Advanced Manufacturing of Sitagliptin Phosphate via Chiral Pool Strategy
Advanced Manufacturing of Sitagliptin Phosphate via Chiral Pool Strategy
In the rapidly evolving landscape of type 2 diabetes therapeutics, the efficient production of Dipeptidyl Peptidase-IV (DPP-IV) inhibitors remains a critical priority for global pharmaceutical manufacturers. Patent CN102153559A discloses a groundbreaking methodology for synthesizing Sitagliptin phosphate and its derivatives, addressing long-standing challenges in cost, scalability, and optical purity. This novel approach utilizes L-aspartic acid as a foundational chiral building block, bypassing the need for complex asymmetric catalysis in the initial stereochemical setup. By integrating amino protection, esterification, reduction, halogenation, and transition metal-mediated coupling, this process offers a robust pathway to the key intermediate (3R)-N-tert-butoxycarbonyl-3-amino-4-(2,4,5-trifluorophenyl)butanoic acid. The structural integrity and therapeutic efficacy of the final API depend heavily on the precision of these synthetic steps, as illustrated by the molecular architecture of the target compound.
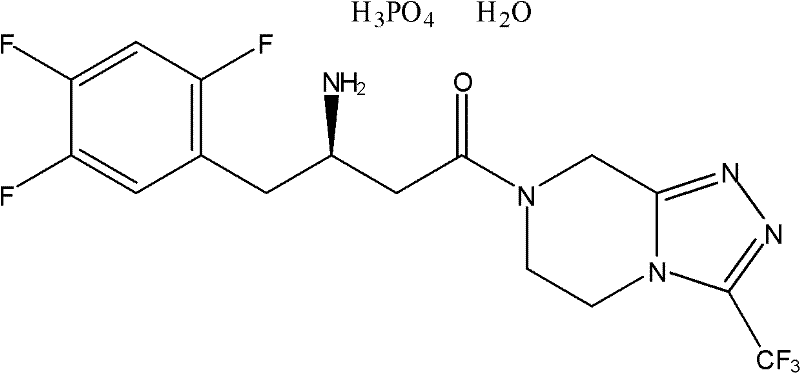
The strategic shift towards chiral pool synthesis represents a significant technological leap, providing a reliable API intermediate supplier with the tools to meet increasing market demand while maintaining stringent quality standards. This report analyzes the technical merits and commercial implications of this patented route, offering deep insights for R&D directors, procurement managers, and supply chain leaders seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sitagliptin has been plagued by processes that are either operationally cumbersome or economically prohibitive due to the reliance on precious metal catalysts. Early methodologies, such as those published by Kim et al., relied on inducing chirality through chiral auxiliaries followed by diazotization reactions to construct the beta-amino acid center. While scientifically valid, these routes are inherently limited to gram-scale research applications and lack the robustness required for industrial production. Furthermore, subsequent improvements by major pharmaceutical entities introduced asymmetric hydrogenation using chiral Rhodium or Ruthenium catalysts. Although these methods improved enantiomeric excess, they introduced a heavy dependency on expensive, scarce transition metals that drive up the cost of goods significantly. The operational complexity of handling air-sensitive catalysts and the rigorous purification required to remove trace metal residues further complicate the manufacturing workflow, creating bottlenecks in cost reduction in API manufacturing.
