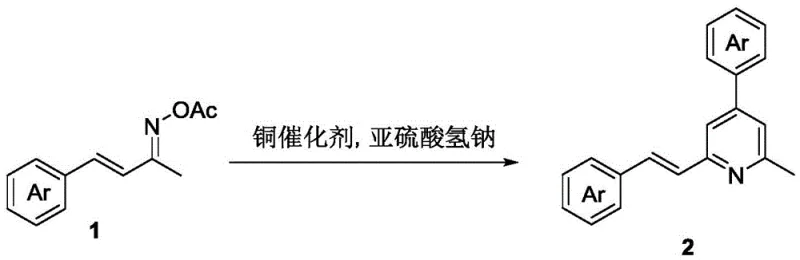
Advanced One-Step Synthesis of Substituted Pyridines for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct nitrogen-containing heterocycles, which serve as the backbone for countless active pharmaceutical ingredients and functional materials. Patent CN108047128B introduces a groundbreaking methodology for the synthesis of (E)-2-methyl-4-phenyl-6-styryl substituted pyridine compounds, addressing critical bottlenecks in traditional organic synthesis. This innovation leverages a copper-catalyzed radical cyclization strategy that transforms simple oxime ester precursors into complex polysubstituted pyridines in a single operational step. By utilizing readily available reagents such as sodium bisulfite and cuprous bromide, this technology offers a robust alternative to conventional multi-step sequences that often suffer from low atom economy and harsh reaction conditions. For R&D Directors and Procurement Managers alike, this patent represents a significant opportunity to streamline supply chains and reduce the cost of goods sold for high-value intermediates. The ability to construct the pyridine core with high regioselectivity and yield underpins the commercial viability of this approach for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of substituted pyridine derivatives has relied heavily on transition metal-catalyzed cross-coupling reactions, such as Suzuki or Heck couplings, which require pre-functionalized halide starting materials. These traditional pathways are often plagued by the necessity for expensive palladium catalysts and specialized phosphine ligands that drive up the overall production cost significantly. Furthermore, conventional methods frequently demand multi-step synthetic sequences to install the necessary substituents, leading to cumulative yield losses and increased waste generation at each stage. The requirement for stringent anhydrous conditions and the sensitivity of organometallic reagents to moisture and oxygen further complicate the operational protocol, making scale-up challenging and risky for process chemists. Additionally, the removal of trace heavy metal residues from the final product to meet pharmaceutical purity standards adds another layer of complexity and expense to the downstream processing. These cumulative inefficiencies create substantial barriers to entry for cost-sensitive applications in the generic drug and agrochemical sectors.
The Novel Approach
In stark contrast, the methodology disclosed in CN108047128B utilizes a direct, one-step cyclization of (2E,3E)-4-phenylbut-3-ene-2-one-O-acetyloxime compounds to forge the pyridine ring system. This novel approach bypasses the need for pre-halogenated substrates and expensive noble metal catalysts, relying instead on an abundant and inexpensive copper source. The reaction proceeds under relatively mild thermal conditions, typically between 80°C and 120°C, which reduces energy consumption and minimizes the thermal degradation of sensitive functional groups. By employing sodium bisulfite as a reducing additive, the system effectively manages the redox cycle of the copper catalyst, ensuring sustained catalytic activity throughout the reaction duration. This streamlined process not only shortens the production timeline but also significantly simplifies the work-up procedure, as there are fewer by-products and reagents to separate. The result is a highly efficient synthetic route that aligns perfectly with the principles of green chemistry and industrial practicality.
Mechanistic Insights into CuBr-Catalyzed Radical Cyclization
The core of this technological advancement lies in its unique radical mechanism, which diverges from standard ionic pathways found in classical heterocycle synthesis. Under the influence of the copper catalyst, the N-O bond of the oxime ester substrate undergoes homolytic cleavage to generate a nitrogen-centered radical species. This highly reactive intermediate then attacks the unsaturated carbon-carbon double bond of a second substrate molecule, initiating a cascade of cyclization events that construct the six-membered pyridine ring. The presence of sodium bisulfite is crucial as it facilitates the reduction of the copper species and assists in the final aromatization step through oxidation. Understanding this mechanistic nuance is vital for R&D teams aiming to optimize the reaction for specific substrates or to troubleshoot potential side reactions during scale-up. The radical nature of the transformation allows for a broader tolerance of functional groups compared to ionic mechanisms, thereby expanding the scope of accessible chemical space for drug discovery programs.
