Scalable Production of Halogenated Anthranilamide Intermediates for Advanced Agrochemicals
The chemical industry continuously seeks robust methodologies for synthesizing complex heterocyclic scaffolds, particularly those serving as critical backbones for next-generation crop protection agents. Patent CN101558056A presents a significant advancement in this domain by disclosing a highly efficient process for producing specific anthranilamide compounds or their salts. This intellectual property focuses on the selective halogenation of precursor compounds represented by Formula (II) to yield the target Formula (I) structures, which are renowned for their efficacy as pest control agents in agriculture and horticulture. The invention addresses the longstanding challenge of achieving high regioselectivity during the functionalization of the pyrazole and benzene rings, ensuring that the halogen atom is introduced precisely at the desired position without compromising the integrity of other sensitive functional groups. By optimizing reaction parameters such as temperature, solvent choice, and base equivalents, the disclosed method facilitates the production of high-purity intermediates essential for formulating potent agrochemical active ingredients.
Furthermore, the patent elucidates multiple synthetic pathways to access the necessary precursors, thereby providing flexibility in manufacturing strategies. Whether through oxidative cyclization or direct amide coupling, the described routes are designed to maximize atom economy and minimize waste generation. This comprehensive approach not only enhances the overall yield of the final anthranilamide derivative but also simplifies the purification process, which is a critical factor in reducing the cost of goods sold for large-scale production. For stakeholders in the agrochemical supply chain, understanding the nuances of this patented technology is vital for securing a reliable source of high-quality intermediates that meet stringent regulatory and performance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing halogenated anthranilamide scaffolds often suffer from poor regioselectivity and harsh reaction conditions that can degrade sensitive moieties within the molecule. Conventional halogenation techniques frequently rely on aggressive reagents or extreme temperatures, leading to the formation of multiple isomeric by-products that are difficult and costly to separate. This lack of specificity results in lower overall yields and necessitates extensive downstream processing, including multiple recrystallization steps or chromatographic purifications, which significantly inflate production costs and extend lead times. Moreover, older methods may utilize stoichiometric amounts of toxic heavy metal catalysts or generate substantial quantities of hazardous waste, posing environmental compliance challenges and increasing the burden on waste management infrastructure. The inability to consistently control the substitution pattern on the pyrazole ring can also compromise the biological activity of the final agrochemical product, rendering batches inconsistent and unreliable for commercial formulation.
The Novel Approach
In contrast, the methodology outlined in CN101558056A introduces a refined strategy centered on the selective halogenation of pre-formed anthranilamide precursors. As illustrated in the reaction scheme below, the transformation of Formula (II) to Formula (I) is achieved under controlled conditions that favor the introduction of the halogen atom at the specific R3 position. 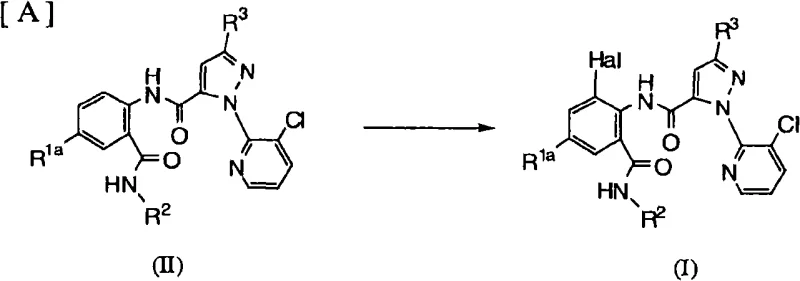 This novel approach utilizes mild halogenating agents such as elemental bromine or chlorine in the presence of suitable bases like sodium hydroxide or alkali metal alkoxides, typically within a temperature range of 0°C to 80°C. By avoiding extreme thermal stress and utilizing chemoselective reagents, the process minimizes side reactions and preserves the stereochemical integrity of chiral centers, such as the cyclopropylalkyl groups often present in these structures. The result is a cleaner reaction profile with fewer impurities, allowing for simpler isolation procedures and higher throughput. This strategic shift from non-selective to selective functionalization represents a paradigm change in manufacturing efficiency, directly addressing the pain points of yield loss and purity inconsistency associated with legacy synthesis routes.