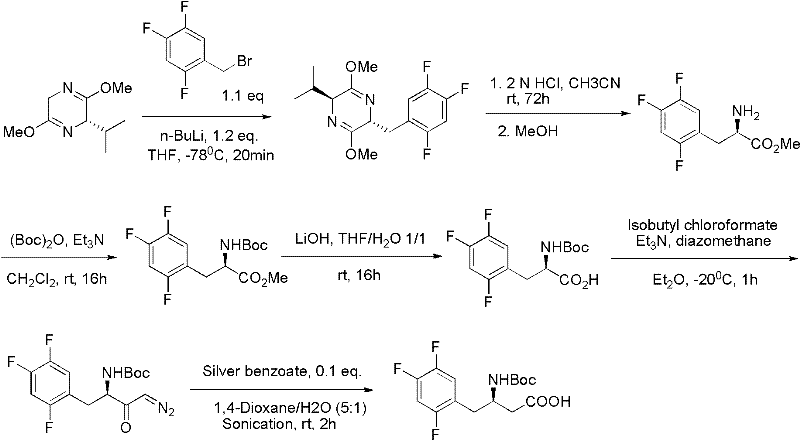
The Novel Approach
In stark contrast, the methodology outlined in CN102153559A revolutionizes the production landscape by adopting a chiral pool strategy starting from L-aspartic acid, a ubiquitous and inexpensive natural amino acid. This approach fundamentally alters the economic equation by eliminating the need for expensive chiral catalysts to establish the stereocenter, as the chirality is inherent in the starting material. The process flow is streamlined into distinct, manageable stages: amino protection, esterification, reduction, iodination, coupling, hydrolysis, amidation, and final salt formation. Each step is designed to proceed under mild reaction conditions, typically ranging from room temperature to moderate heating (30°C to 70°C), which drastically reduces energy consumption and safety risks associated with high-pressure hydrogenation. This novel route not only simplifies the operational protocol but also ensures high optical purity is maintained throughout the synthesis, directly addressing the critical quality attributes required for regulatory approval.
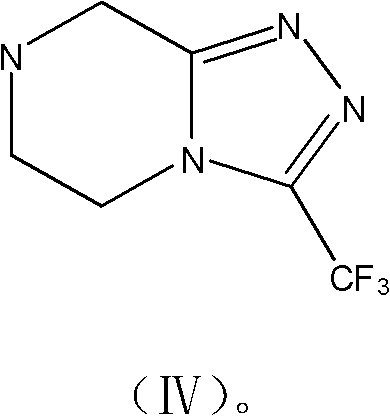
Mechanistic Insights into Zinc-Mediated Cross-Coupling
The cornerstone of this synthetic innovation lies in the transition metal-catalyzed coupling reaction that joins the chiral aliphatic chain with the electron-deficient aromatic ring. The patent details a versatile coupling mechanism where the iodinated intermediate, derived from L-aspartic acid, reacts with 2,4,5-trifluoroiodobenzene or bromobenzene. This transformation is facilitated by the in situ generation of organozinc or organomagnesium species, activated by catalysts such as bis(triphenylphosphine)palladium(II) chloride, palladium dichloride, or even copper-zinc couples. The use of zinc powder activated with 1,2-dibromoethane and trimethylchlorosilane in DMF creates a highly reactive nucleophile that efficiently displaces the halide on the aromatic ring. This mechanistic pathway is superior to traditional Grignard reactions in terms of functional group tolerance, allowing the protected amino ester to remain intact without racemization or side reactions. The careful control of reaction temperature, often maintained around 30°C during the addition of the aryl halide, prevents exothermic runaways and ensures high conversion rates.
Furthermore, the preservation of stereochemical integrity during this coupling phase is paramount for the biological activity of the final drug substance. The mechanism proceeds with retention or controlled inversion depending on the specific activation method, but the use of the chiral pool starting material ensures that the final product achieves the requisite (3R) configuration with high enantiomeric purity. Impurity control is managed through the selection of specific solvents like DMF or benzene mixtures and rigorous workup procedures involving aqueous washes and drying over anhydrous sodium sulfate. The subsequent hydrolysis and amidation steps utilize standard peptide coupling reagents like DCC (dicyclohexylcarbodiimide) to attach the triazolopyrazine moiety, completing the molecular assembly. This detailed understanding of the reaction mechanism allows process chemists to fine-tune parameters for maximum yield and minimal impurity formation, ensuring the production of high-purity pharmaceutical intermediates that meet global pharmacopeial standards.
How to Synthesize Sitagliptin Phosphate Efficiently
The execution of this synthesis requires precise adherence to the sequential chemical transformations described in the patent to ensure reproducibility and safety. The process begins with the protection of L-aspartic acid using di-tert-butyl dicarbonate (Boc anhydride) in a biphasic system, followed by esterification to activate the carboxylic acid for reduction. The reduction step utilizes sodium borohydride in THF to convert the ester to a primary alcohol, which is subsequently converted to an iodide using triphenylphosphine and iodine. The critical coupling step involves the activation of zinc or magnesium to facilitate the carbon-carbon bond formation with the trifluorophenyl ring. Following the coupling, the ester is hydrolyzed using lithium hydroxide, and the resulting acid is coupled with the amine component using DCC. Finally, the Boc protecting group is removed with trifluoroacetic acid, and the free base is converted to the phosphate salt. For the detailed standardized operating procedures and specific molar ratios, please refer to the comprehensive guide below.
