Advanced Synthesis of Halofuginone Intermediates: A Technical Breakthrough for Commercial Scale-Up
Advanced Synthesis of Halofuginone Intermediates: A Technical Breakthrough for Commercial Scale-Up
The global demand for effective coccidiostats and potential anti-fibrotic agents has placed Halofuginone, a potent quinazolinone alkaloid, at the forefront of veterinary and pharmaceutical research. As depicted in 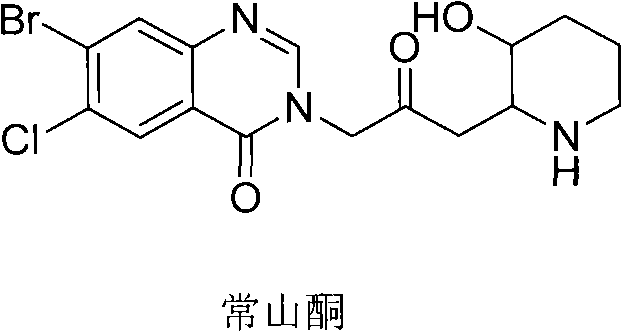 , the molecular architecture of Halofuginone requires precise construction of its heterocyclic framework. Patent CN102070514A, filed in May 2011, introduces a transformative methodology for preparing key intermediates, specifically targeting the synthesis of Compound D and its derivatives. This intellectual property addresses critical bottlenecks in the existing supply chain by optimizing the Van Braun reaction and subsequent Claisen rearrangement. For R&D directors and procurement specialists, understanding this patented route is vital, as it offers a pathway to high-purity pharmaceutical intermediates with significantly reduced environmental footprint and operational complexity compared to legacy processes.
, the molecular architecture of Halofuginone requires precise construction of its heterocyclic framework. Patent CN102070514A, filed in May 2011, introduces a transformative methodology for preparing key intermediates, specifically targeting the synthesis of Compound D and its derivatives. This intellectual property addresses critical bottlenecks in the existing supply chain by optimizing the Van Braun reaction and subsequent Claisen rearrangement. For R&D directors and procurement specialists, understanding this patented route is vital, as it offers a pathway to high-purity pharmaceutical intermediates with significantly reduced environmental footprint and operational complexity compared to legacy processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industry standard largely relied on the synthetic route established by Takeuchi et al., as illustrated in 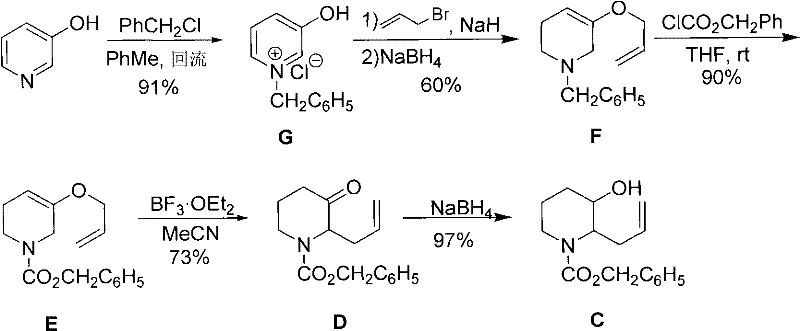 . This conventional approach suffers from severe inefficiencies that hinder commercial scale-up of complex quinazolinone derivatives. Specifically, the Van Braun reaction step traditionally requires a massive excess of benzyl chloroformate—approximately 6 equivalents relative to the substrate—to drive the reaction to completion. Despite this excessive usage, conversion often remains incomplete, leaving significant amounts of unreacted starting material. This necessitates rigorous and costly purification steps, typically involving long chromatography columns and extended gradient elution times. Furthermore, the use of acetonitrile as a solvent for the Claisen rearrangement introduces toxicity concerns and complicates waste management, creating substantial barriers for cost reduction in API manufacturing.
