Scalable Base-Promoted Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Drug Discovery
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to construct fluorinated heterocycles, driven by the unique metabolic stability and lipophilicity that trifluoromethyl groups impart to bioactive molecules. Patent CN113121462A introduces a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing critical bottlenecks in current synthetic workflows. This technology leverages a metal-free, base-promoted cyclization strategy that bypasses the need for hazardous azides and expensive transition metal catalysts. By utilizing readily available trifluoroacetimidoyl chlorides and diazo compounds, the process offers a streamlined pathway to high-value scaffolds essential for modern drug discovery. For R&D teams and procurement specialists alike, this represents a significant shift towards safer, more cost-efficient manufacturing of complex nitrogen-containing heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
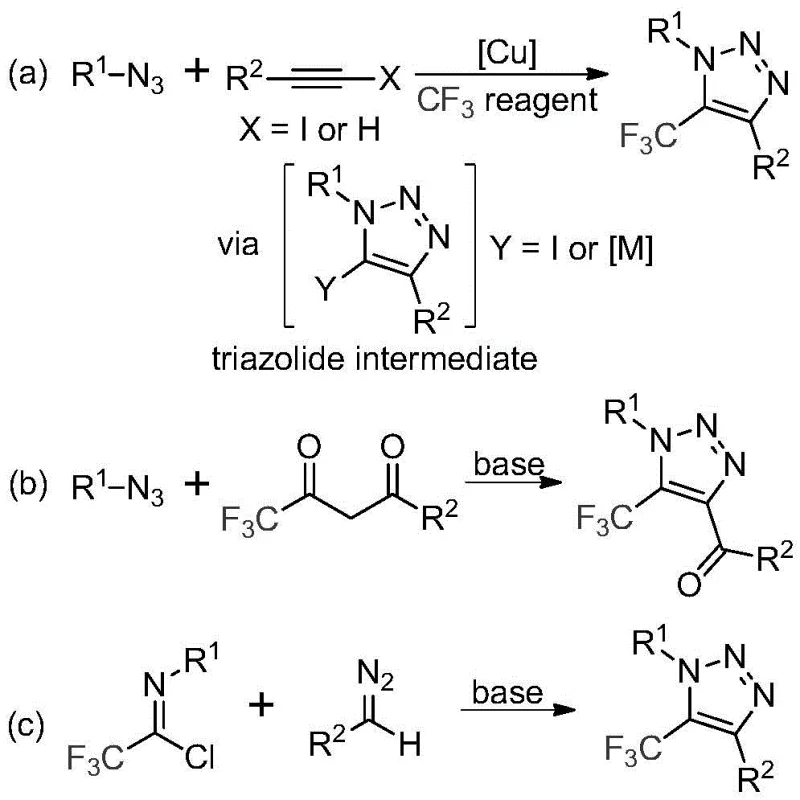
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed azide-alkyne cycloaddition (CuAAC) reactions or 1,3-dipolar cycloadditions involving organic azides. As illustrated in the literature precedents, these traditional routes often necessitate the use of highly toxic and potentially explosive organic azides, posing severe safety risks during scale-up. Furthermore, the requirement for transition metal catalysts, typically copper, introduces significant downstream processing challenges, including the need for rigorous metal scavenging to meet stringent pharmaceutical purity standards. These legacy methods also frequently suffer from limited substrate scope and harsh reaction conditions, which can degrade sensitive functional groups and reduce overall process efficiency.
The Novel Approach
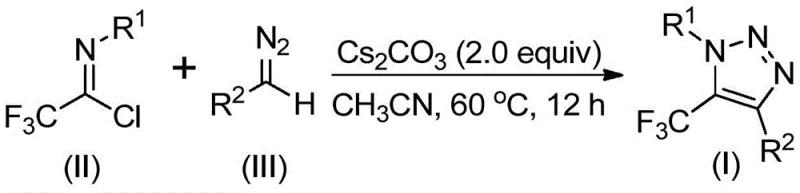
In stark contrast, the methodology disclosed in CN113121462A utilizes a benign, base-promoted reaction between trifluoroacetimidoyl chloride and diazo compounds. This innovative route completely eliminates the need for transition metals and explosive azides, fundamentally altering the safety profile of the synthesis. The reaction proceeds under mild thermal conditions (50-70°C) in common organic solvents like acetonitrile, utilizing inexpensive cesium carbonate as the promoter. This approach not only simplifies the operational procedure but also drastically reduces the environmental footprint by avoiding heavy metal waste. The versatility of this method allows for the facile introduction of diverse substituents at the N1 and C4 positions, enabling the rapid generation of chemical libraries for biological screening without the logistical burdens of handling hazardous reagents.
