Advanced Rhodium-Catalyzed Synthesis of Polysubstituted Isoindoles for Commercial Pharmaceutical Intermediate Production
Introduction to Next-Generation Isoindole Synthesis
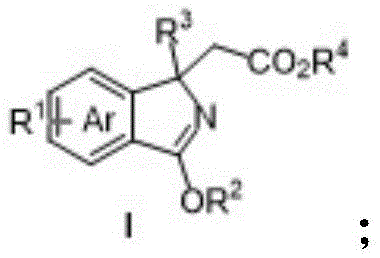
The landscape of organic synthesis for nitrogen-containing heterocycles is undergoing a significant transformation, driven by the urgent need for more efficient and environmentally benign methodologies. Patent CN109879792B introduces a groundbreaking approach to the preparation of polysubstituted isoindole compounds, a structural motif that serves as a critical scaffold in the development of bioactive natural products and advanced pharmaceutical agents. This technology specifically addresses the longstanding challenges associated with constructing isoindole cores, particularly those featuring quaternary carbon centers, which have historically been difficult to access through classical synthetic routes. By leveraging a trivalent rhodium-catalyzed C-H activation strategy coupled with an oxidative Heck reaction, this invention provides a modular and atom-economical pathway to high-value chemical intermediates.
The significance of isoindole derivatives extends far beyond academic curiosity; they are foundational building blocks for potent protein kinase inhibitors such as staurosporine and are integral to the production of high-performance organic pigments like Pigment Yellow 139. However, traditional extraction from natural sources yields insufficient quantities, and legacy synthetic methods often suffer from poor atom economy and severe environmental pollution. The methodology disclosed in this patent represents a paradigm shift, offering a reliable pharmaceutical intermediates supplier with a tool to access these complex architectures through a streamlined, one-pot process that operates under remarkably mild conditions. This report analyzes the technical depth and commercial viability of this innovation for global supply chain integration.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isoindole skeleton has relied on a limited set of classical transformations that are increasingly viewed as obsolete in the context of modern green chemistry standards. Traditional protocols often involve the high-temperature thermal decomposition of 1,2,3,4-tetralin-1,4-imines or the harsh alkylation of 1,2-dibromobenzene followed by rigorous heating steps. These processes are not only energy-intensive but also generate substantial amounts of hazardous by-products, creating significant disposal burdens for manufacturing facilities. Furthermore, conventional strategies struggle immensely with the introduction of quaternary carbon centers at the C1 position of the isoindole ring, a structural feature that is often essential for enhancing the metabolic stability and binding affinity of drug candidates.
Another critical drawback of existing technologies is the reliance on pre-functionalized starting materials, such as aryl halides, which necessitate additional synthetic steps to install the requisite leaving groups prior to cyclization. This increases the overall step count, reduces the cumulative yield, and escalates the cost of goods sold (COGS). Additionally, many classical methods exhibit poor regioselectivity, leading to complex mixtures of isomers that are difficult and costly to separate. For procurement managers and supply chain heads, these inefficiencies translate into longer lead times, higher raw material consumption, and increased regulatory scrutiny due to the presence of genotoxic impurities often associated with harsh halogenation chemistries.
The Novel Approach
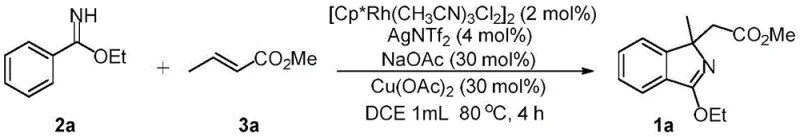
In stark contrast to these legacy methods, the invention described in CN109879792B utilizes a direct C-H bond functionalization strategy that bypasses the need for pre-installed halogen handles. The core of this innovation is the use of a trivalent rhodium catalyst to activate the inert C-H bond ortho to the imidate directing group, followed by an oxidative coupling with an unfunctionalized alkene ester. This tandem reaction sequence effectively merges C-H activation and cyclization into a single operational step, drastically simplifying the synthetic workflow. The ability to use simple imidates and commercially available acrylates as starting materials represents a major advancement in cost reduction in fine chemical manufacturing.