. This conventional approach suffers from severe inefficiencies that hinder commercial scale-up of complex quinazolinone derivatives. Specifically, the Van Braun reaction step traditionally requires a massive excess of benzyl chloroformate—approximately 6 equivalents relative to the substrate—to drive the reaction to completion. Despite this excessive usage, conversion often remains incomplete, leaving significant amounts of unreacted starting material. This necessitates rigorous and costly purification steps, typically involving long chromatography columns and extended gradient elution times. Furthermore, the use of acetonitrile as a solvent for the Claisen rearrangement introduces toxicity concerns and complicates waste management, creating substantial barriers for cost reduction in API manufacturing.
The Novel Approach
The patented methodology fundamentally re-engineers the reaction dynamics to overcome these historical deficiencies. As shown in 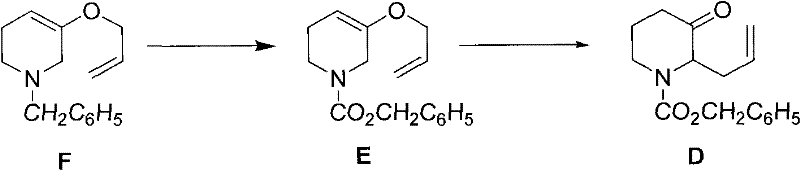 , the core innovation lies in reversing the addition order during the Van Braun reaction. Instead of adding the reagent to the substrate, the solution of Compound F is added dropwise into the benzyl chloroformate solution. This strategic modification ensures that the reagent is always in excess locally, driving the reaction to full completion while allowing the total amount of benzyl chloroformate to be reduced to approximately 4 equivalents. Moreover, the process enables a telescoped one-pot procedure where the intermediate Compound E is not isolated but directly subjected to Claisen rearrangement. This seamless integration not only simplifies the operational workflow but also facilitates the recovery of solvents and reagents, positioning this technology as a superior choice for any reliable pharmaceutical intermediate supplier aiming for sustainability.
, the core innovation lies in reversing the addition order during the Van Braun reaction. Instead of adding the reagent to the substrate, the solution of Compound F is added dropwise into the benzyl chloroformate solution. This strategic modification ensures that the reagent is always in excess locally, driving the reaction to full completion while allowing the total amount of benzyl chloroformate to be reduced to approximately 4 equivalents. Moreover, the process enables a telescoped one-pot procedure where the intermediate Compound E is not isolated but directly subjected to Claisen rearrangement. This seamless integration not only simplifies the operational workflow but also facilitates the recovery of solvents and reagents, positioning this technology as a superior choice for any reliable pharmaceutical intermediate supplier aiming for sustainability.
Mechanistic Insights into Van Braun Reaction and Claisen Rearrangement
The success of this synthetic route hinges on the precise control of nucleophilic substitution and pericyclic rearrangement mechanisms. In the Van Braun reaction, the nitrogen atom of the piperidine ring in Compound F acts as a nucleophile, attacking the carbonyl carbon of benzyl chloroformate. By maintaining the reaction temperature between 0°C and 25°C and controlling the滴加 rate to keep the internal temperature below 0°C initially, the formation of side products is minimized. The reverse addition protocol ensures that the concentration of the electrophile (benzyl chloroformate) remains high relative to the nucleophile throughout the addition phase, effectively suppressing competitive hydrolysis or incomplete substitution. This kinetic control is paramount for achieving the high conversion rates necessary for industrial viability without relying on massive stoichiometric excesses that complicate downstream processing.
Following the formation of the carbamate intermediate (Compound E), the system undergoes a thermal Claisen rearrangement. This [3,3]-sigmatropic rearrangement is highly sensitive to steric and electronic factors. The patent specifies heating the reaction mixture to between 50°C and 80°C, preferably at the reflux temperature of tetrahydrofuran (65-70°C). Under these conditions, the allyl group migrates selectively to the 2-position of the piperidine ring. This regioselectivity is critical because alternative migration paths would generate structural isomers that are chemically similar and notoriously difficult to separate via crystallization or chromatography. The ability to selectively obtain the 2-position product ensures that the final intermediate possesses the correct stereochemical and structural alignment required for the subsequent cyclization into the quinazolinone core of Halofuginone.
