Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Drug Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are known to enhance metabolic stability and bioavailability. Patent CN113045503B introduces a significant advancement in this domain by disclosing a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These scaffolds are critical structural cores found in numerous bioactive molecules, ranging from antifungal and antiviral agents to anticancer drugs like Methaqualone and the natural product Rutaecarpine. The disclosed technology leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing cheap and readily available trifluoroethylimidoyl chloride and various amines as starting materials. This approach represents a paradigm shift from traditional cyclization methods, offering a pathway that is not only operationally simple but also highly scalable for industrial applications. For R&D directors and process chemists, this patent provides a versatile toolkit for accessing diverse chemical space with high reaction efficiency and broad substrate compatibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl-substituted quinazolinones has been fraught with significant technical and economic challenges that hinder large-scale production. Conventional literature methods often rely on the cyclization of anthranilamides with ethyl trifluoroacetate, trifluoroacetic anhydride, or trifluoroacetic acid under varying conditions. Alternatively, strategies involving the reaction of anthranilates with unstable trifluoroacetamides or the cyclization of isatoic anhydride with trifluoroacetic anhydride have been employed. More recent approaches utilize T3P-promoted cascade reactions of anthranilic acid, trifluoroacetic acid, and amines. However, these existing methodologies are generally limited by harsh reaction conditions that require stringent temperature and pressure controls, leading to safety concerns in a manufacturing environment. Furthermore, the substrates required, such as pre-activated anhydrides or unstable amides, are often expensive and difficult to source in bulk quantities, driving up the cost of goods. Perhaps most critically for process development, these traditional routes frequently suffer from low yields and narrow substrate scopes, making it difficult to generate diverse libraries of analogs for structure-activity relationship (SAR) studies without extensive optimization.
The Novel Approach
In stark contrast to the limitations of prior art, the novel approach detailed in CN113045503B utilizes a palladium-catalyzed carbonylation cascade that fundamentally simplifies the synthetic logic. By employing trifluoroethylimidoyl chloride and simple amines as the primary building blocks, the method bypasses the need for expensive pre-activated substrates. A key innovation is the use of TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide substitute, which releases CO in situ under heating conditions. This eliminates the hazards associated with handling high-pressure CO gas cylinders, thereby enhancing workplace safety and reducing infrastructure costs. The reaction proceeds smoothly in common organic solvents like 1,4-dioxane at 110°C, demonstrating remarkable tolerance for various functional groups including halogens, alkyls, and trifluoromethyl groups on the aromatic ring. This broad compatibility allows medicinal chemists to design and synthesize a wide array of substituted trifluoromethyl quinazolinone compounds simply by varying the amine or the imidoyl chloride starting materials, significantly widening the practical utility of the method for drug discovery programs.
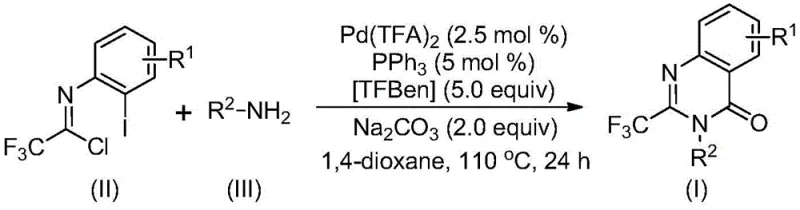
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic pathway proposed for this transformation involves a sophisticated sequence of organometallic steps that ensure high efficiency and selectivity. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond coupling between the trifluoroethylimidoyl chloride and the amine to form a trifluoroacetamidine derivative intermediate. Subsequently, the palladium catalyst, generated from palladium trifluoroacetate and triphenylphosphine, undergoes oxidative addition into the carbon-iodine bond of the aromatic ring, forming a divalent palladium species. Concurrently, the TFBen additive decomposes under thermal conditions to release carbon monoxide, which then inserts into the carbon-palladium bond to generate an acyl-palladium intermediate. This insertion step is crucial for introducing the carbonyl functionality required for the quinazolinone core. Under the influence of the base, typically sodium carbonate, the system promotes the formation of a palladium-nitrogen bond, closing the ring to form a seven-membered palladacycle intermediate. The catalytic cycle is completed by a reductive elimination step, which releases the final 2-trifluoromethyl-substituted quinazolinone product and regenerates the active palladium(0) species for the next turnover. Understanding this mechanism is vital for R&D teams aiming to further optimize ligand systems or catalyst loadings for specific difficult substrates.
