Revolutionizing Quinazolinone Synthesis: A Metal-Free Photocatalytic Route for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for constructing nitrogen-containing heterocycles, which serve as critical scaffolds in drug discovery. Patent CN116159594A presents a groundbreaking advancement in this domain by introducing the application of pyridinium small molecules as photocatalysts in the tandem reaction of alcohols and amines. This innovation specifically targets the synthesis of quinazolinone derivatives and benzothiadiazine 1,1-dioxide derivatives, compounds renowned for their potent anti-inflammatory, antiviral, and anticancer activities. Unlike traditional thermal methods that rely on harsh conditions, this photo-driven approach operates under mild conditions with high atom economy. For R&D directors and procurement managers alike, this technology represents a paradigm shift towards greener chemistry, offering a reliable pharmaceutical intermediate supplier solution that aligns with modern regulatory and environmental standards while maintaining exceptional product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone and benzothiadiazine 1,1-dioxide derivatives has been dominated by condensation reactions involving amidines, ketones, or nitriles, often necessitating the use of harmful oxidants such as potassium permanganate or TBHP. More recent advancements have attempted to utilize alcohols as substrates through metal-catalyzed cross-dehydrogenative coupling; however, these methods typically depend on expensive noble metals like palladium, platinum, or iridium. These homogeneous metal catalysts pose significant challenges in industrial settings, primarily due to the difficulty in separating and recovering the catalyst from the product mixture, which can lead to unacceptable levels of heavy metal residues in the final active pharmaceutical ingredient. Furthermore, the requirement for complex ligands and additional additives increases the overall cost and complexity of the process, creating bottlenecks in cost reduction in API manufacturing and complicating the supply chain for high-purity intermediates.
The Novel Approach
The methodology disclosed in the patent overcomes these hurdles by employing a metal-free pyridinium small molecule photocatalyst that facilitates the tandem reaction between anthranilamides or anthranilsulfonamides and benzyl alcohols. This novel route operates under air atmosphere with 365nm light irradiation, utilizing a benign solvent system of acetonitrile, ethanol, and water. A standout feature of this process is the spontaneous separation of the product; the quinazolinone and benzothiadiazine derivatives precipitate directly from the reaction mixture, thereby eliminating the need for energy-intensive and solvent-heavy silica gel column chromatography. This drastic simplification of the workup procedure not only accelerates the production timeline but also significantly reduces waste generation. 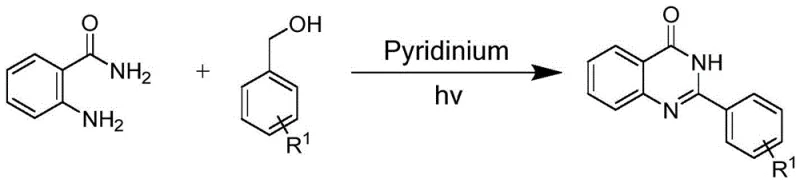
Mechanistic Insights into Pyridinium Small Molecule Photocatalysis
The efficacy of this synthetic strategy lies in the unique dual functionality of the pyridinium small molecule photocatalyst, which acts as both an excellent electron acceptor and a Lewis acid. Upon irradiation, the catalyst undergoes photoinduced electron transfer to generate colored pyridinium radicals, which are easily re-oxidized by molecular oxygen in the air to regenerate the initial state while activating oxygen into superoxide free radicals. This redox cycle drives the aerobic oxidation of the alcohol substrate to the corresponding aldehyde in situ. Simultaneously, the electron-deficient pyridinium unit activates the carbonyl group of the intermediate aldehyde, facilitating nucleophilic attack by the amine functional group of the anthranilamide or anthranilsulfonamide. This synergistic activation enables a seamless one-pot multi-step reaction sequence involving intermolecular tandem addition followed by cyclodehydrogenation to form the target heterocyclic ring system with high selectivity.
