Scalable Synthesis of Bioactive 2,3-Dihydroquinolones via Safe Palladium-Catalyzed Carbonylation
Scalable Synthesis of Bioactive 2,3-Dihydroquinolones via Safe Palladium-Catalyzed Carbonylation
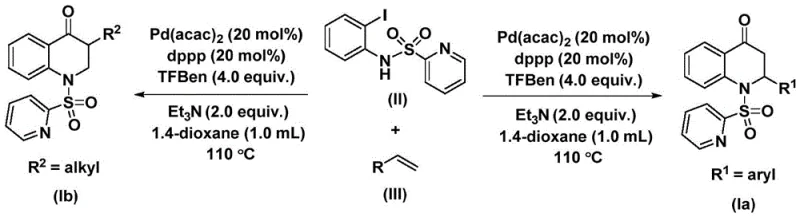
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN112239456B introduces a significant advancement in the preparation of substituted 2,3-dihydroquinolone compounds, a privileged scaffold found in numerous therapeutic agents. This technology leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as key starting materials. The innovation lies not only in the efficiency of the bond formation but also in the strategic use of a solid carbon monoxide surrogate, which circumvents the logistical and safety challenges associated with gaseous CO. For R&D directors and process chemists, this represents a viable pathway to access complex heterocyclic libraries with high functional group tolerance. The method operates under relatively mild thermal conditions and employs readily accessible reagents, positioning it as a strong candidate for the reliable pharmaceutical intermediate supplier market seeking to optimize their API manufacturing pipelines.
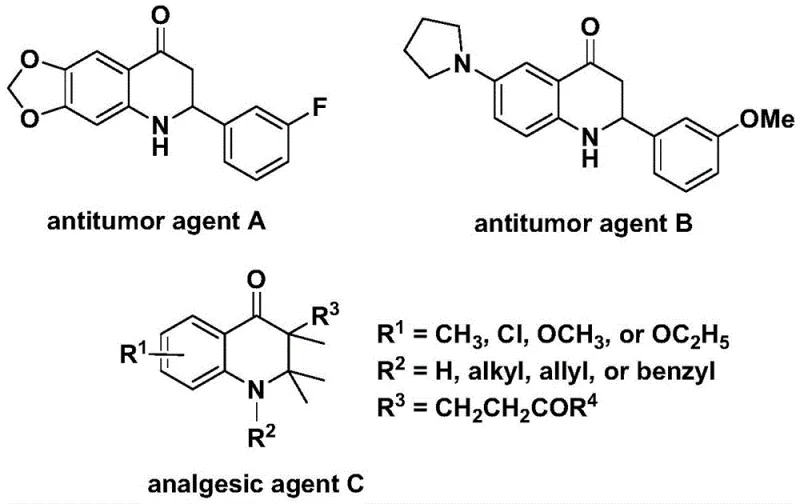
The biological significance of the 2,3-dihydroquinolone core cannot be overstated, serving as a critical structural motif in various medicinal chemistry programs. As illustrated in the provided data, derivatives of this scaffold exhibit potent anti-cancer activity against human cell lines, alongside significant analgesic properties. Traditional synthetic approaches to these skeletons often involve multi-step sequences or harsh reaction conditions that limit their utility in late-stage functionalization. The disclosed invention addresses these limitations by providing a direct carbonylative cyclization strategy. This approach allows for the rapid assembly of the quinolone ring system while simultaneously introducing diversity at the 2- and 3-positions through the selection of different olefin partners. Such versatility is crucial for medicinal chemists aiming to explore structure-activity relationships (SAR) efficiently without being constrained by cumbersome synthetic protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 2,3-dihydroquinolone rings has relied on methodologies that present substantial drawbacks for modern process chemistry. Many conventional routes require the use of gaseous carbon monoxide, a toxic and hazardous reagent that necessitates specialized high-pressure equipment and rigorous safety protocols, thereby increasing capital expenditure and operational complexity. Furthermore, traditional carbonylation reactions often suffer from poor atom economy or require stoichiometric amounts of expensive reagents, leading to elevated production costs and significant waste generation. The substrate scope in older methods is frequently narrow, failing to accommodate sensitive functional groups or sterically hindered olefins, which restricts the chemical space available for drug discovery. Additionally, the purification of products from these reactions can be challenging due to the formation of complex byproduct mixtures, resulting in lower overall yields and extended processing times that are detrimental to supply chain efficiency.
