Advanced Synthesis of Mosapride Intermediate I for Commercial Scale-up
Introduction to the Novel Mosapride Intermediate Synthesis
The pharmaceutical landscape for gastrointestinal prokinetic agents relies heavily on the consistent quality of key building blocks, specifically for drugs like Mosapride Citrate. As detailed in patent CN112225708B, a breakthrough preparation method has been developed for Intermediate I, chemically known as 4-(4-fluorobenzyl)-2-aminomethyl morpholine. This compound serves as the critical morpholine-containing fragment in the final active pharmaceutical ingredient (API). The significance of this development cannot be overstated, as the quality of Intermediate I directly dictates the impurity profile and stability of the final Mosapride Citrate product. Historically, variations in the intermediate's purity have led to batch failures in related substance testing, creating bottlenecks in API production. This new technical disclosure offers a robust, scalable, and economically superior pathway that addresses these long-standing industry pain points.
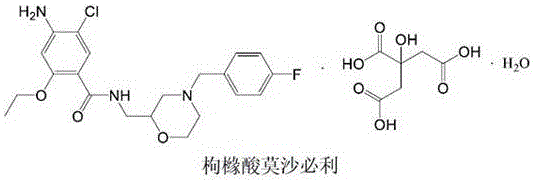
Mosapride Citrate functions as a selective 5-hydroxytryptamine 4 (5-HT4) receptor agonist, widely prescribed for functional dyspepsia due to its ability to promote acetylcholine release without affecting gastric acid secretion. The chemical integrity of the morpholine ring and the fluorobenzyl moiety is paramount for its biological activity. The patent introduces a streamlined synthetic route that bypasses the complexities of previous methodologies. By focusing on the optimization of reaction conditions and the selection of cost-effective starting materials, this process ensures a reliable supply of high-purity intermediates. For procurement and R&D teams, understanding this shift in synthetic strategy is essential for securing long-term supply chains and reducing the cost of goods sold (COGS) for the final medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes for Mosapride Intermediate I have been plagued by inefficiencies that impact both cost and quality. Traditional methods, such as those described in earlier patents like CN1526700A, often commence with p-fluorobenzaldehyde, requiring a multi-step sequence involving nucleophilic addition, reduction with sodium borohydride, and subsequent ring-opening reactions. These pathways frequently necessitate the use of 2-(4-fluorobenzylamino)ethanol as a key intermediate, a reagent that commands a high market price and adds significant cost pressure to the manufacturing process. Furthermore, the reliance on complex protection and deprotection strategies, along with harsh acidic hydrolysis conditions, often results in intermediates with poor stability and undesirable physical properties, such as dark brown oily liquids that are difficult to purify.
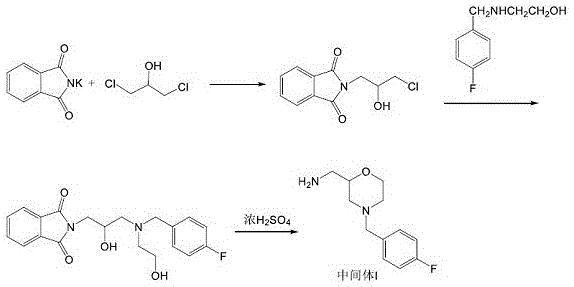
The cumulative effect of these conventional limitations is a prolonged production cycle and a higher risk of impurity carryover. When the intermediate possesses low purity or poor color, it inevitably compromises the quality of the final Mosapride Citrate, leading to potential regulatory non-compliance during related substance detection. Additionally, the use of expensive starting materials and the requirement for multiple isolation steps increase the capital consumption and time-to-market for the API. For supply chain managers, these factors translate into volatile pricing and potential disruptions if the supply of specialized precursors like 2-(4-fluorobenzylamino)ethanol is constrained. Therefore, the industry has urgently required a more direct, economical, and environmentally friendly alternative.
The Novel Approach
The methodology disclosed in CN112225708B represents a paradigm shift by utilizing phthalimide potassium salt and dichloroisopropanol as the primary building blocks. This strategy effectively decouples the synthesis from expensive fluorinated amines, instead introducing the fluorobenzyl group at a later, more controlled stage. The core innovation involves the formation of a morpholine ring through an intramolecular cyclization of a hydroxy-propyl-phthalimide derivative, followed by a highly efficient N-alkylation. This approach not only shortens the overall process route but also significantly enhances the purity of the final intermediate. By avoiding the early introduction of the fluorobenzyl moiety, the process minimizes side reactions associated with the sensitive fluoro-substituted aromatic ring during the rigorous cyclization steps.