This novel approach utilizes mild halogenating agents such as elemental bromine or chlorine in the presence of suitable bases like sodium hydroxide or alkali metal alkoxides, typically within a temperature range of 0°C to 80°C. By avoiding extreme thermal stress and utilizing chemoselective reagents, the process minimizes side reactions and preserves the stereochemical integrity of chiral centers, such as the cyclopropylalkyl groups often present in these structures. The result is a cleaner reaction profile with fewer impurities, allowing for simpler isolation procedures and higher throughput. This strategic shift from non-selective to selective functionalization represents a paradigm change in manufacturing efficiency, directly addressing the pain points of yield loss and purity inconsistency associated with legacy synthesis routes.
Mechanistic Insights into Selective Halogenation and Cyclization
The core mechanistic advantage of this patent lies in the precise control over electrophilic aromatic substitution and oxidative cyclization steps. In the primary transformation, the halogenating agent acts as an electrophile, attacking the electron-rich positions of the pyrazole or benzene ring. The presence of specific substituents and the electronic environment created by the amide linkage direct the incoming halogen to the desired site, ensuring high regioselectivity. The use of bases serves to deprotonate intermediate species or activate the substrate, facilitating the reaction kinetics without promoting unwanted decomposition. For instance, when converting dihydro-pyrazole intermediates to the fully aromatic pyrazole system, oxidizing agents such as 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) or potassium peroxodisulfate are employed. These oxidants effectively remove hydrogen atoms to establish aromaticity, a critical step for stabilizing the final molecular architecture and enhancing its biological potency.
Additionally, the patent details alternative pathways for constructing the central amide bond, which connects the anthranilic acid moiety to the heterocyclic acid component. One such route involves the coupling of a pyrazole carboxylic acid derivative (Formula IV) with an aminobenzamide (Formula V-1), as depicted in the following scheme. 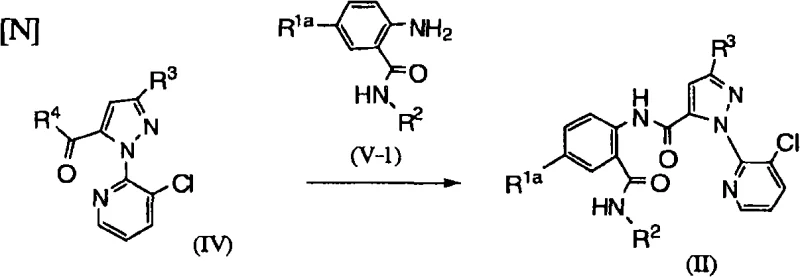 This condensation reaction is typically catalyzed by strong bases like potassium tert-butoxide or sodium hydride in polar aprotic solvents such as DMF or DMSO. The mechanism involves the deprotonation of the amine nucleophile, which then attacks the carbonyl carbon of the activated acid derivative. Careful control of water content is essential here, as moisture can hydrolyze the activated ester or acid chloride intermediates, reducing yield. The patent emphasizes the use of dehydrating agents like anhydrous sodium sulfate to maintain anhydrous conditions, ensuring that the nucleophilic attack proceeds efficiently to form the stable amide linkage. This mechanistic understanding allows process chemists to fine-tune reaction parameters for optimal scalability and reproducibility.
This condensation reaction is typically catalyzed by strong bases like potassium tert-butoxide or sodium hydride in polar aprotic solvents such as DMF or DMSO. The mechanism involves the deprotonation of the amine nucleophile, which then attacks the carbonyl carbon of the activated acid derivative. Careful control of water content is essential here, as moisture can hydrolyze the activated ester or acid chloride intermediates, reducing yield. The patent emphasizes the use of dehydrating agents like anhydrous sodium sulfate to maintain anhydrous conditions, ensuring that the nucleophilic attack proceeds efficiently to form the stable amide linkage. This mechanistic understanding allows process chemists to fine-tune reaction parameters for optimal scalability and reproducibility.
