Revolutionizing Brivaracetam Production: A Scalable Asymmetric Synthesis Strategy for Global Supply Chains
Revolutionizing Brivaracetam Production: A Scalable Asymmetric Synthesis Strategy for Global Supply Chains
The global demand for third-generation antiepileptic drugs continues to surge, placing immense pressure on supply chains to deliver high-purity active pharmaceutical ingredients (APIs) efficiently. Brivaracetam, a structural analogue of levetiracetam with superior binding affinity to the SV2A protein, represents a critical therapeutic asset. However, its complex stereochemistry, featuring two chiral centers at the 2S and 4R positions, has historically posed significant manufacturing challenges. A groundbreaking patent, CN115340482A, filed in late 2022, introduces a transformative synthesis method that fundamentally alters the production landscape. This innovative approach skillfully utilizes 5-hydroxy-4-propylfuran-2-one reacting with chiral sulfinamide derivatives to construct the molecular framework with precise stereocontrol. By bypassing the traditional bottlenecks of chiral chromatographic columns and cumbersome chemical resolution operations, this technology offers a streamlined pathway to high-purity Brivaracetam. For R&D directors and procurement strategists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing paradigms.
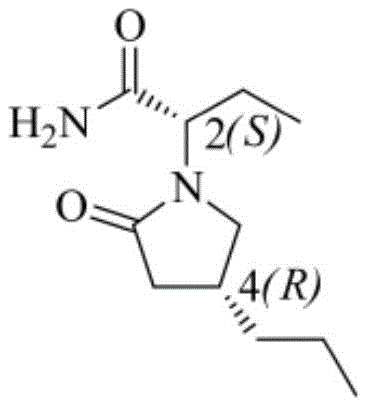
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Brivaracetam has been plagued by inefficiencies inherent in resolution-based strategies. The earliest reported routes involved synthesizing a racemic mixture at the 4-position followed by separation using chiral column chromatography. While chemically straightforward, this method is economically prohibitive for large-scale production due to the massive consumption of solvents and the limited loading capacity of chiral stationary phases. Furthermore, alternative chemical resolution routes require the introduction of resolving agents, such as phenylethylamine, which must later be removed, adding unnecessary unit operations and generating substantial waste. Perhaps most concerning are the asymmetric synthesis routes relying on (R)-epichlorohydrin. Although effective, epichlorohydrin is a known carcinogen and potent irritant with strict exposure limits, creating severe occupational health and safety liabilities for manufacturing facilities. Other reported methods utilizing sodium azide or biological enzymes introduce risks of explosion or biological contamination, respectively, rendering them unsuitable for robust, GMP-compliant commercial scale-up.
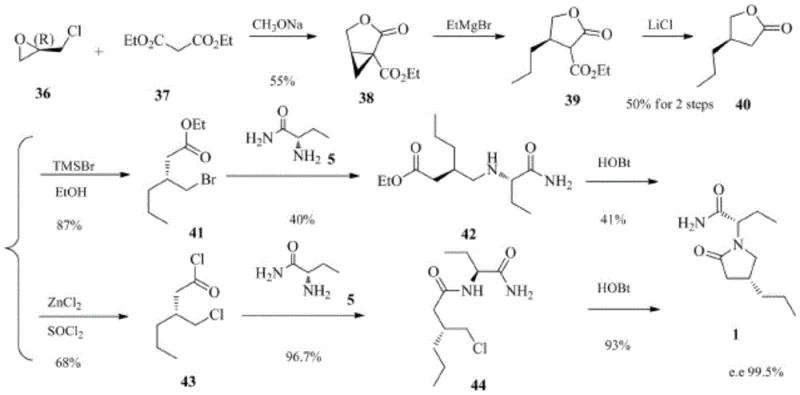
The Novel Approach
The methodology disclosed in patent CN115340482A dismantles these barriers by employing a chiral pool strategy centered on sulfinamide chemistry. Instead of resolving a racemate or using hazardous starting materials, this route builds the chiral lactam ring directly with the desired stereochemistry. The process begins with the condensation of a furanone derivative with a chiral sulfinamide, establishing the stereocenter early in the sequence. This is followed by a selective reduction of the olefinic bond to lock in the 4R configuration, a step that is both highly selective and operationally simple. By avoiding the use of chiral columns and eliminating the need for stoichiometric resolving agents that result in 50% material loss, this novel approach drastically improves the overall yield and material throughput. The reliance on conventional, non-toxic reagents ensures that the process is not only safer for operators but also easier to validate for regulatory compliance, making it an ideal candidate for reliable API intermediate supplier partnerships.
