Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
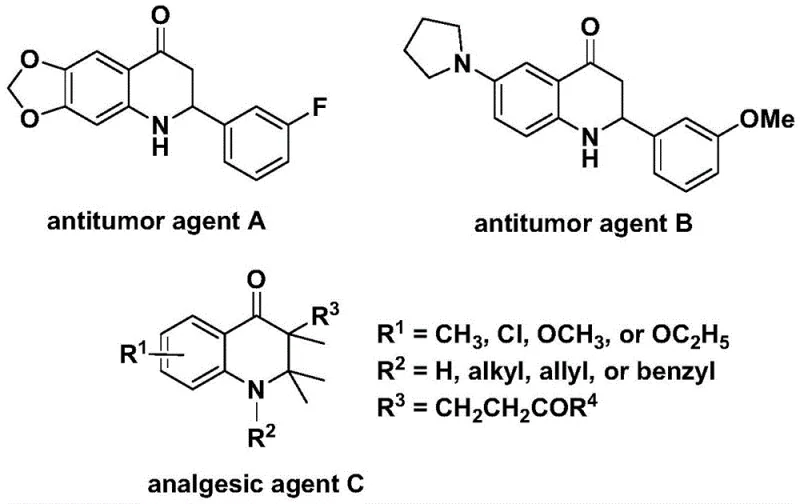
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activities. Patent CN112239456B discloses a robust preparation method for substituted 2,3-dihydroquinolone compounds, a class of molecules renowned for their presence in various bioactive agents including antitumor and analgesic drugs. As illustrated in the structural diversity of known bioactive molecules, the 2,3-dihydroquinolone skeleton serves as a critical pharmacophore in agents such as antitumor agent A, antitumor agent B, and analgesic agent C, highlighting the immense commercial value of accessing these cores efficiently.
This patented technology leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as primary starting materials. The innovation lies not only in the high reaction efficiency and good substrate compatibility but also in the operational simplicity that facilitates gram-level expansion and potential industrial application. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this methodology represents a significant advancement over traditional carbonylation techniques, offering a pathway to high-purity intermediates with reduced safety risks associated with gaseous carbon monoxide handling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles like 2,3-dihydroquinolones has often relied on direct carbonylation using carbon monoxide gas. While effective, this approach presents substantial logistical and safety challenges for large-scale manufacturing. The requirement for high-pressure equipment to handle toxic CO gas increases capital expenditure and imposes rigorous safety protocols that can slow down production timelines. Furthermore, conventional methods may suffer from limited substrate tolerance, where sensitive functional groups on the olefin or aniline precursors are incompatible with harsh reaction conditions, leading to complex impurity profiles that are difficult and costly to remove during downstream processing.
The Novel Approach
The novel approach detailed in the patent circumvents these issues by employing a solid carbon monoxide surrogate, specifically 1,3,5-trimesic acid phenol ester (TFBen). This strategic substitution allows the carbonylation to proceed under standard atmospheric pressure conditions without the need for specialized gas handling infrastructure. The reaction utilizes a palladium catalyst system, specifically bis(acetylacetone)palladium paired with a dppp ligand, to facilitate the coupling of N-pyridine sulfonyl-o-iodoaniline with various olefins. This method not only enhances safety but also improves the overall atom economy and ease of operation, making it an ideal candidate for cost reduction in API manufacturing where safety and scalability are paramount concerns.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The mechanistic pathway of this transformation involves a sophisticated catalytic cycle initiated by the oxidative addition of the palladium catalyst into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This step generates a reactive aryl-palladium intermediate, which is subsequently intercepted by carbon monoxide released in situ from the decomposition of the phenol ester surrogate. The insertion of CO forms an acyl-palladium species, a crucial intermediate that dictates the formation of the carbonyl functionality within the final heterocyclic ring. Following this, the olefin substrate coordinates with the palladium center and undergoes migratory insertion into the acyl-palladium bond, creating a new carbon-carbon bond and extending the molecular framework.
