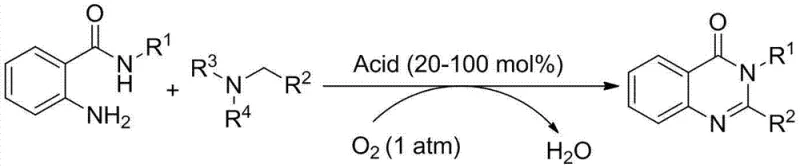
Advanced Metal-Free Synthesis of Quinazolinones for Commercial Pharmaceutical Production
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for constructing privileged scaffolds, and patent CN103408502A presents a transformative approach to synthesizing quinazolinone compounds. This specific intellectual property outlines a novel methodology that replaces traditional transition metal catalysts and toxic stoichiometric oxidants with a simple Bronsted acid catalytic system utilizing molecular oxygen. For R&D directors and process chemists, this represents a significant leap forward in green chemistry, offering a route that eliminates the persistent challenge of heavy metal contamination in active pharmaceutical ingredients. The core innovation lies in the ability to couple anthranilamide derivatives directly with various amines under an oxygen atmosphere, achieving high conversion rates without the need for complex ligand systems or inert gas protection beyond the initial oxygen fill. This technology not only simplifies the operational complexity of the synthesis but also aligns perfectly with modern regulatory demands for cleaner drug substances, making it a highly attractive candidate for reliable quinazolinone supplier partnerships focused on long-term sustainability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied heavily on methodologies that introduce significant safety and purity risks into the manufacturing supply chain. Traditional routes often necessitate the use of harsh, toxic oxidants such as DDQ, manganese dioxide, or potassium permanganate, which generate substantial hazardous waste streams and require rigorous disposal protocols. Furthermore, many contemporary catalytic methods depend on transition metals like palladium, copper, or rhodium, which are not only expensive but also pose a severe risk of residual contamination in the final drug product. For procurement managers, these metal residues translate into costly downstream purification steps, such as scavenger resin treatments or additional chromatography, which drastically inflate the cost of goods sold. Additionally, the sensitivity of these metal catalysts to air and moisture often mandates stringent anhydrous conditions and specialized equipment, complicating the commercial scale-up of complex heterocycles and increasing the lead time for high-purity intermediates needed for clinical trials.
The Novel Approach
In stark contrast, the methodology disclosed in CN103408502A circumvents these bottlenecks by employing inexpensive and readily available Bronsted acids, such as acetic acid, benzoic acid, or diphenylphosphoric acid, as the sole catalytic promoters. This shift from transition metal catalysis to organocatalysis fundamentally alters the economic and safety profile of the synthesis, removing the need for expensive metal precursors and the associated regulatory burden of proving low metal limits. The use of molecular oxygen as the terminal oxidant is particularly advantageous, as it is atom-economical, generates water as the only byproduct, and eliminates the need for storing and handling dangerous chemical oxidants. This novel approach ensures that the resulting quinazolinone products are free from catalyst residues, as confirmed by NMR analysis in the patent examples, thereby streamlining the quality control process. By operating under relatively mild thermal conditions between 100°C and 140°C, the process maintains high selectivity and yield while significantly reducing energy consumption compared to more forcing traditional conditions.
Mechanistic Insights into Bronsted Acid-Catalyzed Oxidative Cyclization
The mechanistic pathway proposed for this transformation involves a synergistic interaction between the Bronsted acid catalyst and the molecular oxygen oxidant to facilitate the oxidative cyclization of the anthranilamide substrate. The acid likely activates the carbonyl group of the anthranilamide or facilitates the dehydrogenation of the amine intermediate, promoting the nucleophilic attack required to close the heterocyclic ring. Molecular oxygen serves as the hydrogen acceptor, regenerating the aromatic system and driving the equilibrium toward the formation of the quinazolinone core. This mechanism avoids the formation of radical species typically associated with metal-catalyzed oxidations, which can lead to unpredictable side reactions and impurity profiles. Instead, the reaction proceeds through a controlled ionic or concerted pathway that preserves the integrity of sensitive functional groups on the amine substrate, such as halogens or methoxy groups, which are common in medicinal chemistry libraries. This high level of chemoselectivity is crucial for maintaining the structural fidelity of complex drug candidates during the synthesis of key intermediates.
From an impurity control perspective, the absence of transition metals eliminates an entire class of potential contaminants that are notoriously difficult to remove to parts-per-million levels. In conventional metal-catalyzed processes, trace metals can coordinate with the product or intermediates, leading to colored impurities or catalyzing decomposition during storage. The Bronsted acid system described here ensures that the crude reaction mixture contains primarily organic byproducts that are easily separated via standard aqueous workup and extraction techniques. The patent data indicates that simple washing with water or saturated salt solutions is sufficient to remove the acid catalyst, followed by organic extraction to isolate the product. This simplicity in workup translates directly to higher overall recovery rates and reduced solvent usage, contributing to a greener manufacturing footprint. The robustness of this mechanism across a wide range of substrates, including primary, secondary, and tertiary amines, further underscores its utility for generating diverse chemical libraries for drug discovery.
