Scalable Catalyst-Free Production of High-Purity Xanthone for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with environmental sustainability. A significant breakthrough in this domain is documented in patent CN109651327B, filed in early 2021, which discloses a novel process for preparing xanthone under strictly catalyst-free conditions. This technology represents a paradigm shift from traditional oxidative methods that rely heavily on transition metals or corrosive acids. By utilizing molecular oxygen as the sole oxidant in a specific non-polar solvent environment, this method achieves the direct conversion of xanthene to xanthone with remarkable efficiency. The elimination of catalysts not only simplifies the reaction matrix but also fundamentally alters the downstream purification landscape, offering a cleaner profile that is highly attractive for the production of high-value pharmaceutical intermediates. This report analyzes the technical merits and commercial implications of this catalyst-free aerobic oxidation strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of xanthone derivatives has been plagued by significant operational and environmental challenges inherent to conventional methodologies. Traditional routes often depend on the use of phenyl salicylate rearrangement or etherification reactions involving toxic reagents like phosphorus oxychloride, which generate substantial hazardous waste streams and require complex neutralization procedures. Furthermore, catalytic oxidation methods, while an improvement, typically necessitate the use of expensive transition metal complexes or strong mineral acids such as nitric acid. These additives introduce severe complications in the form of metal residue contamination, requiring rigorous and costly purification steps to meet stringent pharmaceutical purity standards. The presence of such impurities can compromise the biological activity of the final API and poses regulatory hurdles during drug approval processes. Additionally, the disposal of spent catalysts and acidic byproducts creates a heavy burden on waste management systems, driving up the overall cost of manufacturing and increasing the environmental footprint of the production facility.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN109651327B introduces a streamlined, catalyst-free oxidation pathway that leverages the intrinsic reactivity of xanthene under controlled high-pressure oxygen conditions. This innovative approach dissolves the xanthene substrate in non-polar alkane solvents—specifically n-hexane, n-heptane, n-octane, or cyclohexane—and subjects the mixture to elevated temperatures ranging from 120°C to 180°C. The reaction is driven solely by the introduction of oxygen gas at pressures between 0.2 and 4 MPa, creating a clean reaction environment devoid of extraneous chemical promoters. 
This direct oxidative transformation bypasses the need for any additive, thereby preventing the formation of catalyst-derived impurities at the source. The result is a reaction mixture that is inherently cleaner, allowing for simpler isolation techniques and significantly higher product purity. The ability to recycle the alkane solvent further enhances the economic viability of this process, creating a closed-loop system that minimizes raw material consumption. By replacing hazardous reagents with benign oxygen and recyclable solvents, this method aligns perfectly with modern green chemistry principles while delivering a product quality that exceeds that of acid-catalyzed counterparts.
Mechanistic Insights into Catalyst-Free Aerobic Oxidation
The success of this catalyst-free protocol lies in the unique interaction between the non-polar solvent environment and the electronic structure of the xanthene molecule. In typical polar solvents, reaction molecules tend to undergo ionization, which can stabilize intermediates in ways that hinder direct oxidation or lead to side reactions. However, the use of non-polar alkanes inhibits this ionization, forcing the reaction to proceed through a radical mechanism. Under the high-temperature and high-pressure oxygen conditions, the benzene rings on the xanthene molecule play a crucial activation role, facilitating the generation of superoxide radicals directly from molecular oxygen. These highly reactive radical species are then capable of attacking the methylene bridge of the xanthene core, abstracting hydrogen atoms and initiating the oxidation sequence that ultimately yields the carbonyl group of the xanthone structure. 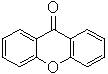
This mechanistic pathway effectively circumvents the high activation energy barrier that usually necessitates a catalyst. The absence of metal centers means there is no risk of metal leaching into the product, a critical factor for pharmaceutical applications where heavy metal limits are strictly enforced by agencies like the FDA and EMA. Furthermore, the radical nature of the reaction ensures a high degree of selectivity for the benzylic position, minimizing the formation of over-oxidized byproducts or ring-opened degradation products. The control of oxygen pressure is paramount here; maintaining the pressure between 0.5 and 3 MPa ensures a sufficient concentration of dissolved oxygen to sustain the radical chain reaction without leading to uncontrolled combustion or safety hazards. This precise tuning of physical parameters replaces the chemical tuning usually achieved by catalysts, demonstrating a sophisticated understanding of physical organic chemistry applied to industrial synthesis.