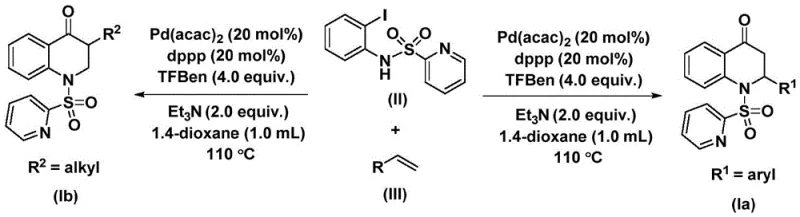
The final stage of the catalytic cycle involves an intramolecular reductive elimination that closes the six-membered ring, releasing the substituted 2,3-dihydroquinolone product and regenerating the active palladium catalyst for the next turnover. This mechanism ensures high chemoselectivity, as the palladium center specifically targets the iodine moiety and the olefin double bond, minimizing side reactions such as homocoupling or polymerization of the olefin. The ability to tune the electronic properties of the olefin, whether it be an aryl group for 2-aryl substituted products or an alkyl/silyl group for 3-alkyl substituted products, allows for precise control over the final molecular architecture, as demonstrated by the successful synthesis of compounds Ia and Ib.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The synthesis protocol outlined in the patent provides a straightforward procedure accessible to standard organic synthesis laboratories. The process begins with the careful weighing and mixing of the palladium catalyst, ligand, base, CO surrogate, and substrates in an aprotic organic solvent such as 1,4-dioxane. The reaction mixture is then heated to elevated temperatures, typically around 110°C, to drive the carbonylation and cyclization to completion over a period of 24 to 48 hours. Detailed standardized synthesis steps for implementing this protocol are provided in the guide below.
- Charge a reaction vessel with palladium bis(acetylacetonate), 1,3-bis(diphenylphosphino)propane, triethylamine, 1,3,5-trimesic acid phenol ester, N-pyridine sulfonyl-o-iodoaniline, and the desired olefin substrate in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to a temperature between 100°C and 120°C, preferably 110°C, and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the reaction mixture, mix the crude sample with silica gel, and purify the resulting substituted 2,3-dihydroquinolone compound via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits regarding cost structure and supply continuity. The shift from gaseous CO to a solid surrogate drastically simplifies the reactor setup, removing the need for expensive high-pressure autoclaves and specialized gas delivery systems. This reduction in equipment complexity translates directly to lower capital investment and maintenance costs, facilitating a more agile manufacturing environment capable of responding quickly to market demands for complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The utilization of commercially available starting materials, such as simple olefins and iodoanilines, combined with a recyclable palladium catalyst system, significantly lowers the raw material cost profile. By eliminating the hazards associated with toxic gas handling, the facility saves on safety compliance costs and insurance premiums, while the high conversion rates minimize waste generation and solvent usage, contributing to substantial cost savings in the overall production budget.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of functional groups including halogens, alkyls, and silyl groups, ensures consistent batch-to-batch quality. This reliability reduces the risk of production delays caused by failed batches or difficult purifications, thereby securing a steady flow of high-quality intermediates essential for maintaining uninterrupted API production schedules and meeting strict delivery deadlines.
- Scalability and Environmental Compliance: The method has been demonstrated to be scalable from milligram to gram levels with high efficiency, indicating strong potential for ton-scale commercial production. The use of a solid CO source reduces the environmental footprint by preventing the release of greenhouse gases, aligning with increasingly stringent global environmental regulations and supporting the company's sustainability goals through greener chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed carbonylation process. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, ensuring that potential partners have a clear understanding of the technology's capabilities and limitations before initiating collaboration.
Q: What is the advantage of using 1,3,5-trimesic acid phenol ester in this synthesis?
A: Using 1,3,5-trimesic acid phenol ester serves as a solid carbon monoxide surrogate, eliminating the need for handling toxic and hazardous CO gas directly, thereby significantly enhancing operational safety and simplifying equipment requirements for industrial scale-up.
Q: What types of olefin substrates are compatible with this palladium-catalyzed method?
A: The method demonstrates broad substrate compatibility, successfully accommodating both aryl-substituted olefins to form 2-aryl derivatives and alkyl or silyl-substituted olefins to form 3-alkyl derivatives, allowing for diverse functionalization of the dihydroquinolone core.
Q: What are the typical reaction conditions required for high yields?
A: Optimal results are achieved using Pd(acac)2 as the catalyst with dppp as the ligand in 1,4-dioxane solvent at 110°C for 48 hours, utilizing triethylamine as a base to facilitate the carbonylative cyclization process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity 2,3-dihydroquinolone intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to leverage our technical expertise to optimize your supply chain for these valuable heterocyclic building blocks. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals.
