Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access privileged scaffolds like quinazolinones, which are ubiquitous in bioactive molecules ranging from anticonvulsants to antitumor agents. Patent CN112125856A discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone derivatives, addressing critical bottlenecks in current synthetic routes. As illustrated in 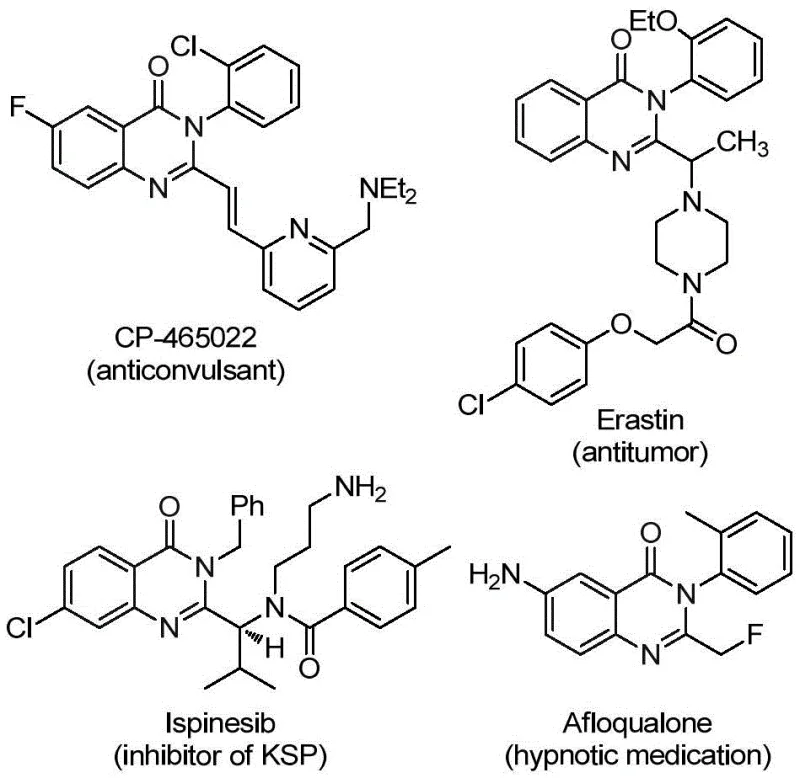 , the quinazolinone core is present in numerous commercial drugs, yet introducing a trifluoromethyl group at the 2-position has historically been challenging due to harsh conditions and limited substrate tolerance. This new technology leverages a transition metal palladium-catalyzed carbonylation tandem reaction, offering a streamlined pathway that enhances both chemical efficiency and operational safety for large-scale manufacturing.
, the quinazolinone core is present in numerous commercial drugs, yet introducing a trifluoromethyl group at the 2-position has historically been challenging due to harsh conditions and limited substrate tolerance. This new technology leverages a transition metal palladium-catalyzed carbonylation tandem reaction, offering a streamlined pathway that enhances both chemical efficiency and operational safety for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic strategies for constructing the 2-trifluoromethyl quinazolinone skeleton often rely on cumbersome multi-step sequences or hazardous reagents that pose significant risks in an industrial setting. Common literature methods involve the cyclization of anthranilamides with ethyl trifluoroacetate or trifluoroacetic anhydride, which frequently require aggressive reaction conditions and expensive pre-activated substrates. Furthermore, alternative routes utilizing isatoic anhydrides or T3P-promoted tandem reactions often suffer from narrow substrate scopes and inconsistent yields, making them unsuitable for the diverse library synthesis required in modern drug discovery. Perhaps most critically, many classical carbonylation approaches depend on the direct use of carbon monoxide gas, a colorless, odorless, and highly toxic substance that necessitates specialized high-pressure equipment and rigorous safety protocols, thereby inflating capital expenditure and operational complexity for chemical manufacturers.
The Novel Approach
In stark contrast, the methodology described in CN112125856A introduces a sophisticated yet practical solution by employing 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide substitute. This innovation effectively eliminates the need for gaseous CO, allowing the reaction to proceed under standard atmospheric pressure in common laboratory glassware or standard reactors. The core transformation involves a palladium-catalyzed coupling between readily available o-iodoanilines and trifluoroacetimidoyl chlorides, as depicted in the general reaction scheme 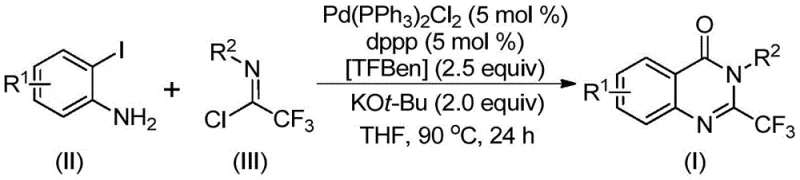 . This approach not only simplifies the operational workflow but also dramatically broadens the functional group tolerance, enabling the synthesis of derivatives with diverse electronic and steric properties without compromising yield or purity, thus representing a significant leap forward in process chemistry.
. This approach not only simplifies the operational workflow but also dramatically broadens the functional group tolerance, enabling the synthesis of derivatives with diverse electronic and steric properties without compromising yield or purity, thus representing a significant leap forward in process chemistry.
Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Reaction
The success of this synthesis relies on a meticulously orchestrated catalytic cycle initiated by the base-promoted intermolecular carbon-nitrogen bond coupling. Initially, potassium tert-butoxide facilitates the formation of a trifluoroacetamidine derivative from the starting imidoyl chloride and amine. Subsequently, the palladium catalyst, specifically Pd(PPh3)2Cl2 coordinated with the bidentate ligand dppp, undergoes oxidative addition into the carbon-iodine bond of the o-iodoaniline moiety to generate a reactive divalent palladium intermediate. Under the thermal conditions of 90°C, the solid CO source TFBen decomposes in situ to release carbon monoxide, which then inserts into the carbon-palladium bond to form a crucial acyl-palladium species. This step is pivotal as it introduces the carbonyl functionality required for the quinazolinone ring closure without external gas feed.
