Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Global Pharmaceutical Manufacturing
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Global Pharmaceutical Manufacturing
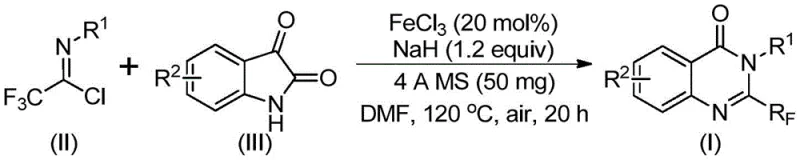
The pharmaceutical industry continuously seeks robust synthetic methodologies to access nitrogen-containing heterocycles, particularly quinazolinones, which serve as privileged scaffolds in medicinal chemistry. Patent CN111675662B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing critical bottlenecks in current manufacturing processes. This technology leverages an inexpensive iron catalyst to facilitate the cyclization of readily available starting materials, specifically trifluoroethylimidoyl chloride and isatin derivatives. The introduction of the trifluoromethyl group is strategically vital, as it significantly enhances the electronegativity, metabolic stability, and lipophilicity of drug candidates, thereby improving their bioavailability and therapeutic efficacy. By shifting away from precious metal catalysis and harsh fluorinating agents, this invention provides a sustainable and economically viable pathway for producing high-value pharmaceutical intermediates.
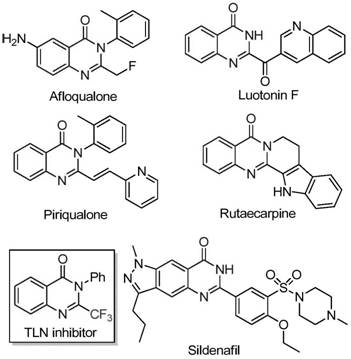
Quinazolinone compounds are ubiquitous in nature and pharmacology, exhibiting a broad spectrum of biological activities including anti-cancer, anticonvulsant, anti-inflammatory, antifungal, and antimalarial properties. The structural diversity shown in known bioactive molecules underscores the demand for efficient synthetic routes that can introduce functional groups like trifluoromethyl moieties with high precision. Traditional methods often rely on the cyclization of synthons such as anthranilamide or isatoic anhydride with trifluoroacetic anhydride or ethyl trifluoroacetate. However, these conventional approaches are frequently plagued by severe reaction conditions, limited substrate scope, and the use of expensive or hazardous reagents. The novel method described in the patent overcomes these limitations by utilizing a tandem reaction sequence promoted by sodium hydride and catalyzed by ferric chloride, enabling the direct construction of the quinazolinone core with excellent yields and functional group compatibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for trifluoromethylated quinazolinones have relied heavily on the use of strong electrophilic trifluoromethylating agents or pre-functionalized building blocks that are costly and difficult to handle on a large scale. For instance, the use of trifluoroacetic anhydride often requires strictly anhydrous conditions and generates stoichiometric amounts of acidic waste, complicating waste management and increasing environmental compliance costs. Furthermore, many existing protocols utilize precious metal catalysts such as palladium or copper, which not only drive up raw material costs but also necessitate rigorous purification steps to remove trace metal residues to parts-per-million levels, a mandatory requirement for API manufacturing. These factors collectively contribute to prolonged lead times and reduced overall process efficiency, making the supply chain for these critical intermediates vulnerable to fluctuations in metal prices and regulatory scrutiny regarding heavy metal impurities.
The Novel Approach
The methodology outlined in CN111675662B represents a paradigm shift by employing earth-abundant iron (FeCl3) as the catalyst and simple isatin derivatives as the cyclization precursors. This approach utilizes trifluoroethylimidoyl chloride, which can be easily synthesized from aromatic amines, triphenylphosphine, and trifluoroacetic acid, ensuring a stable and cost-effective supply of starting materials. The reaction proceeds through an initial alkali-promoted formation of a carbon-nitrogen bond, followed by an iron-catalyzed decarbonylation and cyclization cascade. This dual-activation strategy allows the reaction to proceed under relatively mild thermal conditions (initially at 40°C, then heating to 120°C) and, crucially, under aerobic conditions (in air), eliminating the need for expensive inert gas setups. The result is a streamlined process that delivers high-purity 2-trifluoromethyl quinazolinones with yields often exceeding 90% for various substrates, demonstrating exceptional robustness and scalability.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between base promotion and Lewis acid catalysis. Initially, sodium hydride acts as a strong base to deprotonate the isatin nitrogen, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step forms a transient trifluoroacetamidine intermediate, establishing the crucial carbon-nitrogen bond required for ring closure. Subsequently, the ferric chloride catalyst coordinates with the carbonyl oxygen and the imine nitrogen, facilitating the decarbonylation step which is the rate-determining phase of the reaction. The iron center stabilizes the transition state, lowering the activation energy required for the extrusion of carbon monoxide and the subsequent intramolecular cyclization to form the fused quinazolinone ring system. This mechanism explains the high tolerance for electron-withdrawing and electron-donating groups on the aromatic rings, as the iron catalyst effectively modulates the electronic density throughout the reaction coordinate.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based trifluoromethylation methods. Because the trifluoromethyl group is introduced via a stable imidoyl chloride precursor rather than a reactive radical source, the formation of side products resulting from non-selective radical addition is minimized. The use of 4A molecular sieves in the reaction mixture further ensures the removal of trace moisture, which could otherwise hydrolyze the imidoyl chloride or deactivate the sodium hydride, leading to incomplete conversion. The specificity of the iron-catalyzed decarbonylation ensures that the final product profile is clean, with the primary impurities being unreacted starting materials that are easily removed during the standard silica gel column chromatography purification step described in the patent examples.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The synthesis protocol detailed in the patent provides a reproducible framework for laboratory and pilot-scale production. The process begins with the precise weighing of ferric chloride (20 mol%) and sodium hydride (1.2 equivalents), which are added to a reaction vessel along with 4A molecular sieves to maintain anhydrous conditions. The substrates, trifluoroethylimidoyl chloride and the chosen isatin derivative, are dissolved in an aprotic polar solvent, with DMF identified as the optimal medium for maximizing conversion rates. The reaction mixture is stirred at a moderate temperature of 40°C for approximately 10 hours to allow the initial coupling to occur, after which the temperature is raised to 120°C for an additional 18 to 20 hours to drive the cyclization to completion. Detailed standardized synthesis steps follow below.
