Advanced Palladium-Catalyzed Carbonylation for Scalable Alkynone Manufacturing Solutions
Advanced Palladium-Catalyzed Carbonylation for Scalable Alkynone Manufacturing Solutions
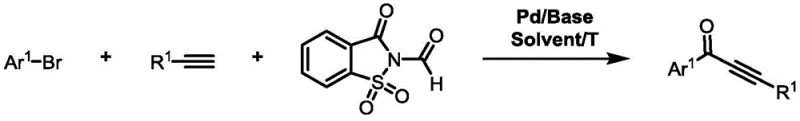
The landscape of fine chemical synthesis is constantly evolving, driven by the dual imperatives of safety and cost-efficiency. A significant breakthrough in this domain is detailed in patent CN113563164A, which introduces a robust methodology for the preparation of alkynone compounds. These conjugated structural units are pivotal intermediates in the construction of complex pharmaceutical agents and advanced functional materials. The core innovation lies in the strategic replacement of hazardous gaseous carbon monoxide with N-formyl saccharin, a solid and stable reagent. This shift not only mitigates the extreme safety risks associated with traditional carbonylation processes but also streamlines the operational workflow for large-scale production. By leveraging a palladium-catalyzed insertion reaction between brominated aromatic hydrocarbons and terminal alkynes, this technology offers a greener, more accessible pathway to high-value chemical building blocks that are essential for modern drug discovery and development pipelines.
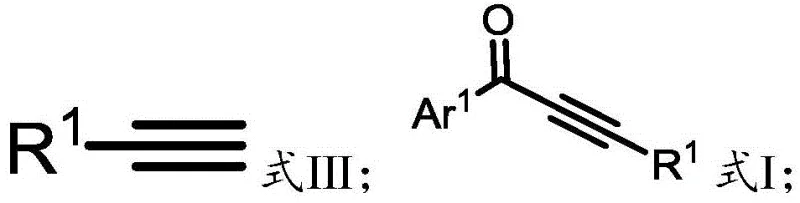
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkynones has relied heavily on methods that pose significant logistical and safety challenges for industrial manufacturers. Traditional routes often necessitate the use of acyl chlorides or anhydrides reacting with alkynyl metal reagents, which can be moisture-sensitive and generate substantial stoichiometric waste. More critically, palladium-catalyzed carbonylation reactions have typically depended on the direct use of carbon monoxide gas. Handling CO requires specialized high-pressure equipment and rigorous safety protocols due to its high toxicity and flammability, creating a barrier to entry for many facilities. Furthermore, conventional protocols frequently mandate the use of aryl iodides as substrates because of their higher reactivity in oxidative addition steps. However, aryl iodides are exponentially more expensive than their bromide counterparts, inflating the raw material costs and rendering the process economically unviable for cost-sensitive applications such as generic pharmaceutical intermediate manufacturing.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this synthetic landscape by introducing N-formyl saccharin as a superior carbon monoxide substitute. This solid reagent releases CO in situ under mild thermal conditions, effectively bypassing the need for gas cylinders and high-pressure reactors. The process is designed to work efficiently with aryl bromides, which are widely available and significantly cheaper than iodides, thus unlocking substantial cost reduction in fine chemical manufacturing. The reaction operates under relatively mild temperatures, typically around 100°C to 120°C, using common bases like potassium carbonate and standard solvents such as toluene. This combination of factors results in a catalytic system that is not only green and environmentally friendly but also operationally simple, allowing for easier separation and purification of the final alkynone products with high efficiency and yield.
Mechanistic Insights into Pd-Catalyzed Carbonyl Insertion
At the heart of this transformation is a sophisticated palladium-catalyzed cycle that orchestrates the coupling of three distinct components: the aryl bromide, the terminal alkyne, and the CO source derived from N-formyl saccharin. The mechanism initiates with the oxidative addition of the palladium(0) species into the carbon-bromine bond of the aromatic substrate, forming an aryl-palladium(II) intermediate. This step is crucial and is facilitated by the specific electronic properties of the ligand environment, allowing even the less reactive aryl bromides to participate effectively. Subsequently, the N-formyl saccharin undergoes decomposition or activation to release carbon monoxide, which then inserts into the palladium-carbon bond to generate an acyl-palladium complex. This acyl species then reacts with the terminal alkyne, likely via a transmetallation or coordination-insertion pathway typical of Sonogashira-type couplings, followed by reductive elimination to release the desired alkynone product and regenerate the active palladium catalyst.
