Scalable Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles Using Glucose as a Sustainable Carbon Source
Scalable Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles Using Glucose as a Sustainable Carbon Source
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and cost-effective routes for synthesizing complex heterocyclic scaffolds, particularly those containing fluorine atoms which are critical for modulating metabolic stability and bioavailability. Patent CN113880781A introduces a groundbreaking methodology for the preparation of 3-trifluoromethyl-substituted 1,2,4-triazole compounds, utilizing glucose—a ubiquitous and renewable biomass resource—as the primary carbon source. This innovation represents a significant departure from traditional synthetic pathways that often rely on expensive or hazardous reagents, offering a greener alternative that aligns with modern green chemistry principles. The structural motif of 3-trifluoromethyl-1,2,4-triazole is a privileged scaffold found in numerous high-value active pharmaceutical ingredients (APIs), including blockbuster drugs like Sitagliptin, as well as various agrochemicals and functional materials. By leveraging the natural abundance of glucose, this process not only reduces raw material costs but also simplifies the supply chain logistics for manufacturers seeking a reliable pharmaceutical intermediate supplier.
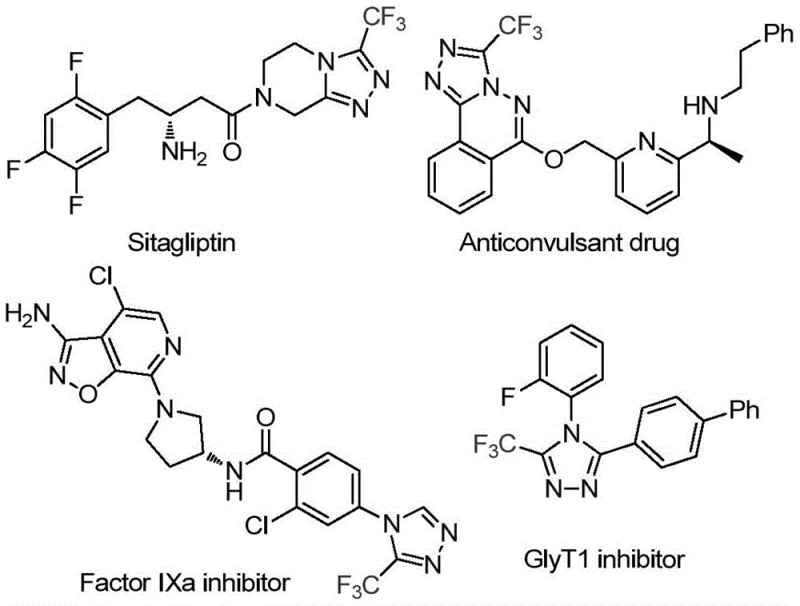
The strategic importance of this technology cannot be overstated for R&D directors and procurement managers alike, as it addresses the dual challenges of economic viability and environmental compliance. The ability to access these valuable nitrogen-containing heterocycles through a cascade reaction driven by a simple sugar opens new avenues for the design of diverse libraries of compounds. Furthermore, the mild reaction conditions described in the patent suggest that this method can be readily integrated into existing manufacturing infrastructure without requiring specialized high-pressure or cryogenic equipment. As we delve deeper into the technical specifics, it becomes clear that this glucose-mediated approach offers a robust solution for the cost reduction in API manufacturing, providing a competitive edge in the global market for specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant technical and economic hurdles that hinder large-scale production. Traditional methodologies often necessitate the use of highly reactive and expensive fluorinating agents, or they require multi-step sequences involving harsh conditions such as strong bases, elevated temperatures, or strictly anhydrous environments. These conventional routes frequently suffer from poor atom economy, generating substantial amounts of chemical waste that complicate downstream purification and increase the burden on waste treatment facilities. Moreover, the reliance on specialized reagents that are sensitive to moisture or oxygen imposes stringent operational requirements, demanding inert atmosphere gloveboxes or Schlenk lines which drastically increase capital expenditure and operational complexity. For supply chain heads, these factors translate into longer lead times, higher inventory costs for sensitive reagents, and increased risk of batch-to-batch variability due to the sensitivity of the reaction parameters. The cumulative effect of these limitations is a manufacturing process that is neither economically efficient nor environmentally sustainable, creating a pressing need for innovation in this chemical space.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in patent CN113880781A utilizes a trifluoromethanesulfonic acid (TfOH) catalyzed cascade cyclization reaction that transforms glucose directly into the desired triazole scaffold. This method operates under remarkably mild conditions, typically requiring temperatures between 70°C and 90°C for just 2 to 4 hours, eliminating the need for energy-intensive heating or cooling cycles. The use of glucose as a carbon-carbon synthon is particularly ingenious; under acidic conditions, glucose undergoes cleavage to generate an aldehyde intermediate in situ, which then condenses with trifluoroacetimidoyl hydrazide to form a hydrazone. This intermediate subsequently undergoes intramolecular nucleophilic addition and cyclization, followed by oxidative aromatization using tert-butyl hydroperoxide (TBHP) to yield the final 3-trifluoromethyl-1,2,4-triazole product. This one-pot strategy significantly streamlines the synthetic route, reducing the number of isolation steps and minimizing solvent consumption. The robustness of this system allows for the tolerance of various functional groups on the aromatic ring, enabling the synthesis of a wide array of derivatives without the need for protecting group chemistry, thereby enhancing the overall efficiency and versatility of the process.
