryogenic temperatures, facilitating easier commercial scale-up and reducing energy consumption.
Q: What purity levels can be achieved with this catalytic oxidation method?
A: The patented method demonstrates the capability to produce Zolpidem with purity levels exceeding 99% directly after recrystallization. The avoidance of complex side reactions associated with toxic alkylating agents results in a cleaner impurity profile, reducing the burden on downstream purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Zolpidem Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic oxidative amidation route for Zolpidem production. As a leading CDMO partner, we possess the technical expertise and infrastructure to rapidly adapt this patented methodology to your specific production needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering high-purity Zolpidem that meets stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest international standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your current production metrics, highlighting exactly where this new route can drive value. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive proposal for how we can support your long-term Zolpidem sourcing strategy with reliability and excellence.