How to Synthesize Halofuginone Intermediate Efficiently
Implementing this patented process requires strict adherence to the optimized parameters regarding solvent choice, temperature gradients, and reagent ratios. The preferred embodiment utilizes tetrahydrofuran (THF) as the primary solvent due to its favorable boiling point and solubility profile for both the Van Braun and Claisen steps. The process begins with the preparation of a solution of Compound F in THF, which is then carefully introduced into a solution of benzyl chloroformate. Following the completion of the carbamate formation, the reaction mixture is heated to induce rearrangement. A key feature of this protocol is the integrated recovery system: THF is recovered via atmospheric distillation, followed by the recovery of excess benzyl chloroformate under reduced pressure. This closed-loop approach maximizes atom economy. For detailed operational parameters and specific molar ratios, please refer to the standardized synthesis guide below.
- Perform Van Braun reaction by dropwise adding Compound F solution into benzyl chloroformate solution at 0-25°C to ensure complete conversion.
- Directly subject the resulting mixture to Claisen rearrangement at 50-80°C in a one-pot procedure to yield Compound D.
- Recover solvents and excess reagents via distillation to maximize efficiency and minimize environmental impact.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route represents a strategic opportunity to optimize the supply of critical veterinary and pharmaceutical ingredients. The elimination of excessive reagent usage directly translates to raw material cost savings, while the simplified workup procedures reduce labor and utility costs associated with prolonged purification. By adopting a process that minimizes the use of toxic solvents like acetonitrile and reduces the generation of hazardous waste, manufacturers can also achieve significant improvements in environmental compliance and safety standards. These factors collectively enhance the resilience of the supply chain against regulatory shifts and raw material price volatility.
- Cost Reduction in Manufacturing: The reduction of benzyl chloroformate usage from 6 equivalents to approximately 4 equivalents results in substantial direct material savings. Furthermore, the ability to recover and recycle both the solvent (THF) and the excess reagent via distillation drastically lowers the net consumption of chemicals per kilogram of product. The one-pot nature of the synthesis eliminates the isolation and drying steps for intermediate Compound E, thereby reducing energy consumption and processing time. These efficiencies compound to offer a significantly lower cost of goods sold (COGS) compared to the traditional Takeuchi route.
- Enhanced Supply Chain Reliability: The robustness of the reverse-addition Van Braun reaction ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed reactions or off-spec material. Since the process relies on commercially available starting materials and standard unit operations (distillation, filtration), it is less susceptible to supply disruptions associated with specialized catalysts or exotic reagents. This reliability is crucial for maintaining continuous production schedules for high-purity halofuginone intermediates required by downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scale, avoiding the bottlenecks of column chromatography which are impractical at multi-ton scales. By replacing chromatographic purification with distillation and crystallization techniques, the method facilitates commercial scale-up from pilot plants to full industrial production. Additionally, the reduced toxicity profile and the implementation of solvent recovery systems align with modern green chemistry principles, minimizing the environmental burden and simplifying the permitting process for new manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims presented in CN102070514A, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer.
Q: How does the new Van Braun protocol improve upon traditional methods?
A: The novel protocol reverses the addition order, dropping Compound F into benzyl chloroformate. This ensures complete reaction of the starting material, reduces the required excess of benzyl chloroformate from 6 equivalents to approximately 4 equivalents, and simplifies downstream purification by minimizing unreacted reagents.
Q: What are the scalability advantages of the one-pot synthesis described in CN102070514A?
A: The one-pot method eliminates the isolation of intermediate Compound E, significantly reducing solvent consumption and operational time. It allows for the direct recovery of tetrahydrofuran and benzyl chloroformate via distillation, enhancing overall process economics and environmental compliance for large-scale production.
Q: Why is regioselectivity critical in the Claisen rearrangement step?
A: High regioselectivity for the 2-position product is essential to avoid isomeric impurities that are difficult to separate. The optimized conditions in this patent ensure selective formation of the desired carbon-carbon bond, maintaining high purity standards required for subsequent pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halofuginone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the broader pharmaceutical value chain. Our technical team has extensively analyzed the pathways described in CN102070514A and possesses the expertise to implement these advanced protocols at an industrial level. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Halofuginone intermediate meets the exacting standards required for veterinary and human health applications.
We invite global partners to collaborate with us to leverage this innovative technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a sustainable and cost-effective supply of high-quality pharmaceutical intermediates.