From a quality control perspective, the choice of N-formyl saccharin and the specific reaction conditions play a vital role in impurity management. The mild thermal profile prevents the thermal degradation of sensitive functional groups that might be present on the aromatic ring or the alkyne chain. Moreover, the use of a solid CO source ensures a controlled, steady release of carbon monoxide, preventing local concentration spikes that could lead to side reactions such as homocoupling of the alkyne or over-carbonylation. The byproduct of the CO source, saccharin, is chemically inert under these conditions and can be easily removed during the aqueous workup or column chromatography, ensuring that the final API intermediate meets stringent purity specifications required by regulatory bodies. This mechanistic elegance translates directly into a cleaner reaction profile and reduced downstream processing burdens.
How to Synthesize Alkynone Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and environmental controls to maximize yield and reproducibility. The process involves charging a reactor with the aryl bromide substrate, the terminal alkyne, and N-formyl saccharin in a molar ratio that favors complete conversion, typically utilizing a slight excess of the alkyne and the CO source. The reaction is conducted in a non-aqueous solvent system, such as toluene or dioxane, under an inert nitrogen atmosphere to prevent catalyst oxidation. A palladium catalyst, such as palladium acetate, is introduced along with a base like potassium carbonate to facilitate the deprotonation of the alkyne and neutralize acidic byproducts. The detailed standardized synthesis steps are outlined in the guide below.
- Combine aryl bromide, terminal alkyne, N-formyl saccharin, and base (e.g., K2CO3) in a non-aqueous solvent like toluene under inert atmosphere.
- Add palladium catalyst (e.g., Pd(OAc)2) and heat the mixture to 100-120°C with stirring until substrate consumption is confirmed by TLC.
- Purify the crude reaction mixture via silica gel column chromatography using petroleum ether and diethyl ether to isolate the high-purity alkynone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology represents a strategic opportunity to optimize both cost structures and operational resilience. The shift from aryl iodides to aryl bromides immediately lowers the bill of materials, as bromides are commodity chemicals with stable global supply chains compared to the more volatile iodide market. Furthermore, the elimination of gaseous carbon monoxide removes the need for specialized gas handling infrastructure and the associated regulatory compliance costs, simplifying the facility requirements for production. The use of N-formyl saccharin, a stable solid, enhances storage safety and reduces the risks of supply disruption caused by hazardous material transport restrictions. These factors collectively contribute to a more robust and cost-effective supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive raw materials with cost-effective alternatives. By enabling the use of aryl bromides instead of aryl iodides, the direct material costs are significantly reduced without sacrificing reaction efficiency. Additionally, the avoidance of high-pressure gas equipment and the use of standard heating methods lower the capital expenditure and energy consumption associated with the process. The high yields reported in the patent embodiments mean that less raw material is wasted, further improving the overall atom economy and reducing the cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of commercially available and stable reagents. N-formyl saccharin is easy to store and handle, unlike compressed gases which require special cylinders and logistics. The robustness of the catalytic system allows for consistent batch-to-batch performance, minimizing the risk of production delays due to failed reactions. This stability ensures that downstream customers receive their orders on time, maintaining the continuity of their own manufacturing schedules for finished drugs or agrochemicals.
- Scalability and Environmental Compliance: The process is inherently scalable due to its mild operating conditions and the absence of hazardous gases. Scaling up from laboratory to pilot or commercial plant does not require complex engineering solutions for gas containment, making the transition smoother and faster. From an environmental standpoint, the catalytic system is greener, generating less hazardous waste compared to traditional methods involving acid chlorides or toxic gases. This aligns with increasingly strict environmental regulations and corporate sustainability goals, reducing the burden of waste treatment and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkynone synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production lines or new product development projects.
Q: Why is N-formyl saccharin preferred over carbon monoxide gas in this synthesis?
A: N-formyl saccharin serves as a solid, stable, and non-toxic carbon monoxide surrogate, eliminating the severe safety hazards associated with handling explosive and highly toxic CO gas in industrial settings.
Q: Can this method utilize cheaper aryl bromides instead of aryl iodides?
A: Yes, the patented process specifically enables the efficient use of aryl bromides, which are significantly more cost-effective than aryl iodides, thereby reducing raw material costs without compromising yield.
Q: What represents the typical purity level achievable with this protocol?
A: The method consistently delivers alkynone compounds with purity exceeding 99%, as demonstrated in multiple embodiments where rigorous purification via column chromatography ensures minimal impurity profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkynone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed carbonylation technology for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including inert atmosphere operations and rigorous QC labs that enforce stringent purity specifications to guarantee the quality of every batch we deliver.
We invite you to collaborate with us to leverage this advanced synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how switching to this bromide-based protocol can optimize your budget. Please contact our technical procurement team today to request specific COA data for similar alkynone compounds and to discuss route feasibility assessments for your target molecules, ensuring a secure and efficient supply chain for your critical intermediates.