Moreover, this novel approach demonstrates exceptional versatility in constructing quaternary carbon centers, a feat that is notoriously difficult to achieve with high fidelity using nucleophilic substitution or Grignard-type reactions. The reaction proceeds under mild thermal conditions, typically around 80°C, and tolerates an air atmosphere, eliminating the need for expensive inert gas infrastructure. This robustness makes the process highly attractive for commercial scale-up of complex heterocyclic scaffolds. By enabling the direct assembly of polysubstituted isoindoles from readily available feedstocks, this technology offers a sustainable and economically superior alternative to traditional synthesis, aligning perfectly with the industry's shift towards greener and more efficient production models.

Mechanistic Insights into Rh(III)-Catalyzed Oxidative Heck Cyclization
The mechanistic underpinning of this transformation relies on the unique ability of cationic Cp*Rh(III) species to coordinate with the nitrogen atom of the imidate substrate, thereby directing the metal center to the adjacent ortho-C-H bond. This coordination initiates a concerted metalation-deprotonation (CMD) process, facilitated by the acetate base, which results in the formation of a stable five-membered rhodacycle intermediate. This step is crucial as it determines the regioselectivity of the reaction, ensuring that functionalization occurs exclusively at the desired position on the aromatic ring. The resulting organometallic species then undergoes migratory insertion with the alkene substrate, forming a new carbon-carbon bond and extending the metal-alkyl chain.
Following the insertion step, the catalytic cycle proceeds through a beta-hydride elimination or an oxidative pathway that ultimately leads to the formation of the isoindole double bond and the regeneration of the active Rh(III) catalyst. The presence of copper(II) acetate serves a dual purpose: it acts as a terminal oxidant to re-oxidize any reduced rhodium species back to the active Rh(III) state, and it assists in the final aromatization or cyclization steps. The silver additive, such as AgNTf2, plays a vital role in abstracting chloride ligands from the rhodium precursor to generate the highly electrophilic cationic rhodium species required for efficient C-H activation. This intricate interplay of catalysts and additives ensures high turnover numbers and minimizes the formation of side products, thereby delivering high-purity isoindole derivatives suitable for sensitive pharmaceutical applications.
From an impurity control perspective, the high chemoselectivity of the rhodium catalyst is paramount. Unlike palladium-catalyzed cross-couplings which might suffer from homocoupling of aryl halides or beta-hydride elimination side reactions leading to linear by-products, this oxidative Heck variant is tightly controlled by the chelating assistance of the imidate group. This intrinsic directing effect suppresses non-specific background reactions, resulting in a cleaner crude reaction profile. For R&D directors, this means simplified downstream processing and a reduced burden on analytical quality control laboratories, as the impurity spectrum is predictable and manageable. The mechanism effectively converts simple, low-cost building blocks into complex, high-value architectures with minimal waste generation.
How to Synthesize Polysubstituted Isoindoles Efficiently
The practical implementation of this synthesis protocol is designed for ease of execution, requiring standard laboratory equipment and commercially available reagents. The process begins by combining the imidate substrate and the alkene coupling partner in a chlorinated solvent, followed by the sequential addition of the rhodium catalyst system and oxidants. The reaction is then heated to a moderate temperature, allowing the catalytic cycle to proceed to completion within a few hours. Detailed standardized synthesis steps for replicating this high-efficiency transformation are provided in the guide below, ensuring consistent results across different production batches.
- Dissolve the imidate substrate (Formula II) and the alkene ester (Formula III) in an inert solvent such as 1,2-dichloroethane (DCE) within a reaction vessel under an air atmosphere.
- Add the trivalent rhodium catalyst (e.g., [Cp*RhCl2]2), silver additive (AgNTf2), copper oxidant (Cu(OAc)2), and sodium acetate base to the reaction mixture.