How to Synthesize Quinazolinone Compounds Efficiently
The practical implementation of this synthesis route is designed for ease of operation, requiring standard laboratory or plant equipment without the need for specialized high-pressure reactors or gloveboxes. The general procedure involves charging a reaction vessel with the anthranilamide starting material and the selected Bronsted acid catalyst, followed by evacuating the system and backfilling with oxygen to establish the necessary oxidative environment. Once the solvent and amine coupling partner are added, the mixture is sealed and heated to the specified temperature range, typically between 100°C and 140°C, for a duration of 10 to 40 hours depending on the specific substrate reactivity. The detailed standardized synthesis steps, including specific molar ratios and workup parameters for various derivatives, are outlined in the guide below to ensure reproducibility and optimal yield for your specific application.
- Charge a reaction vessel with anthranilamide compound and Bronsted acid catalyst (20-100 mol%), then evacuate and backfill with molecular oxygen.
- Add organic solvent and amine substrate, seal the vessel, and heat the mixture to 100-140°C for 10-40 hours to facilitate oxidative cyclization.
- Upon completion, cool to room temperature, wash with water or brine, extract with organic solvent, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis technology offers profound strategic advantages that extend beyond simple chemical efficiency. The elimination of transition metal catalysts removes a major cost driver from the bill of materials, as precious metals like palladium or rhodium are subject to volatile market pricing and supply constraints. Furthermore, the reliance on commodity chemicals such as acetic acid, benzoic acid, and molecular oxygen ensures a stable and secure supply chain, reducing the risk of production delays caused by the shortage of specialized reagents. The simplified purification process, which avoids expensive metal scavenging resins and complex chromatographic separations, significantly lowers the operational expenditure associated with manufacturing. This cost reduction in pharmaceutical intermediate manufacturing allows for more competitive pricing structures while maintaining high margins, making it an ideal solution for large-scale commercial production where every percentage point of yield and cost efficiency matters.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with inexpensive Bronsted acids results in a drastic reduction in raw material costs. Since the catalyst loading is moderate and the acids are commodity chemicals, the overall cost of goods is significantly optimized. Additionally, the removal of metal scavenging steps reduces the consumption of auxiliary materials and solvents, further enhancing the economic viability of the process for high-volume production runs.
- Enhanced Supply Chain Reliability: By utilizing widely available reagents like oxygen and common organic acids, the process mitigates the risk of supply chain disruptions often associated with specialized catalytic systems. The robustness of the reaction conditions allows for flexible sourcing of raw materials, ensuring continuous production capability even during market fluctuations. This reliability is critical for maintaining consistent delivery schedules for critical drug intermediates, thereby strengthening the partnership between the manufacturer and the end-user.
- Scalability and Environmental Compliance: The use of molecular oxygen as a green oxidant aligns with increasingly stringent environmental regulations regarding waste disposal and emissions. The process generates water as the primary byproduct, minimizing the environmental footprint and simplifying waste treatment protocols. This environmental compliance facilitates smoother regulatory approvals and supports the company's sustainability goals, while the mild reaction conditions ensure that the process can be safely scaled from pilot plant to multi-ton commercial production without significant engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Bronsted acid-catalyzed synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing manufacturing workflows and for assessing its potential impact on product quality and regulatory compliance.
Q: Does this synthesis method leave toxic metal residues in the final API?
A: No. Unlike conventional methods using transition metals like Pd or Cu, this patent utilizes Bronsted acids (e.g., phosphoric acid, benzoic acid) as catalysts. NMR analysis confirms zero metal residue, which is critical for regulatory compliance in oncology drug manufacturing.
Q: What is the substrate scope for the amine component in this reaction?
A: The method demonstrates broad compatibility with primary, secondary, and tertiary amines. Successful examples include triethylamine, benzylamine derivatives, and heteroaryl amines, allowing for diverse functionalization at the 2-position of the quinazolinone core.
Q: Is this process scalable for industrial production of quinazolinone intermediates?
A: Yes. The process uses molecular oxygen (1 atm) as a green oxidant and avoids expensive, air-sensitive metal catalysts. The simple workup procedure involving aqueous washing and extraction supports robust commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to meet the evolving demands of the global pharmaceutical market. Our team of expert process chemists has thoroughly evaluated the methodology described in CN103408502A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to translate this laboratory innovation into industrial reality. We are committed to delivering high-purity quinazolinone intermediates that meet stringent purity specifications, leveraging our rigorous QC labs to ensure that every batch is free from metal residues and other critical impurities. Our state-of-the-art facilities are equipped to handle the specific thermal and pressure requirements of this oxidative cyclization process, guaranteeing consistent quality and supply continuity for our partners.
We invite you to collaborate with us to explore the full potential of this metal-free synthesis route for your specific drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener methodology for your supply chain. We encourage you to contact us today to obtain specific COA data for our quinazolinone portfolio and to discuss route feasibility assessments tailored to your unique project timelines and budget constraints, ensuring a seamless transition from development to commercial supply.