Mechanistic Insights into Base-Promoted Cyclization
The core of this transformation lies in a sophisticated intermolecular nucleophilic addition-elimination sequence followed by an intramolecular cyclization. Initially, the base activates the diazo compound, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroacetimidoyl chloride. This step forms a key intermediate through the displacement of the chloride leaving group. Subsequently, the system undergoes a 5-endo-dig cyclization, where the internal nitrogen nucleophile attacks the activated triple bond or equivalent electrophilic center, closing the triazole ring. This mechanism is distinct from radical pathways often seen in trifluoromethylation reactions, offering superior control over regioselectivity and minimizing the formation of unwanted byproducts.
From an impurity control perspective, the absence of transition metals eliminates the risk of metal-catalyzed side reactions such as homocoupling or oxidative degradation of sensitive substrates. The use of cesium carbonate, a mild inorganic base, ensures that acid-sensitive functional groups on the aromatic rings remain intact throughout the process. Furthermore, the reaction conditions are sufficiently robust to tolerate a wide range of electronic environments on the aryl rings, whether electron-donating or electron-withdrawing. This mechanistic clarity allows process chemists to predict outcomes with high confidence, facilitating the design of derivatives with specific physicochemical properties required for optimal bioavailability in final drug candidates.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The operational simplicity of this protocol makes it highly attractive for both laboratory discovery and pilot plant operations. The standard procedure involves mixing the trifluoroacetimidoyl chloride, the specific diazo compound, cesium carbonate, and activated molecular sieves in an anhydrous organic solvent. The molecular sieves play a crucial role in sequestering trace moisture, which could otherwise hydrolyze the reactive imidoyl chloride or decompose the diazo species. Following the reaction period, the workup is straightforward, typically involving filtration to remove inorganic salts and silica gel, followed by standard chromatographic purification. For detailed standardized synthesis steps and specific molar ratios optimized for different substrates, please refer to the guide below.
- Combine cesium carbonate, molecular sieves, trifluoroacetimidoyl chloride, and the specific diazo compound in an organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature range of 50-70°C and maintain stirring for 8 to 16 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical novelty. The shift away from precious metal catalysts and hazardous azides translates directly into reduced raw material costs and lower regulatory compliance burdens. By utilizing commodity chemicals like cesium carbonate and acetonitrile, manufacturers can insulate their supply chains from the volatility associated with specialized reagent markets. Moreover, the simplified purification process reduces solvent consumption and waste disposal costs, aligning with modern green chemistry initiatives that are increasingly mandated by global regulatory bodies.
- Cost Reduction in Manufacturing: The elimination of expensive copper catalysts and ligands removes a significant cost center from the bill of materials. Additionally, the avoidance of heavy metal scavengers and the associated validation testing for residual metals significantly lowers the cost of goods sold (COGS). The use of cheap, bulk-available bases like cesium carbonate further drives down operational expenses, making the production of high-purity 5-trifluoromethyl-1,2,3-triazoles economically viable even at large scales.
- Enhanced Supply Chain Reliability: The starting materials, specifically trifluoroacetimidoyl chlorides and diazo compounds, are derived from widely available precursors such as aromatic amines and acyl chlorides. This ensures a stable and continuous supply of feedstock, reducing the risk of production delays caused by specialty reagent shortages. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, enhancing overall manufacturing reliability and consistency.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be effective at the gram level with clear pathways for kilogram and tonne-scale expansion. The absence of toxic azides simplifies safety protocols and facility requirements, allowing for production in standard multipurpose reactors without the need for specialized blast-proof infrastructure. This ease of scale-up, combined with reduced hazardous waste generation, positions this method as a sustainable choice for long-term commercial manufacturing of complex heterocyclic scaffolds.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing a realistic overview of the method's capabilities and limitations for potential adopters.
Q: Does this synthesis method require toxic organic azides?
A: No, unlike conventional copper-catalyzed methods, this novel protocol utilizes stable diazo compounds and trifluoroacetimidoyl chlorides, completely eliminating the safety hazards associated with handling explosive organic azides.
Q: What is the substrate scope for R1 and R2 groups?
A: The method demonstrates broad tolerance, accommodating various aryl groups including phenyl, substituted phenyl (methyl, methoxy, halogen), and phenethyl for R1, while R2 can be aroyl, alkoxycarbonyl, phospholipid, or trifluoromethyl groups.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is easily expandable to gram-level reactions and beyond, utilizing cheap, commercially available starting materials and simple post-processing techniques suitable for manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in accelerating the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-trifluoromethyl-1,2,3-triazole meets the exacting standards required by the global pharmaceutical industry.
We invite you to leverage our technical expertise to optimize your supply chain and reduce development timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your commercial goals.