Mechanistic Insights into TfOH-Catalyzed Cascade Cyclization
The mechanistic pathway of this transformation is a fascinating example of biomimetic chemistry, where a simple sugar is deconstructed and rebuilt into a complex heterocycle through a series of acid-promoted steps. The reaction initiates with the activation of glucose by the strong Brønsted acid catalyst, trifluoromethanesulfonic acid, which facilitates the cleavage of the carbohydrate backbone to release a reactive aldehyde species. This aldehyde immediately engages in a condensation reaction with the trifluoroacetimidoyl hydrazide, forming a hydrazone intermediate that serves as the precursor for ring closure. The subsequent intramolecular nucleophilic attack by the nitrogen atom onto the electrophilic carbon center drives the cyclization process, constructing the five-membered triazole ring system. Finally, the introduction of tert-butyl hydroperoxide acts as a terminal oxidant, facilitating the dehydrogenation and aromatization of the dihydro-triazole intermediate to yield the stable, aromatic 3-trifluoromethyl-1,2,4-triazole core. This cascade sequence is highly efficient because each step flows logically into the next without the need for intermediate workups, maximizing the throughput of the reaction vessel.
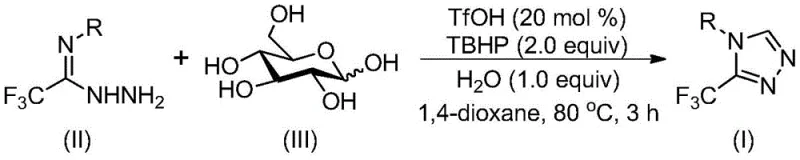
From an impurity control perspective, this mechanism offers distinct advantages over traditional metal-catalyzed couplings. Since the reaction relies on organocatalysis (TfOH) rather than transition metals, there is no risk of heavy metal contamination in the final product, a critical quality attribute for pharmaceutical intermediates intended for human use. The absence of metal catalysts also simplifies the purification protocol, often allowing for straightforward filtration and column chromatography without the need for specialized scavengers to remove trace metals. Furthermore, the use of aqueous TBHP and water as an additive helps to moderate the reaction exotherm and solubilize polar intermediates, reducing the formation of polymeric byproducts that often plague acid-catalyzed carbohydrate reactions. The selectivity of the cyclization is governed by the electronic properties of the hydrazide and the steric environment of the glucose-derived aldehyde, ensuring that the desired 1,2,4-regioisomer is formed predominantly. This high level of chemoselectivity ensures a clean crude profile, which is essential for maintaining high purity specifications in commercial manufacturing.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale discovery and pilot-plant production. The procedure involves charging a reaction vessel with the requisite amounts of trifluoromethanesulfonic acid, 70% aqueous tert-butyl hydroperoxide, water, trifluoroethylimide hydrazide, and glucose in a suitable organic solvent such as 1,4-dioxane. The mixture is then heated to the specified temperature range and stirred until conversion is complete, as monitored by standard analytical techniques. Following the reaction, the workup is uncomplicated, typically involving filtration to remove any insoluble particulates followed by purification via silica gel column chromatography to isolate the target compound in high purity. The detailed standardized synthesis steps, including precise molar ratios and specific safety precautions for handling strong acids and oxidizers, are outlined in the comprehensive guide below.