From an impurity control perspective, the metal-free nature of this catalytic system is paramount for ensuring the purity profile required for pharmaceutical applications. Traditional metal-catalyzed routes often introduce trace metal impurities that require rigorous and costly purification steps to meet ICH Q3D guidelines. By eliminating transition metals entirely, this photocatalytic method inherently produces a cleaner crude product. Furthermore, the specificity of the photo-induced radical mechanism minimizes side reactions commonly associated with harsh thermal oxidants, resulting in yields consistently above 90% for a wide range of substrates. The structural integrity of the catalyst, shown below, ensures stability throughout the reaction, preventing catalyst degradation products from contaminating the final batch. 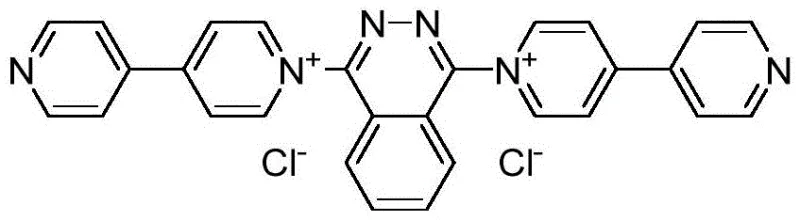
How to Synthesize Quinazolinone Derivatives Efficiently
To implement this technology for the commercial scale-up of complex pharmaceutical intermediates, the process begins with the preparation of the pyridinium photocatalyst via a Menshutkin reaction between 4,4'-bipyridine and 1,4-dichlorophthalazine. For the synthesis of the target heterocycles, the anthranilamide or anthranilsulfonamide is mixed with the aromatic alcohol and a catalytic amount (3%-5%) of the pyridinium powder in the specified ternary solvent system. The reaction vessel is then exposed to 365nm light under ambient air conditions for 5 to 10 hours. Detailed standardized synthesis steps see the guide below.
- Dissolve anthranilamide, aromatic alcohol, and the pyridinium small molecule photocatalyst (3%-5% loading) in a solvent mixture of acetonitrile, ethanol, and water.
- Stir the reaction mixture under an air atmosphere while irradiating with 365nm light to initiate the tandem addition and cyclodehydrogenation.
- After 5-10 hours, collect the spontaneously precipitated product via filtration, eliminating the need for silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible operational efficiencies and risk mitigation. The elimination of noble metals removes the volatility associated with the pricing and availability of resources like palladium and iridium, stabilizing the cost structure of raw materials. Additionally, the use of alcohols derived from renewable plant fermentation ensures a sustainable and abundant feedstock supply, reducing dependency on petrochemical-derived precursors. The simplified downstream processing, characterized by spontaneous precipitation and filtration rather than chromatography, drastically reduces solvent consumption and waste disposal costs, contributing to substantial cost savings in the overall manufacturing budget.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the avoidance of stoichiometric oxidants significantly lower the direct material costs associated with production. Moreover, the elimination of silica gel column chromatography reduces the consumption of large volumes of organic solvents and the labor hours required for purification, leading to a more economically viable process for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials such as benzyl alcohols and anthranilamides, which are commercially accessible in bulk quantities, the supply chain becomes more robust against disruptions. The metal-free nature of the catalyst also simplifies regulatory compliance regarding heavy metal limits, reducing the risk of batch rejection and ensuring consistent delivery schedules for critical drug substances.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of air as the terminal oxidant make this process highly scalable from laboratory to industrial production without the safety hazards associated with high-pressure oxygen or explosive peroxides. The green chemistry principles embedded in this method, including high atom economy and reduced waste generation, align perfectly with increasingly stringent environmental regulations, facilitating smoother permitting and operation of manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyridinium photocatalytic system. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of this technology for industrial partners seeking to optimize their synthetic routes.
Q: Does this photocatalytic method require expensive transition metal catalysts?
A: No, the method described in patent CN116159594A utilizes a metal-free pyridinium small molecule photocatalyst, completely eliminating the need for costly noble metals like Palladium or Iridium and avoiding heavy metal contamination in the final API.
Q: How is product purification handled in this new synthetic route?
A: A key advantage of this technology is spontaneous separation. The quinazolinone and benzothiadiazine derivatives precipitate directly from the reaction mixture, allowing for simple filtration without the need for tedious silica gel column chromatography.
Q: What are the environmental benefits of using alcohols as substrates?
A: Using alcohols derived from renewable plant fermentation as substrates enhances atom economy. The only by-products are molecular hydrogen or water, making the process significantly greener compared to traditional methods requiring stoichiometric oxidants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinone Supplier
As the global demand for high-quality heterocyclic intermediates continues to rise, partnering with an experienced CDMO is essential for translating innovative academic research into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale photocatalysis to industrial manufacturing is seamless and efficient. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of quinazolinone or benzothiadiazine derivative meets the highest international standards, providing our clients with the confidence needed to advance their drug development pipelines.
We invite potential partners to engage with our technical procurement team to discuss how this metal-free photocatalytic technology can be integrated into your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits tailored to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project leverages the full potential of this green and efficient synthetic methodology.