Furthermore, the novel approach leverages a specific base system in the final alkylation step, utilizing a combination of tert-butanol metal salts and triethylamine. This dual-base strategy has been empirically shown to drastically improve yields compared to single-base systems, ensuring that the conversion of the morpholine precursor to the final Intermediate I is both high-yielding and clean. The result is a solid product with excellent purity profiles, eliminating the issues of dark coloration and instability seen in older methods. For manufacturers, this translates to a simplified downstream processing workflow, reduced solvent usage, and a final product that consistently meets the stringent specifications required for API synthesis, thereby securing the quality of the entire pharmaceutical value chain.
Mechanistic Insights into the Phthalimide-Mediated Cyclization
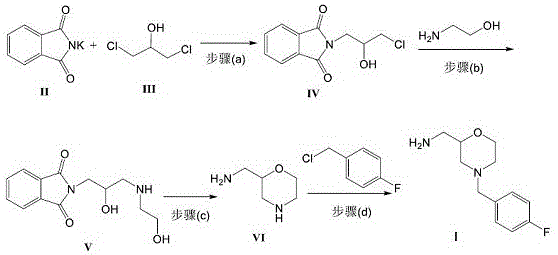
The success of this synthetic route hinges on the precise control of nucleophilic substitution and cyclization mechanisms. The process initiates with the N-alkylation of phthalimide potassium salt by dichloroisopropanol. In this step, the phthalimide anion acts as a potent nucleophile, displacing one of the chlorine atoms to form N-(2-hydroxy-3-chloropropyl)phthalimide. The presence of the remaining hydroxyl and chloro groups sets the stage for the subsequent ring construction. Following this, the introduction of 2-aminoethanol triggers a condensation reaction where the primary amine attacks the remaining chloro-substituted carbon. This step is critical as it installs the nitrogen atom required for the morpholine ring while maintaining the phthalimide protecting group, which prevents unwanted polymerization or side reactions of the amine during the harsh conditions of the subsequent cyclization.
The cyclization step (Step c in the patent) is the mechanistic cornerstone of this process. Under strong acidic conditions, typically using p-toluenesulfonic acid or concentrated sulfuric acid in a high-boiling solvent like toluene, the molecule undergoes dehydration and intramolecular nucleophilic attack. The secondary amine, liberated or activated within the chain, attacks the carbon bearing the hydroxyl group, closing the six-membered morpholine ring. Simultaneously, the acidic environment facilitates the hydrolysis of the phthalimide group, releasing the primary amine functionality as 2-aminomethyl morpholine (Compound VI). This tandem cyclization-deprotection sequence is highly efficient, converting a linear precursor into the cyclic core with high atom economy. The final step involves the N-alkylation of this morpholine derivative with 4-fluorobenzyl chloride, where the optimized base system ensures complete conversion without degrading the sensitive fluorobenzyl group.
How to Synthesize 4-(4-fluorobenzyl)-2-aminomethyl morpholine Efficiently
The practical implementation of this synthesis requires strict adherence to the optimized reaction parameters outlined in the patent to ensure reproducibility and high yield. The process is designed to be scalable, moving seamlessly from laboratory benchtop to industrial reactor vessels. Key to the operation is the management of stoichiometry, particularly in the final alkylation step where the ratio of base to substrate is critical for maximizing yield. The use of polar aprotic solvents like DMF or toluene facilitates the solubility of intermediates and promotes the nucleophilic displacement reactions. Operators must monitor the reaction progress carefully, especially during the acid-catalyzed cyclization, to ensure complete water removal which drives the equilibrium towards the desired morpholine product.
- React phthalimide potassium salt with dichloroisopropanol under alkaline conditions to form N-(2-hydroxy-3-chloropropyl)phthalimide.
- Condense the intermediate with 2-aminoethanol to generate a hydroxy-propyl-phthalimide derivative.
- Perform acid-catalyzed cyclization and deprotection to obtain 2-aminomethyl morpholine (Compound VI).