Following the CO insertion, the basic environment promotes an intramolecular nucleophilic attack by the nitrogen atom onto the acyl-palladium center, leading to the formation of a seven-membered ring palladium intermediate. This cyclic intermediate is thermodynamically driven towards the final product through a reductive elimination step, which regenerates the active palladium(0) catalyst and releases the desired 2-trifluoromethyl substituted quinazolinone. This mechanism ensures high atom economy and minimizes the formation of side products, as the tandem nature of the reaction prevents the isolation of unstable intermediates. The choice of THF as the solvent is also critical, as it effectively solubilizes all organic and inorganic components while stabilizing the polar transition states involved in the catalytic cycle, ensuring consistent performance across different substrate combinations.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis protocol requires precise control over stoichiometry and reaction parameters to maximize yield and minimize impurity profiles. The process is designed to be scalable, utilizing commercially available reagents that do not require complex pre-synthesis or purification. Operators should note that the ratio of the palladium catalyst to ligand is optimized at 1:1 (5 mol% each) to maintain catalytic activity throughout the 16 to 30-hour reaction window. Detailed standardized operating procedures regarding reagent addition order, temperature ramping, and work-up protocols are essential for reproducibility. For a comprehensive step-by-step guide on executing this synthesis in a GMP-compliant environment, please refer to the technical instructions below.
- Combine palladium catalyst Pd(PPh3)2Cl2, ligand dppp, base KOt-Bu, solid CO source TFBen, trifluoroacetimidoyl chloride, and o-iodoaniline in THF solvent.
- Heat the reaction mixture to 90°C and maintain stirring for 16 to 30 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl quinazolinone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical yield. By shifting away from hazardous gaseous reagents and expensive activated substrates, manufacturers can significantly reduce the overhead costs associated with safety compliance, specialized equipment maintenance, and waste disposal. The use of stable, solid starting materials like TFBen and trifluoroacetimidoyl chlorides ensures a more predictable supply chain, as these commodities are widely available from multiple global vendors, mitigating the risk of single-source dependency. Furthermore, the robustness of the reaction conditions allows for flexible scheduling and easier scale-up from kilogram to multi-ton production without the need for exotic high-pressure reactors.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide gas removes the necessity for expensive gas handling infrastructure and rigorous safety monitoring systems, leading to substantial operational cost savings. Additionally, the use of cheap and readily available o-iodoanilines and imidoyl chlorides as starting materials lowers the raw material cost basis compared to traditional methods requiring pre-functionalized anthranilic acid derivatives. The high conversion rates observed, with yields reaching up to 99% in optimized examples, further enhance cost efficiency by minimizing raw material waste and reducing the load on downstream purification processes.
- Enhanced Supply Chain Reliability: The reliance on stable solid reagents rather than pressurized gases or unstable liquids significantly de-risks the logistics and storage aspects of the supply chain. Since the key building blocks are common fine chemicals with established global supply networks, procurement teams can secure long-term contracts with better pricing stability. The method's tolerance for various substituents means that a single platform technology can be used to produce a wide array of derivatives, allowing for agile response to changing market demands without requalifying entirely new synthetic routes for each new analog.
- Scalability and Environmental Compliance: From an environmental perspective, replacing gaseous CO with a solid surrogate drastically reduces the potential for accidental emissions and aligns with increasingly stringent green chemistry regulations. The reaction proceeds in THF, a solvent that is easily recovered and recycled in standard distillation units, supporting sustainable manufacturing practices. The simplicity of the post-treatment process, involving filtration and standard column chromatography or crystallization, facilitates rapid scale-up from pilot plant to commercial production, ensuring that lead times for high-purity pharmaceutical intermediates can be consistently met even during peak demand periods.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into existing production lines. The following questions address common inquiries regarding substrate compatibility, safety protocols, and scalability, derived directly from the experimental data and technical specifications outlined in the patent documentation. These insights are intended to provide clarity on how this technology can be adapted to specific project requirements while maintaining high standards of quality and safety.
Q: What are the safety advantages of this synthesis method compared to traditional carbonylation?
A: This method utilizes 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide substitute, completely avoiding the handling of toxic and hazardous carbon monoxide gas, which significantly improves operational safety and reduces regulatory compliance burdens.
Q: What is the substrate scope for the R1 and R2 groups in this reaction?
A: The process demonstrates excellent compatibility with various substituents. R1 can be H, alkyl, halogen (F, Cl, Br), or trifluoromethyl, while R2 accommodates substituted or unsubstituted aryl groups including phenyl, naphthyl, and those with electron-withdrawing or donating groups like nitro or tert-butyl.
Q: What are the typical reaction conditions required for high yield?
A: Optimal results are achieved using Pd(PPh3)2Cl2 (5 mol%) and dppp (5 mol%) as the catalytic system in THF solvent at 90°C for 16 to 30 hours, with potassium tert-butoxide serving as the base to promote the tandem cyclization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates for the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards. Our expertise in palladium-catalyzed transformations allows us to optimize this specific quinazolinone synthesis for maximum yield and minimal impurity generation.
We invite potential partners to engage with our technical procurement team to discuss how this innovative route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this safer, more efficient methodology. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to initiate route feasibility assessments for your proprietary targets, ensuring a reliable supply of high-purity 2-trifluoromethyl quinazolinone derivatives for your pipeline.