- Heat the reaction mixture to 80°C for approximately 4 hours, then cool, filter through celite, and purify the crude product via silica gel chromatography to obtain the target isoindole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this Rh(III)-catalyzed technology offers compelling economic and operational benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials; by utilizing unfunctionalized alkenes and simple imidates, manufacturers can source starting materials from bulk chemical suppliers at a fraction of the cost of specialized halogenated precursors. This shift significantly reduces the dependency on volatile halogen markets and mitigates the risk of supply disruptions associated with complex custom synthesis of starting materials. Furthermore, the operational simplicity of running reactions under air atmosphere removes the capital expenditure requirement for extensive inert gas manifolds and glovebox facilities.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps, such as halogenation or lithiation, directly translates to a reduction in raw material costs and processing time. By consolidating multiple synthetic transformations into a single pot, the process minimizes solvent usage, energy consumption for heating and cooling cycles, and labor hours required for intermediate isolation. Although specific percentage savings depend on the specific target molecule, the qualitative impact on the cost of goods is substantial due to the reduced step count and higher overall throughput. Additionally, the use of earth-abundant copper salts as co-oxidants helps keep reagent costs low compared to processes relying solely on precious metal oxidants.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system against air and moisture variations enhances supply chain reliability by reducing the sensitivity of the manufacturing process to environmental fluctuations. The broad substrate scope, which tolerates various functional groups including halogens and nitro groups, allows for a flexible sourcing strategy where different substituted imidates can be utilized without re-optimizing the entire process. This flexibility ensures that if one specific raw material faces a shortage, alternative analogs can be swiftly substituted to maintain production continuity, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From a scalability perspective, the reaction conditions (80°C, standard pressure) are easily transferable from laboratory glassware to industrial stainless steel reactors without requiring exotic high-pressure or cryogenic equipment. The improved atom economy and reduced generation of halogenated waste streams align with increasingly stringent environmental regulations, lowering the cost of waste treatment and disposal. The ability to produce complex quaternary carbon centers efficiently also opens up new avenues for producing high-margin specialty chemicals that were previously too expensive to manufacture at scale, providing a competitive edge in the market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed isoindole synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for technical teams evaluating this process for adoption. Understanding these nuances is critical for assessing the feasibility of integrating this methodology into existing production pipelines.
Q: What are the primary advantages of this Rhodium-catalyzed method over traditional isoindole synthesis?
A: Unlike conventional methods that require harsh conditions like high-temperature heating of tetralin-imines or complex multi-step alkylations of dibromobenzenes, this Rh(III)-catalyzed approach utilizes direct C-H activation. This results in significantly fewer synthetic steps, milder reaction temperatures (around 80°C), and the ability to construct difficult quaternary carbon centers efficiently.
Q: Does this synthesis method tolerate diverse functional groups on the aromatic ring?
A: Yes, the methodology demonstrates excellent functional group tolerance. The patent data confirms successful synthesis with substrates containing electron-withdrawing groups such as bromo, iodo, and nitro substituents, as well as electron-donating groups, allowing for the modular construction of complex derivatives suitable for further cross-coupling transformations.
Q: Is the reaction sensitive to atmospheric conditions?
A: The process is notably robust regarding atmospheric conditions. The experimental examples specify that reactions can be conducted under a standard air atmosphere (one atmosphere of air) rather than requiring strict inert gas protection like nitrogen or argon, which simplifies operational complexity and reduces equipment costs for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Isoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in reshaping the landscape of pharmaceutical intermediate production. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the Rh(III)-catalyzed isoindole synthesis are seamlessly translated into robust industrial processes. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs dedicated to characterizing every batch to meet the exacting standards of global regulatory bodies.
We invite forward-thinking pharmaceutical and agrochemical companies to collaborate with us to leverage this cutting-edge synthesis platform for their pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific target molecules, demonstrating exactly how this technology can optimize your budget. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on the foundation of the most efficient and sustainable chemistry available.