- Combine trifluoromethanesulfonic acid, tert-butyl hydroperoxide (70% aqueous), water, trifluoroethylimide hydrazide, and glucose in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 2 to 4 hours to ensure complete conversion.
- Upon completion, perform post-processing including filtration and silica gel column chromatography to isolate the pure 3-trifluoromethyl-substituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this glucose-based synthesis route presents a compelling value proposition centered on cost optimization and supply security. The shift from petrochemical-derived precursors to biomass-based feedstocks like glucose fundamentally alters the cost structure of the manufacturing process, decoupling production expenses from the volatility of fossil fuel markets. Since glucose is one of the most abundant and inexpensive carbohydrates available globally, its utilization as a key starting material drives a substantial reduction in raw material costs. Additionally, the elimination of expensive transition metal catalysts and the avoidance of stringent anhydrous conditions further contribute to significant cost savings by reducing the need for specialized equipment and energy-intensive drying processes. The simplified workup procedure also translates to lower labor costs and reduced solvent usage, enhancing the overall economic efficiency of the production line.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the use of low-cost, renewable starting materials and the minimization of processing steps. By replacing expensive synthetic building blocks with glucose, manufacturers can achieve a drastic reduction in the bill of materials, which is a major component of the total production cost. Furthermore, the mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for exotic alloys resistant to extreme corrosion or high pressure, thereby lowering capital depreciation costs. The high reaction efficiency and selectivity mean that less material is wasted in side reactions, improving the overall yield and reducing the cost per kilogram of the final API intermediate. These factors combined create a leaner, more cost-effective manufacturing model that enhances profit margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the widespread availability of the key reagents used in this synthesis. Glucose is a commodity chemical produced in massive quantities worldwide for the food and fermentation industries, ensuring a stable and continuous supply that is immune to the geopolitical disruptions often affecting specialized chemical intermediates. Similarly, trifluoromethanesulfonic acid and tert-butyl hydroperoxide are commercially available bulk chemicals with established supply chains, reducing the risk of stockouts or delivery delays. The robustness of the reaction, which tolerates the presence of water and does not require inert atmospheres, also means that production can continue uninterrupted even if there are minor fluctuations in utility quality or environmental controls. This reliability is crucial for maintaining consistent inventory levels and meeting the just-in-time delivery demands of downstream pharmaceutical customers.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this process aligns perfectly with the increasing regulatory pressure for greener manufacturing practices. The use of a biomass feedstock reduces the carbon footprint of the synthesis, while the absence of heavy metals eliminates the generation of toxic hazardous waste streams that require costly disposal. The reaction has already been demonstrated to be effective at the gram scale, and the simplicity of the one-pot protocol suggests that scaling up to kilogram or tonne levels will be straightforward without encountering significant engineering bottlenecks. The ability to run the reaction in common solvents like 1,4-dioxane, which can be recovered and recycled, further supports sustainable operations. For companies aiming to meet ESG (Environmental, Social, and Governance) goals, adopting this technology demonstrates a commitment to sustainable chemistry and responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this glucose-mediated triazole synthesis. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on the feasibility and scope of the technology. Understanding these details is essential for technical teams evaluating the integration of this route into their existing portfolios.
Q: What are the key advantages of using glucose in this synthesis?
A: Using glucose as a carbon source offers significant advantages including low cost, wide availability as a biomass feedstock, and the elimination of harsh anhydrous or anaerobic reaction conditions, thereby simplifying the operational complexity.
Q: What is the typical reaction temperature and time for this process?
A: The reaction typically proceeds under mild thermal conditions, requiring heating to between 70°C and 90°C for a duration of 2 to 4 hours, which is conducive to energy-efficient manufacturing.
Q: Can this method be scaled for industrial production?
A: Yes, the patent data indicates that the method has been successfully extended to gram-level reactions with high efficiency, demonstrating strong potential for commercial scale-up due to the simplicity of the workup and the stability of the reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this glucose-based synthetic route for the production of high-value fluorinated heterocycles. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients can transition seamlessly from bench-scale discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of acid-catalyzed cascade reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole meets the highest industry standards. We are committed to delivering not just a chemical product, but a comprehensive solution that optimizes your supply chain and accelerates your time to market.
We invite you to engage with our technical procurement team to discuss how this innovative technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this biomass-derived route for your specific application. We encourage you to contact us today to obtain specific COA data for our reference standards and to schedule a consultation for route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and commercial viability.