- Protect the amino group of L-aspartic acid using Boc anhydride and perform esterification to form the protected ester intermediate.
- Reduce the ester to an alcohol and convert it to an iodide or bromide leaving group to prepare for coupling.
- Perform a transition metal-catalyzed coupling reaction with 2,4,5-trifluoroiodobenzene or bromobenzene using Zinc or Magnesium reagents.
- Hydrolyze the ester, amidate with the triazolopyrazine moiety, deprotect the Boc group, and form the phosphate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this L-aspartic acid-based route offers transformative advantages that extend beyond mere technical feasibility. The primary driver for adoption is the substantial cost optimization achieved by replacing scarce, high-value chiral catalysts with abundant, commodity-grade amino acids. This shift decouples the production cost from the volatile pricing of precious metals like Rhodium and Ruthenium, leading to a more stable and predictable cost structure. Additionally, the mild reaction conditions reduce the capital expenditure required for specialized high-pressure reactors, allowing existing multipurpose facilities to be utilized effectively. This flexibility enhances supply chain resilience, enabling manufacturers to respond swiftly to market fluctuations without the lead time penalties associated with sourcing specialized catalytic systems.
- Cost Reduction in Manufacturing: The elimination of expensive chiral hydrogenation catalysts represents a direct and significant reduction in raw material costs. By utilizing L-aspartic acid, which is produced on a massive industrial scale for the food and feed industries, the process leverages economies of scale that are unavailable to synthetic chiral catalysts. Furthermore, the avoidance of high-pressure hydrogenation equipment reduces both CAPEX and OPEX, as standard glass-lined or stainless steel reactors can be employed. The simplified purification steps, which do not require extensive chromatography to remove trace metal contaminants, further lower the processing costs, resulting in a highly competitive price point for the final API intermediate.
- Enhanced Supply Chain Reliability: Reliance on a chiral pool starting material like L-aspartic acid mitigates the risk of supply disruptions commonly associated with complex synthetic reagents. L-aspartic acid is a globally traded commodity with a robust and diversified supply base, ensuring continuous availability even during geopolitical or logistical disturbances. This stability is crucial for maintaining uninterrupted production schedules for life-saving diabetes medications. Moreover, the use of common solvents such as ethyl acetate, THF, and DMF simplifies logistics and inventory management, reducing the administrative burden on the supply chain team and minimizing the risk of production stoppages due to solvent shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily transferable from pilot plant to commercial production scales. The mild temperatures and atmospheric pressure operations minimize energy consumption and safety hazards, aligning with modern green chemistry principles. The waste profile is more manageable compared to heavy metal-catalyzed processes, as the primary byproducts are organic salts and spent zinc or magnesium species that can be treated using standard effluent protocols. This environmental compatibility facilitates easier regulatory approval and reduces the long-term liability associated with hazardous waste disposal, making it a sustainable choice for long-term manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on process capabilities and quality outcomes. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing frameworks.
Q: What is the primary advantage of using L-aspartic acid as a starting material?
A: Using L-aspartic acid leverages the chiral pool strategy, eliminating the need for expensive asymmetric hydrogenation catalysts like Rhodium or Ruthenium complexes, thereby significantly reducing raw material costs and simplifying optical purity control.
Q: How does this method improve supply chain reliability compared to prior art?
A: The method utilizes readily available commodity chemicals such as L-aspartic acid and avoids reliance on scarce precious metal catalysts, ensuring consistent raw material availability and reducing lead times for high-purity pharmaceutical intermediates.
Q: Is this synthesis route suitable for commercial scale-up?
A: Yes, the reaction conditions are mild (typically 30°C to 70°C) and utilize standard solvents like DMF, THF, and ethyl acetate, making the process highly adaptable for commercial scale-up of complex pharmaceutical intermediates without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Phosphate Supplier
The technical potential of this L-aspartic acid-derived synthesis route is immense, offering a clear pathway to high-quality, cost-effective Sitagliptin phosphate production. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels that exceed industry standards. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated process engineering team is ready to optimize this specific route to maximize yield and minimize impurities for your specific needs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can enhance your supply chain efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and reliability in your API sourcing strategy.
