Advanced Iodine-Promoted Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Commercial API Production
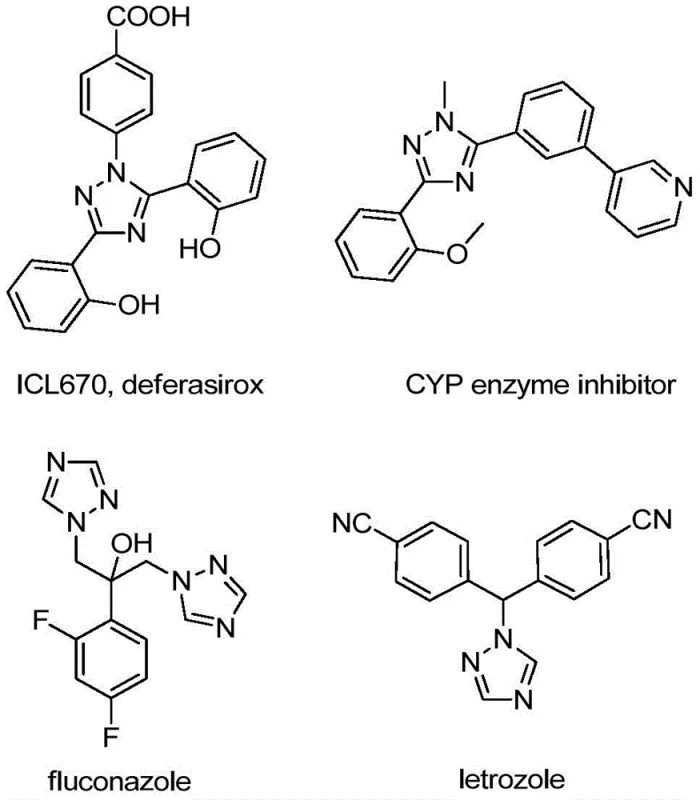
The pharmaceutical and agrochemical industries continuously seek robust methodologies for constructing nitrogen-containing heterocycles, particularly the 1,2,4-triazole scaffold, which serves as a critical pharmacophore in numerous bioactive molecules. As illustrated by prominent drugs such as fluconazole and letrozole, the incorporation of a trifluoromethyl group significantly enhances metabolic stability and lipophilicity, yet traditional synthetic routes often suffer from harsh conditions or expensive reagents. Patent CN110467579B introduces a transformative preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds that addresses these longstanding challenges through a simple, efficient, and metal-free approach. This innovation leverages inexpensive starting materials like hydrazones and trifluoroethylimide chloride, promoted by elemental iodine under mild heating, to achieve high yields without the need for rigorous anhydrous or anaerobic environments. By eliminating the dependency on toxic heavy metal catalysts and complex trifluoromethylating agents, this technology offers a streamlined pathway for producing high-purity intermediates essential for modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted nitrogen heterocycles has relied heavily on two primary strategies, both of which present significant drawbacks for large-scale manufacturing and process safety. The first approach involves the direct trifluoromethylation of pre-synthesized nitrogen heterocycles, which necessitates the use of specialized and often hazardous trifluoromethylating reagents that are costly and difficult to handle in bulk quantities. The second mainstream method utilizes trifluoromethyl synthons, such as trifluorodiazoethane, which poses severe safety risks due to its explosive nature, or trifluoroethylimide halides which have seen limited application due to reactivity challenges. Furthermore, many conventional protocols demand strict anhydrous and oxygen-free conditions, requiring specialized equipment like Schlenk lines or gloveboxes that drastically increase capital expenditure and operational complexity. The reliance on transition metal catalysts in some existing routes also introduces the risk of heavy metal contamination, necessitating additional purification steps to meet stringent regulatory standards for pharmaceutical ingredients.
The Novel Approach
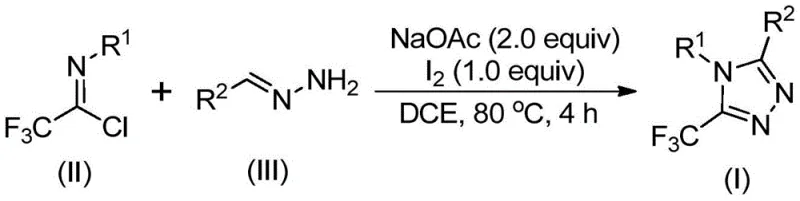
In stark contrast to these cumbersome traditional methods, the novel approach disclosed in the patent utilizes a non-metallic iodine-promoted strategy that fundamentally simplifies the synthetic workflow while maintaining high efficiency. By employing readily available hydrazones and trifluoroethylimide chloride as starting materials, the process bypasses the need for dangerous diazo compounds or expensive metal catalysts entirely. The reaction proceeds smoothly in common organic solvents such as dichloroethane at moderate temperatures around 80°C, eliminating the energy costs associated with cryogenic conditions or extreme heating. Crucially, the tolerance for ambient moisture and oxygen means that the reaction can be conducted in standard reactors without the need for inert gas protection, significantly lowering the barrier for technology transfer from laboratory to plant. This methodological shift not only enhances operational safety but also widens the substrate scope, allowing for the facile synthesis of variously substituted 1,2,4-triazole compounds through simple substrate design modifications.
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is a sophisticated sequence of base-promoted condensation and oxidative cyclization events that ensure high regioselectivity and yield. Initially, sodium acetate acts as a mild base to facilitate the intermolecular carbon-nitrogen bond formation between the trifluoroethylimide chloride and the hydrazone, generating a trifluoroacetamidine intermediate. This intermediate subsequently undergoes isomerization, positioning the reactive centers appropriately for the subsequent oxidative step mediated by elemental iodine. The iodine species promotes an oxidative iodination that generates a key iodinated intermediate, which is primed for intramolecular electrophilic substitution. This cyclization step is critical as it closes the five-membered triazole ring, followed immediately by aromatization to yield the final stable 5-trifluoromethyl substituted 1,2,4-triazole structure. The elegance of this mechanism lies in the dual role of the reagents, where iodine serves as both an oxidant and a promoter, driving the reaction to completion without the need for external oxidizing agents that could degrade sensitive functional groups.