How to Synthesize Halogenated Anthranilamide Efficiently
To implement this synthesis effectively, one must adhere to strict protocols regarding reagent quality and reaction monitoring. The process begins with the preparation of the key intermediate, ensuring that all starting materials meet specified purity criteria to prevent the propagation of impurities. The halogenation step requires precise temperature control, often necessitating cooling baths to manage the exothermic nature of the reaction when adding halogens. Following the reaction, quenching procedures must be carefully executed to neutralize excess reagents safely before extraction. The detailed standardized synthesis steps见下方的指南。
- Prepare the precursor compound of Formula (II) via amide coupling or cyclization reactions.
- React Formula (II) with a halogenating agent such as bromine or chlorine in the presence of a base and solvent.
- Maintain reaction temperature between 0°C and 80°C to ensure selectivity and isolate the final Formula (I) product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the processes described in CN101558056A offers tangible strategic benefits beyond mere technical superiority. The streamlined nature of the synthesis reduces the number of unit operations required, which directly correlates to lower capital expenditure on equipment and reduced operational complexity. By minimizing the formation of difficult-to-remove impurities, the need for expensive chromatographic purification is eliminated, allowing for cost-effective crystallization-based isolation methods. This simplification of the downstream process significantly shortens the manufacturing cycle time, enabling faster response to market demand fluctuations and reducing inventory holding costs. Furthermore, the use of readily available and commodity-grade solvents and reagents mitigates supply risk, ensuring continuity of supply even during periods of raw material volatility.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reliance on stoichiometric halogenating agents drastically simplify the waste stream and reduce the cost associated with heavy metal removal and disposal. The high selectivity of the reaction means that less starting material is wasted on by-products, leading to substantial cost savings in raw material consumption. Additionally, the ability to operate at moderate temperatures reduces energy consumption for heating and cooling, further contributing to a lower overall cost of production. These efficiencies compound over large production volumes, making the process economically attractive for commercial scale-up.
- Enhanced Supply Chain Reliability: The synthetic routes utilize common chemical building blocks that are widely available from multiple global suppliers, reducing dependency on single-source vendors. The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with agrochemical formulators. By avoiding exotic or highly regulated reagents, the procurement team can navigate regulatory hurdles more easily and secure materials with shorter lead times. This reliability strengthens the supply chain against disruptions and ensures a steady flow of high-purity intermediates to downstream customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and conditions that are compatible with standard reactor setups found in multipurpose chemical plants. The reduced generation of hazardous waste aligns with increasingly stringent environmental regulations, lowering the compliance burden and potential liability. The ability to scale from pilot plant quantities to multi-ton production without significant process re-engineering provides a clear path for meeting growing market demand. This environmental and operational compatibility makes the technology a sustainable choice for long-term manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on feasibility and performance. Understanding these aspects helps stakeholders make informed decisions about integrating this synthesis route into their existing production portfolios. The responses cover key areas such as reagent selection, safety considerations, and quality control metrics.
Q: What is the key advantage of the halogenation method in CN101558056A?
A: The method allows for selective halogenation at specific positions on the benzene or pyrazole ring under mild conditions, improving purity and yield compared to non-selective methods.
Q: What types of halogenating agents are suitable for this process?
A: The patent specifies the use of elemental chlorine or bromine, often in the presence of bases like sodium hydroxide or potassium tert-butoxide.
Q: Is this process scalable for industrial production?
A: Yes, the reaction conditions (0-80°C) and use of common solvents like DMF or toluene suggest high feasibility for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anthranilamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value agrochemical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the methodologies described in CN101558056A can be seamlessly transferred to our manufacturing facilities. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of anthranilamide intermediate performs consistently in your final formulations. Our state-of-the-art infrastructure allows us to handle complex halogenation and coupling reactions with the utmost precision and safety.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your production efficiency and reduce your overall manufacturing costs. Let us be your partner in driving innovation and reliability in the agrochemical sector.