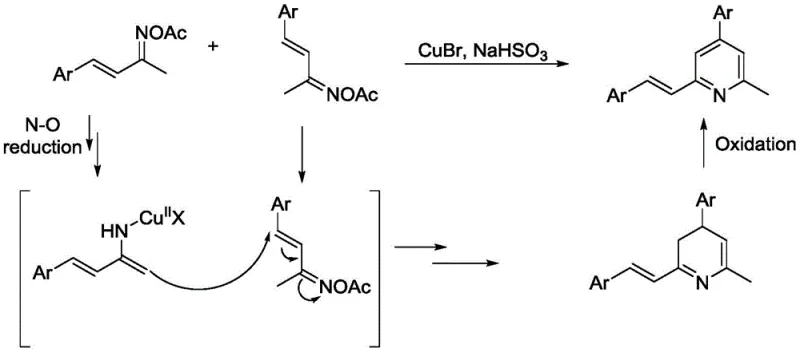
Impurity control is another critical aspect where this mechanism offers distinct advantages over traditional methods. Because the reaction is a self-coupling process of a single type of precursor, the potential for cross-coupling impurities between different starting materials is inherently eliminated. The primary by-products are typically derived from incomplete conversion or minor decomposition of the oxime ester, which are generally easier to separate than the complex metal-ligand complexes found in palladium-catalyzed reactions. The use of column chromatography for purification, as described in the patent examples, effectively removes residual copper salts and organic impurities to yield high-purity products. For quality control teams, this means a more predictable impurity profile and a simpler validation process for regulatory filings. The robustness of the radical pathway ensures consistent product quality across different batches, which is a key requirement for GMP manufacturing environments.
How to Synthesize (E)-2-methyl-4-phenyl-6-styryl Substituted Pyridine Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and reproducibility. The standard protocol involves charging a reaction vessel with the oxime ester substrate, cuprous bromide catalyst, and sodium bisulfite in a solvent such as 1,4-dioxane under an inert nitrogen atmosphere. The mixture is then heated to a constant temperature of approximately 100°C and stirred for a period of 12 hours to ensure complete conversion. Following the reaction, the mixture is cooled to room temperature and extracted with ethyl acetate, followed by solvent removal under reduced pressure. The crude residue is then subjected to column chromatography to isolate the target pyridine compound with high purity. This straightforward procedure minimizes the need for specialized equipment, making it accessible for both laboratory research and pilot plant operations.
- Prepare the reaction mixture by adding (2E,3E)-4-phenylbut-3-en-2-one-O-acetyloxime, sodium bisulfite, and cuprous bromide to 1,4-dioxane under nitrogen.
- Heat the reaction mixture to 100°C and stir for 12 hours to facilitate the radical cyclization and N-O bond cleavage.
- Cool the mixture, extract with ethyl acetate, and purify the crude product via column chromatography to isolate the target pyridine derivative.

Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patent offers compelling advantages that directly address the pain points of procurement and supply chain management in the fine chemical sector. The shift from expensive palladium catalysts to inexpensive copper salts results in a drastic reduction in raw material costs, which can significantly improve the margin profile of the final product. Furthermore, the simplification of the synthetic route from multiple steps to a single step reduces labor costs, utility consumption, and reactor occupancy time, leading to overall operational efficiency. The use of common, commercially available solvents and reagents ensures that supply chain disruptions are minimized, as there is no reliance on exotic or single-source materials. This reliability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts and complex ligands removes a major cost driver from the bill of materials, allowing for substantial savings in direct production costs. The one-step nature of the reaction also reduces the consumption of solvents and energy associated with intermediate isolation and purification steps. By simplifying the process flow, manufacturers can achieve higher throughput with existing infrastructure, effectively lowering the fixed cost per unit of production. These cumulative savings can be passed on to customers or reinvested into further process optimization initiatives.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like cuprous bromide and sodium bisulfite ensures a stable and diversified supply base, reducing the risk of shortages that often plague specialized reagents. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, further enhancing supply chain resilience. This stability allows for more accurate forecasting and inventory management, enabling procurement teams to negotiate better terms with suppliers. Ultimately, a reliable supply of key intermediates supports the uninterrupted manufacturing of finished drug products.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of highly toxic reagents make this process easier to scale from laboratory to commercial production without significant engineering hurdles. The reduced waste generation and simpler work-up procedure contribute to a lower environmental footprint, aligning with increasingly stringent regulatory requirements for green manufacturing. Easier waste treatment and lower emissions translate to reduced compliance costs and a stronger sustainability profile for the manufacturing site. This scalability ensures that the technology can meet growing market demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyridine synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for potential adopters. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production lines or research pipelines. The insights provided here aim to clarify the operational benefits and technical capabilities of the copper-catalyzed radical cyclization process.
Q: What are the primary advantages of this copper-catalyzed method over traditional cross-coupling?
A: This method eliminates the need for expensive palladium catalysts and specialized phosphine ligands, utilizing cheap cuprous bromide instead. It simplifies the process from multi-step to one-step, significantly reducing operational complexity and raw material costs.
Q: How does the reaction handle substrate diversity for pharmaceutical applications?
A: The protocol demonstrates wide substrate adaptability, tolerating electron-withdrawing and electron-donating groups on the benzene ring, as well as heterocycles like thiophene and furan, ensuring versatility for diverse API intermediate synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction uses mild conditions (80-120°C) and common solvents like 1,4-dioxane or toluene. The absence of sensitive reagents and the high yield profile make it highly amenable to commercial scale-up and continuous manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis route for the production of high-value pyridine intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to market. Our state-of-the-art facilities are equipped to handle the specific requirements of radical chemistry, with rigorous QC labs dedicated to maintaining stringent purity specifications for every batch. We are committed to leveraging this innovative technology to deliver cost-effective and high-quality solutions for your pharmaceutical and agrochemical needs.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this advanced synthetic method. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments for your target molecules. Let us help you engineer a more efficient and profitable production strategy for your critical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