Mechanistic Insights into Sulfinamide-Mediated Asymmetric Induction
The core brilliance of this synthesis lies in the mechanistic elegance of the chiral sulfinamide auxiliary. In the initial step, the 5-hydroxy-4-propylfuran-2-one reacts with the sulfinamide derivative under Lewis acid catalysis. The sulfur atom in the sulfinamide acts as a powerful stereochemical director, guiding the nucleophilic attack to occur from a specific face of the molecule. This interaction creates a transient intermediate with a defined three-dimensional structure, effectively encoding the chirality required for the final drug substance. Following this condensation, the olefinic bond within the heterocyclic ring undergoes catalytic hydrogenation. Crucially, the existing chiral environment created by the sulfinamide group influences the approach of hydrogen to the double bond, ensuring that the reduction proceeds with high diastereoselectivity to yield the 4R configuration exclusively. This cascade of stereochemical events eliminates the formation of unwanted diastereomers that would otherwise require difficult separation downstream.
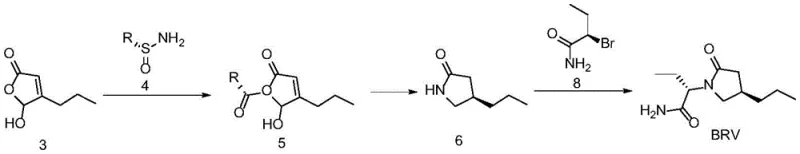
Following the establishment of the chiral lactam core, the synthesis proceeds through a hydrolysis step to remove the sulfinamide auxiliary, revealing the free amine necessary for the final coupling. The final stage involves an N-alkylation reaction with (R)-2-bromobutyramide. This step is facilitated by phase transfer catalysts in a biphasic system, which enhances the reaction rate and ensures complete conversion. From an impurity control perspective, this route is exceptionally clean. Because the chirality is established via covalent bonding with a high-purity auxiliary rather than physical separation, the risk of enantiomeric contamination is minimized. The selective nature of the hydrogenation and the specificity of the alkylation mean that the impurity profile is dominated by known, manageable byproducts rather than complex stereoisomers. This predictability is invaluable for quality control teams aiming to meet stringent pharmacopeial standards for high-purity pharmaceutical intermediates.
How to Synthesize Brivaracetam Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the asymmetric induction. The process is divided into three distinct operational stages: the condensation and reduction sequence to form the chiral lactam, the hydrolysis to unmask the amine, and the final alkylation. Each step utilizes standard chemical engineering unit operations, such as filtration, extraction, and distillation, ensuring compatibility with existing multipurpose reactor trains. The mild reaction temperatures, ranging from ambient to moderate reflux, further simplify the thermal management requirements. For detailed procedural specifics regarding molar ratios, solvent choices, and workup protocols, please refer to the standardized synthesis guide below.
- Condensation of 5-hydroxy-4-propylfuran-2-one with chiral sulfinamide derivatives using a Lewis acid and reducing agent to form the chiral intermediate.
- Selective catalytic hydrogenation of the olefinic bond followed by acid hydrolysis to generate the key lactam ring intermediate with 4R configuration.