- Mix ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in anhydrous DMF solvent.
- Stir the reaction mixture at 40°C for approximately 10 hours to initiate the alkali-promoted carbon-nitrogen bond formation.
- Heat the reaction to 120°C and maintain for 18-20 hours under air to complete the iron-catalyzed decarbonylation and cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed technology translates into tangible strategic benefits regarding cost stability and operational flexibility. The substitution of expensive precious metals with commodity-grade iron chloride drastically reduces the raw material cost baseline, insulating the production budget from the volatile pricing trends associated with palladium and rhodium markets. Furthermore, the ability to run the reaction under air rather than inert atmosphere simplifies the engineering requirements for reactor vessels, allowing for the utilization of standard glass-lined or stainless steel equipment without the need for complex nitrogen blanketing systems. This operational simplicity directly correlates to reduced capital expenditure for new production lines and lower maintenance costs for existing facilities.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of costly catalysts and the simplification of downstream processing. By avoiding precious metals, manufacturers save significantly on both the initial purchase of the catalyst and the subsequent costs associated with metal scavenging resins and analytical testing for residual metals. Additionally, the high atom economy of the cyclization reaction and the use of commercially available isatin derivatives mean that the cost of goods sold (COGS) for the final API intermediate is substantially lower compared to legacy routes. The robustness of the reaction also minimizes batch failures, ensuring consistent output and reducing waste disposal costs associated with failed runs.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals such as isatin, aromatic amines, and iron salts ensures a resilient supply chain that is less susceptible to geopolitical disruptions or single-source supplier bottlenecks. Unlike specialized fluorinating reagents that may have long lead times or restricted availability, the starting materials for this process are produced globally in large volumes. This abundance allows for flexible sourcing strategies and the ability to scale production rapidly in response to market demand without waiting for specialized reagent shipments. The stability of the reagents also simplifies storage and logistics, as they do not require extreme cold chain management or hazardous material handling protocols beyond standard industrial practices.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with green chemistry principles by utilizing a non-toxic iron catalyst and generating minimal hazardous waste. The absence of heavy metals simplifies the treatment of effluent streams, reducing the burden on wastewater treatment facilities and ensuring easier compliance with stringent environmental regulations. The process is inherently scalable, having been demonstrated to work efficiently from milligram to gram scales in the patent examples, suggesting a smooth trajectory for expansion to multi-kilogram and ton-scale commercial production. The use of DMF, while requiring careful handling, is a well-understood solvent in the industry with established recovery and recycling protocols, further enhancing the sustainability profile of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of the method for industrial partners seeking to optimize their quinazolinone supply chains.
Q: What are the key advantages of using FeCl3 over precious metal catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers significant cost reductions compared to traditional palladium or rhodium catalysts. As an earth-abundant base metal, iron eliminates the need for expensive heavy metal scavenging steps required to meet strict pharmaceutical residual metal limits, thereby simplifying downstream processing and reducing overall production costs.
Q: How does this method improve substrate tolerance compared to conventional trifluoroacetylation routes?
A: This protocol utilizes readily available isatin derivatives and trifluoroethylimidoyl chlorides, which exhibit excellent functional group tolerance. Unlike methods requiring harsh trifluoroacetic anhydride conditions, this iron-catalyzed approach accommodates various substituents such as halogens, alkyl, and methoxy groups on the aromatic rings without compromising yield or selectivity.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It operates under aerobic conditions (in air) rather than requiring inert gas protection, uses common solvents like DMF, and employs inexpensive reagents. The simple workup involving filtration and standard column chromatography makes it highly viable for commercial scale-up from gram to multi-kilogram levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists has thoroughly analyzed the FeCl3-catalyzed route described in CN111675662B and is fully prepared to leverage this technology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and risk-free. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone intermediate we deliver meets the highest international quality standards required by global regulatory bodies.
We invite you to engage with our technical procurement team to discuss how this cost-effective synthesis route can be integrated into your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. We encourage you to request specific COA data and route feasibility assessments for your target molecules, allowing us to demonstrate concretely how our manufacturing capabilities can reduce your lead time for high-purity pharmaceutical intermediates while optimizing your overall production budget.