How to Synthesize Xanthone Efficiently
Implementing this synthesis route requires careful attention to reactor design and process parameters to ensure both safety and optimal yield. The procedure begins with the dissolution of the xanthene starting material in a selected non-polar solvent, with cyclohexane and n-octane showing particularly promising results in experimental trials. Once the solution is prepared, the reactor must be sealed and purged before introducing oxygen to reach the target pressure. The system is then heated to the specified range, typically optimized around 140°C to 160°C for the best balance of reaction rate and selectivity. Agitation is critical to maintain gas-liquid mass transfer, ensuring that the oxygen is continuously replenished at the reaction site. The detailed standardized synthesis steps, including specific molar ratios and work-up procedures, are outlined below.
- Dissolve xanthene starting material in a non-polar organic solvent such as cyclohexane, n-hexane, n-heptane, or n-octane within a high-pressure reactor.
- Introduce molecular oxygen into the reactor system until the internal pressure reaches a range of 0.2 to 4 MPa, ensuring sufficient oxidant concentration.
- Seal the reactor and heat the mixture to a temperature between 120°C and 180°C, maintaining agitation for 2 to 40 hours to complete the oxidation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalyst-free technology offers profound strategic advantages that extend beyond simple chemical efficiency. The primary value driver is the drastic simplification of the supply chain; by eliminating the need for specialized transition metal catalysts or corrosive acids, the procurement team removes a significant source of cost volatility and supply risk. Catalysts often involve precious metals with fluctuating market prices and complex logistics, whereas oxygen and alkanes are commodity chemicals available globally with stable pricing structures. This shift transforms the cost structure of xanthone manufacturing from a catalyst-dependent model to a utility-dependent model, providing greater predictability in long-term budgeting. Furthermore, the reduction in hazardous waste generation translates directly into lower disposal costs and reduced regulatory compliance burdens, enhancing the overall margin profile of the product.
- Cost Reduction in Manufacturing: The elimination of catalysts removes the entire unit operation associated with catalyst removal, such as filtration through silica or treatment with scavengers, which are both time-consuming and expensive. Without the need to purchase, handle, and dispose of toxic reagents like phosphorus oxychloride or nitric acid, the operational expenditure is significantly lowered. The ability to recycle the organic solvent multiple times without degradation further amplifies these savings, reducing the volume of fresh solvent required per kilogram of product. Consequently, the total cost of goods sold (COGS) is optimized through a combination of reduced raw material intensity and streamlined processing steps.
- Enhanced Supply Chain Reliability: Relying on abundant feedstocks like oxygen and hexane ensures a robust supply chain that is resilient to geopolitical disruptions or shortages of specialty chemicals. Traditional methods relying on specific proprietary catalysts can face bottlenecks if the supplier encounters production issues, potentially halting the entire manufacturing line. In contrast, the inputs for this novel process are foundational industrial chemicals with deep and redundant supply networks. This reliability allows for more aggressive production scheduling and shorter lead times, enabling the manufacturer to respond rapidly to fluctuations in market demand for xanthone derivatives.
- Scalability and Environmental Compliance: The simplicity of the reaction mixture facilitates easier scale-up from pilot plant to commercial tonnage production. The absence of exothermic catalyst initiation events reduces the thermal runaway risks often associated with large-scale oxidations, making the process safer to operate in large reactors. From an environmental perspective, the "green" nature of using oxygen as the oxidant and generating minimal waste aligns with increasingly strict global environmental regulations. This compliance future-proofs the manufacturing asset against tightening emission standards and avoids potential fines or shutdowns related to waste discharge, securing the long-term viability of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this catalyst-free xanthone synthesis. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on the feasibility and optimization of the process. Understanding these nuances is essential for R&D teams evaluating the technology for potential technology transfer or licensing.
Q: Why is a catalyst not required for this xanthene oxidation?
A: The process utilizes specific non-polar alkane solvents which inhibit ionization and promote the generation of superoxide radicals directly activated by the benzene rings, eliminating the need for external metallic or acidic catalysts.
Q: What are the optimal solvent conditions for maximum yield?
A: Experimental data indicates that cyclohexane and n-octane provide superior yields (up to 81%) compared to other alkanes, likely due to their specific solvation properties and stability under high-temperature oxidative conditions.
Q: How does this method improve supply chain reliability?
A: By removing the dependency on expensive transition metal catalysts or corrosive nitric acid, the supply chain is simplified to basic bulk commodities (oxygen and alkanes), reducing procurement risks and lead times.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Xanthone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of catalyst-free oxidation technologies in modernizing the production of key pharmaceutical intermediates like xanthone. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial realities. Our facilities are equipped with state-of-the-art high-pressure reactors and rigorous QC labs capable of meeting stringent purity specifications required by top-tier global pharmaceutical clients. We are committed to leveraging advanced process chemistry to deliver high-quality intermediates that accelerate your drug development timelines while maintaining the highest standards of safety and compliance.
We invite you to collaborate with us to explore how this efficient synthesis route can be integrated into your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in green chemistry can drive value for your organization.