- N-alkylation of the lactam intermediate with (R)-2-bromobutyramide under phase transfer catalysis to yield the final Brivaracetam API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers compelling economic and logistical benefits. The primary advantage stems from the elimination of chiral chromatography, a process notorious for its high operational expenditure due to the cost of chiral resins and the vast volumes of HPLC-grade solvents required. By replacing this with a catalytic asymmetric synthesis, the cost reduction in Brivaracetam manufacturing is substantial, driven by lower raw material consumption and reduced waste disposal fees. Furthermore, the avoidance of hazardous reagents like epichlorohydrin and sodium azide simplifies the regulatory burden associated with handling toxic substances, thereby reducing insurance premiums and safety compliance costs. The use of commercially available, commodity-grade starting materials ensures that the supply chain remains resilient against raw material shortages, enhancing supply chain reliability for long-term production contracts.
- Cost Reduction in Manufacturing: The economic model of this process is superior because it maximizes atom economy. Traditional resolution routes inherently discard half of the synthesized material (the unwanted enantiomer), effectively doubling the cost of goods for the starting materials. In contrast, this asymmetric route directs the synthesis towards the desired isomer from the outset, significantly reducing the raw material input per kilogram of final API. Additionally, the removal of the chiral column step eliminates a major bottleneck in production throughput, allowing for faster batch cycles and better utilization of capital equipment. The simplified workup procedures, which avoid complex purification steps, further contribute to lower utility and labor costs per unit produced.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialized, single-source reagents. This synthesis method mitigates that risk by utilizing common organic building blocks such as valeraldehyde and glyoxylic acid, which are produced globally in large volumes. The robustness of the chemical transformations, which tolerate minor variations in reaction conditions without compromising stereochemical integrity, ensures consistent output quality even when scaling up. This stability reduces the likelihood of batch failures and production delays, providing a dependable source of high-purity API intermediates for downstream formulation partners. The ability to source reagents from multiple vendors enhances negotiation leverage and protects against market volatility.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route aligns perfectly with modern green chemistry principles. The process operates under mild conditions, reducing energy consumption for heating and cooling. The solvents employed, such as isopropyl acetate and ethanol, are relatively benign and easily recoverable, minimizing the environmental footprint of the manufacturing site. By avoiding the generation of heavy metal waste or toxic azide byproducts, the facility reduces its liability and simplifies wastewater treatment processes. This environmental stewardship not only meets current regulatory standards but also future-proofs the manufacturing site against increasingly stringent global environmental regulations, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of adopting this route for commercial production. Understanding these details is crucial for technical teams evaluating process transfer and for strategic planners assessing the long-term value of this manufacturing method.
Q: How does this new synthesis method improve upon traditional chiral column resolution?
A: Traditional methods rely on separating racemic mixtures using expensive chiral columns, which limits throughput and increases solvent waste. This novel asymmetric route constructs the chiral center directly during synthesis, eliminating the need for resolution steps and significantly improving atom economy and production scalability.
Q: What are the safety advantages of this route compared to epichlorohydrin-based synthesis?
A: Conventional asymmetric routes often utilize (R)-epichlorohydrin, a highly toxic and volatile compound posing significant occupational health risks. The disclosed method employs conventional, easily obtained reagents like valeraldehyde and glyoxylic acid derivatives, operating under mild conditions that drastically reduce potential safety hazards in industrial settings.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is specifically designed for industrial viability. It avoids complex operations like biological enzymatic steps or hazardous reagents like sodium azide. The use of standard catalysts (Pd/C) and common solvents, combined with mild reaction temperatures, ensures the process can be safely scaled from pilot plants to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brivaracetam Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex antiepileptic drugs depends on a partnership grounded in technical excellence and supply security. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering Brivaracetam and its key intermediates with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our infrastructure is designed to handle the specific requirements of asymmetric synthesis, including dedicated zones for chiral processing and advanced containment systems for safe chemical handling.
We invite you to collaborate with us to optimize your supply chain for Brivaracetam. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this novel synthesis route can enhance your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of this critical medication, ensuring that patients worldwide have uninterrupted access to life-changing therapy.