Controlling impurity profiles in heterocyclic synthesis is paramount for pharmaceutical applications, and this iodine-promoted method offers inherent advantages in minimizing side reactions. The mild basicity of sodium acetate prevents the decomposition of sensitive hydrazone substrates that might occur under stronger alkaline conditions, thereby reducing the formation of polymeric byproducts or hydrolysis impurities. Furthermore, the specific reactivity of elemental iodine towards the amidine intermediate ensures that the cyclization occurs selectively at the desired position, avoiding the formation of regioisomers that are common in uncatalyzed thermal cyclizations. The absence of transition metals eliminates the risk of metal-ligand complexation byproducts, which are notoriously difficult to remove and can catalyze degradation during storage. Post-treatment is equally straightforward, involving simple filtration and silica gel chromatography, which effectively removes residual iodine and inorganic salts, resulting in a final product with high chemical purity suitable for downstream coupling reactions in API synthesis.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this synthesis effectively, operators must adhere to the specific molar ratios and temperature profiles outlined in the patent to ensure optimal conversion and minimize waste. The process begins with the precise mixing of sodium acetate, trifluoroethylimide chloride, and hydrazone in a suitable aprotic solvent, with dichloroethane being the preferred medium for maximizing solubility and reaction rate. Maintaining the reaction temperature between 80°C and 100°C for the initial 2 to 4 hours is critical to allow the condensation to reach equilibrium before the introduction of the oxidant. Following this incubation period, elemental iodine is added to the system, and the reaction is allowed to proceed for an additional 1 to 2 hours to complete the oxidative cyclization. Detailed standardized synthesis steps see the guide below.
- Mix sodium acetate, trifluoroethylimide chloride, and hydrazone in an organic solvent like dichloroethane.
- Heat the reaction mixture to 80°C and maintain for 2 to 4 hours to facilitate initial condensation.
- Add elemental iodine to the system and continue reacting for 1 to 2 hours to complete the oxidative cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates directly into enhanced operational resilience and significant cost optimization opportunities across the manufacturing value chain. By eliminating the requirement for expensive transition metal catalysts such as palladium or copper, the raw material costs are drastically reduced, and the supply chain is no longer vulnerable to the price volatility associated with precious metals. The use of elemental iodine and sodium acetate, which are commodity chemicals available from multiple global suppliers, ensures a stable and diversified sourcing strategy that mitigates the risk of single-source bottlenecks. Additionally, the removal of heavy metals from the process simplifies the purification workflow, reducing the consumption of specialized scavenging resins and lowering the overall cost of goods sold while accelerating the batch release timeline.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost catalytic systems with inexpensive stoichiometric reagents that are widely available in the bulk chemical market. The avoidance of specialized trifluoromethylating agents, which often carry a premium price tag due to complex synthesis and handling requirements, further contributes to substantial cost savings per kilogram of produced intermediate. Moreover, the simplified post-treatment process reduces the demand for extensive chromatographic purification or metal scavenging steps, leading to lower solvent consumption and waste disposal costs. These cumulative efficiencies result in a more competitive cost structure that allows for better margin management in the production of high-value pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on commercially available starting materials like hydrazones and imidoyl chlorides ensures that production schedules are not disrupted by long lead times for exotic reagents. The robustness of the reaction conditions, which do not require strict inert atmospheres, means that manufacturing can be performed in standard multipurpose reactors without the need for dedicated anhydrous lines, increasing facility utilization rates. This flexibility allows for rapid scale-up and responsive production planning, enabling suppliers to meet fluctuating market demands with shorter turnaround times. The stability of the reagents also simplifies storage and transportation requirements, reducing the logistical overhead associated with handling moisture-sensitive or pyrophoric materials.
- Scalability and Environmental Compliance: The environmental profile of this method aligns well with modern green chemistry initiatives, as it avoids the generation of heavy metal waste streams that require costly and regulated disposal procedures. The ability to scale the reaction from gram to multi-kilogram levels without significant loss in efficiency demonstrates its viability for commercial production, ensuring a consistent supply of high-quality intermediates. The use of common organic solvents that can be readily recovered and recycled further minimizes the environmental footprint of the manufacturing process. This compliance with environmental standards not only reduces regulatory risk but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology in an industrial setting. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the method described in CN110467579B utilizes inexpensive elemental iodine and sodium acetate, avoiding costly heavy metal catalysts and simplifying purification.
Q: What are the reaction conditions regarding moisture and oxygen?
A: The process is robust and does not require strict anhydrous or anaerobic conditions, making it highly suitable for standard industrial reactor setups without specialized inert atmosphere equipment.
Q: Is this method scalable for large volume production?
A: Yes, the patent explicitly demonstrates that the method can be easily expanded to the gram level and beyond, providing a viable pathway for industrial large-scale production applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing the supply chain for next-generation therapeutics, and we possess the technical expertise to bring this patented methodology to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 5-trifluoromethyl-1,2,4-triazole intermediate meets the highest quality standards required by global regulatory bodies. Our commitment to process excellence ensures that clients receive materials that are not only chemically pure but also consistent in quality, supporting the reliability of their downstream drug manufacturing processes.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs and volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic advantages of switching to this iodine-promoted route for your specific target molecules. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the performance and profitability of your pharmaceutical development pipeline.