From an impurity control perspective, the mild nature of this catalytic system offers distinct advantages over harsh acidic or basic cyclizations. Traditional methods often lead to decomposition of sensitive functional groups or polymerization side reactions due to the aggressive reagents used. In this palladium-catalyzed protocol, the reaction conditions are neutral to slightly basic, preserving sensitive moieties such as esters or halides that might otherwise be compromised. The use of a solid CO source like TFBen ensures a steady, controlled release of carbon monoxide, preventing local concentration spikes that could lead to over-carbonylation or other side reactions. Furthermore, the high selectivity of the palladium catalyst for the specific C-I bond activation minimizes the formation of regioisomers or homocoupling byproducts. This clean reaction profile simplifies the downstream purification process, often allowing for straightforward filtration and column chromatography to achieve high-purity products, which is essential for meeting the stringent quality standards required for pharmaceutical intermediates.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The experimental procedure outlined in the patent provides a clear roadmap for executing this synthesis with high reproducibility. The process begins by charging a reaction vessel, such as a Schlenk tube, with the precise stoichiometric amounts of the catalyst system, specifically palladium trifluoroacetate and triphenylphosphine, along with the base sodium carbonate and the CO source TFBen. To this solid mixture, the trifluoroethylimidoyl chloride substrate and the chosen amine nucleophile are added, followed by the organic solvent, preferably 1,4-dioxane, to ensure complete dissolution of all components. The detailed standardized synthesis steps for scaling this reaction are provided in the guide below, ensuring that operators can replicate the high yields reported in the patent examples.
- Combine palladium trifluoroacetate, triphenylphosphine, TFBen, sodium carbonate, trifluoroethylimidoyl chloride, and amine in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 110°C and stir for 16 to 30 hours to allow the carbonylation cascade to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The primary driver for cost reduction lies in the raw material selection; the method utilizes trifluoroethylimidoyl chlorides and simple amines, which are commodity chemicals available from multiple global suppliers at competitive prices. Unlike traditional routes that require specialized, expensive reagents like T3P or unstable anhydrides, this process relies on robust, shelf-stable starting materials that simplify inventory management and reduce storage costs. Additionally, the elimination of high-pressure gas equipment for carbon monoxide delivery reduces capital expenditure on reactor infrastructure and lowers maintenance overheads. The streamlined workup procedure, involving simple filtration and standard chromatography, minimizes solvent consumption and waste generation, contributing to a more sustainable and cost-effective manufacturing process that aligns with modern green chemistry principles.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the use of inexpensive catalysts and ligands in low molar percentages, combined with the avoidance of costly activating agents. By replacing hazardous gaseous CO with a solid surrogate, the process eliminates the need for specialized high-pressure reactors and the associated safety compliance costs, leading to substantial operational savings. The high reaction efficiency and yields reported across a broad range of substrates mean that less raw material is wasted, maximizing the output per batch and reducing the cost per kilogram of the final API intermediate. Furthermore, the simplified purification requirements reduce the consumption of silica gel and elution solvents, which are often significant cost drivers in fine chemical production.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, commercially sourced reagents rather than custom-synthesized specialty chemicals. The starting amines and imidoyl chlorides are part of standard chemical catalogs, ensuring that procurement teams can source materials from multiple vendors to mitigate supply disruption risks. The robustness of the reaction conditions, which tolerate a wide variety of functional groups, means that the process is less sensitive to minor variations in raw material quality, reducing the rate of batch failures. This reliability ensures consistent production schedules and on-time delivery to downstream customers, which is critical for maintaining trust in long-term supply agreements within the pharmaceutical sector.
- Scalability and Environmental Compliance: The method has been demonstrated to be scalable, with the patent noting successful extension to gram-level synthesis, indicating strong potential for kilogram and ton-scale production. The use of 1,4-dioxane as a solvent, while requiring careful handling, is a well-understood industrial solvent with established recovery and recycling protocols, facilitating compliance with environmental regulations. The absence of heavy metal waste streams typical of stoichiometric oxidants or harsh acids simplifies wastewater treatment processes. The overall atom economy of the carbonylation cascade is superior to multi-step traditional routes, resulting in less chemical waste and a smaller environmental footprint, which is increasingly important for meeting corporate sustainability goals and regulatory standards in chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and descriptions provided in the patent documentation, offering clarity on reaction parameters and applicability. Understanding these details is crucial for process engineers evaluating the feasibility of integrating this method into existing production lines.
Q: What are the advantages of using TFBen over traditional CO gas sources?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a solid carbon monoxide substitute, eliminating the need for high-pressure CO gas equipment and significantly improving operational safety and handling convenience in standard laboratory or plant settings.
Q: What is the substrate scope for the amine component in this reaction?
A: The method demonstrates excellent compatibility with various amines including alkyl amines (n-Bu, t-Bu), benzyl amines, and substituted aryl amines, allowing for the synthesis of diverse quinazolinone derivatives suitable for SAR studies.
Q: Can this method be applied to the synthesis of complex drug molecules?
A: Yes, the patent explicitly demonstrates the successful application of this methodology in the efficient total synthesis of the bioactive natural product Rutaecarpine with a high overall yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed technology for the rapid development of novel therapeutic agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium chemistry, including rigorous QC labs capable of detecting trace metal residues to meet stringent purity specifications required by global regulatory bodies. We are committed to delivering high-quality intermediates that empower your drug discovery pipelines with speed and reliability.
We invite you to leverage our technical expertise to optimize your supply chain for quinazolinone-based projects. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this novel route can reduce your overall cost of goods. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us be your strategic partner in bringing the next generation of fluorinated pharmaceuticals to market efficiently.