- React Compound VI with 4-fluorobenzyl chloride using a tert-butoxide/triethylamine base system to yield the final Mosapride Intermediate I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic advantages beyond mere technical feasibility. The primary driver for cost reduction lies in the substitution of expensive raw materials with commodity chemicals. By eliminating the dependency on 2-(4-fluorobenzylamino)ethanol, a specialized and costly intermediate, manufacturers can leverage the widespread availability and low price point of 2-aminoethanol and phthalimide salts. This fundamental shift in the bill of materials (BOM) structure results in substantial cost savings per kilogram of the final intermediate, directly improving the margin profile for the API producer. Moreover, the shortened process route reduces the number of unit operations, thereby lowering labor costs, energy consumption, and waste disposal fees associated with multiple isolation and purification steps.
- Cost Reduction in Manufacturing: The economic benefits are further amplified by the high yields achieved in each step, particularly the final alkylation which historically suffered from lower efficiency. The patent data indicates that optimizing the base system leads to superior conversion rates, meaning less raw material is wasted in the form of unreacted starting materials or byproducts. This efficiency gain reduces the effective cost of goods sold (COGS) and minimizes the environmental footprint of the manufacturing process. Additionally, the ability to produce a high-purity solid intermediate simplifies the logistics of storage and transport, as stable solids are far easier and cheaper to handle than unstable oily liquids that require special containment or immediate processing.
- Enhanced Supply Chain Reliability: From a supply chain perspective, relying on commoditized starting materials significantly de-risks the procurement process. Phthalimide potassium salt and dichloroisopropanol are produced by numerous global chemical suppliers, ensuring a competitive market and preventing single-source bottlenecks. This diversification of the supply base guarantees continuity of supply even during market fluctuations or geopolitical disruptions that might affect specialized fine chemical vendors. The robustness of the synthesis also means that production schedules are more predictable, with fewer delays caused by failed batches or the need for re-processing. This reliability is crucial for maintaining the just-in-time inventory models often employed by large-scale API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, utilizing standard reaction conditions and solvents that are compatible with existing stainless steel reactor infrastructure. The avoidance of exotic catalysts or extreme cryogenic conditions simplifies the engineering requirements for plant modification. Furthermore, the improved purity of the intermediate reduces the burden on downstream purification processes in the API synthesis, leading to less solvent waste and lower emissions. This alignment with green chemistry principles not only aids in meeting increasingly strict environmental regulations but also enhances the corporate sustainability profile of the manufacturing entity, a factor of growing importance to stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this new synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements. The answers reflect the consensus on how this specific chemical pathway resolves historical challenges in Mosapride production.
Q: Why is the purity of Mosapride Intermediate I critical for the final API?
A: Intermediate I (4-(4-fluorobenzyl)-2-aminomethyl morpholine) is a key structural component of Mosapride Citrate. Historical data indicates that intermediates with poor stability, dark color, or low purity directly lead to final products failing related substance tests. High-purity synthesis ensures the final API meets stringent pharmacopeial standards for safety and efficacy.
Q: How does this new method reduce manufacturing costs compared to prior art?
A: Traditional routes often rely on expensive precursors like 2-(4-fluorobenzylamino)ethanol. This novel process utilizes low-cost, commercially abundant starting materials such as phthalimide potassium salt and 2-aminoethanol. By eliminating the need for pre-synthesized expensive amines and shortening the overall reaction sequence, significant raw material cost reductions are achieved.
Q: What represents the key innovation in the final alkylation step?
A: The innovation lies in the specific base system used for the final N-alkylation of the morpholine ring. The patent specifies a combination of a tert-butanol metal salt (like sodium tert-butoxide) and triethylamine. This dual-base system optimizes the nucleophilicity of the amine while minimizing side reactions, resulting in significantly improved yields and product purity compared to single-base systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4-fluorobenzyl)-2-aminomethyl morpholine Supplier
As the global demand for high-quality gastrointestinal therapeutics continues to rise, the need for a dependable source of critical intermediates like 4-(4-fluorobenzyl)-2-aminomethyl morpholine has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep technical expertise to deliver solutions that align with the rigorous standards of the pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercial manufacturing. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate we supply supports the safety and efficacy of your final drug product.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating exactly how switching to our optimized intermediate can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a stable, high-quality supply of Mosapride intermediates, driving your project forward with confidence and efficiency.