The Novel Approach
In contrast, the novel methodology described in the patent utilizes a palladium-catalyzed system that integrates a solid CO surrogate, specifically 1,3,5-trimesic acid phenol ester, to drive the carbonylation. This strategic substitution of gaseous CO with a stable solid source dramatically simplifies the reaction setup, allowing it to be performed in standard laboratory glassware or reactors without the need for high-pressure gas lines. The reaction proceeds efficiently at 110 °C in solvents like dioxane, demonstrating excellent compatibility with a wide range of olefins, including those bearing electron-withdrawing or electron-donating groups on aryl rings, as well as simple alkyl chains. This broad substrate tolerance enables the synthesis of both 2-aryl and 3-alkyl substituted variants, providing a versatile platform for generating diverse chemical libraries. The use of a catalytic amount of palladium combined with a bidentate phosphine ligand ensures high turnover numbers, making the process economically attractive for cost reduction in API manufacturing.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The mechanistic pathway of this transformation involves a sophisticated catalytic cycle initiated by the oxidative addition of the palladium catalyst into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This step generates a reactive aryl-palladium intermediate, which is pivotal for the subsequent insertion events. Following oxidative addition, the carbon monoxide released in situ from the decomposition of the phenol ester surrogate inserts into the palladium-carbon bond, forming an acyl-palladium species. This acyl intermediate then undergoes coordination and migratory insertion with the olefin substrate, establishing the new carbon-carbon bond that defines the 3-position of the quinolone ring. The cycle concludes with an intramolecular nucleophilic attack or reductive elimination step that closes the ring and regenerates the active palladium catalyst. Understanding this mechanism is vital for R&D teams, as it highlights the critical role of the ligand environment in stabilizing the various palladium intermediates and preventing catalyst deactivation pathways such as palladium black formation.
Impurity control in this system is inherently managed by the specificity of the palladium catalysis and the choice of the CO surrogate. Unlike gaseous CO methods where pressure fluctuations can lead to incomplete reactions or polymerization side products, the steady release of CO from the solid surrogate ensures a consistent concentration of the carbonyl source throughout the reaction duration. This controlled release minimizes the formation of decarbonylated byproducts or oligomeric species that often plague carbonylation reactions. Furthermore, the use of triethylamine as an additive helps to neutralize any acidic byproducts generated during the reaction, maintaining a stable pH environment that protects acid-sensitive functional groups on the substrate. The resulting crude reaction mixtures are typically cleaner, facilitating easier downstream purification via standard silica gel chromatography, which is essential for achieving the high-purity specifications required for pharmaceutical intermediates.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The synthesis protocol outlined in the patent provides a straightforward procedure for accessing these valuable heterocycles. The process begins by charging a reaction vessel with the palladium catalyst, ligand, base, CO surrogate, and the two primary coupling partners in an appropriate organic solvent. The mixture is then heated to facilitate the catalytic cycle, with reaction times typically ranging from 24 to 48 hours to ensure full conversion. Detailed standardized synthesis steps follow below for technical reference.
- Combine palladium bis(acetylacetonate), dppp ligand, triethylamine, and the CO surrogate (1,3,5-trimesic acid phenol ester) with N-pyridine sulfonyl-o-iodoaniline and the chosen olefin in an organic solvent like dioxane.
- Heat the reaction mixture to 110 °C and maintain stirring for 24 to 48 hours to ensure complete conversion of the starting materials into the cyclic product.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the high-purity substituted 2,3-dihydroquinolone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits regarding cost stability and operational safety. The elimination of gaseous carbon monoxide removes a major safety hazard from the manufacturing floor, reducing the need for specialized containment infrastructure and lowering insurance and compliance costs associated with handling toxic gases. Moreover, the reagents employed, such as the palladium catalyst and the olefin substrates, are generally commercially available commodities, ensuring a stable supply chain without reliance on exotic or custom-synthesized starting materials. This accessibility translates directly into reduced lead times for raw material sourcing and mitigates the risk of supply disruptions that can halt production lines. The robustness of the reaction conditions also implies a lower rate of batch failures, contributing to more predictable production schedules and reliable delivery timelines for downstream customers.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the use of a catalytic rather than stoichiometric amount of palladium, significantly lowering the consumption of this precious metal. Additionally, the replacement of high-pressure CO equipment with standard heating manifolds reduces capital expenditure on reactor hardware. The simplified workup procedure, which involves basic filtration and chromatography, minimizes solvent usage and labor hours compared to more complex multi-step syntheses. These factors collectively drive down the cost of goods sold (COGS), allowing for more competitive pricing strategies in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on shelf-stable solid reagents like the CO surrogate and o-iodoaniline derivatives ensures that inventory can be maintained without the degradation issues associated with gaseous reagents. This stability allows manufacturers to stockpile key inputs effectively, buffering against market volatility or logistical delays. The broad substrate scope means that a single manufacturing platform can be adapted to produce a variety of analogues simply by switching the olefin input, providing flexibility to respond rapidly to changing customer demands without retooling the entire production line.
- Scalability and Environmental Compliance: From an environmental perspective, the use of a solid CO surrogate aligns with green chemistry principles by reducing the risk of atmospheric emissions of toxic gases. The reaction generates fewer hazardous byproducts, simplifying waste treatment and disposal procedures. The method has been demonstrated to be scalable, with the potential to transition from gram-scale optimization to multi-kilogram commercial production without significant loss in efficiency. This scalability ensures that the supply chain can grow in tandem with the clinical or commercial success of the final drug product, supporting long-term partnership goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance for potential partners.
Q: What are the safety advantages of this carbonylation method compared to traditional CO gas methods?
A: This method utilizes 1,3,5-trimesic acid phenol ester as a solid carbon monoxide surrogate, eliminating the need for handling toxic and hazardous gaseous carbon monoxide, thereby significantly enhancing operational safety in industrial settings.
Q: What types of olefin substrates are compatible with this palladium-catalyzed system?
A: The process demonstrates excellent substrate compatibility, successfully accommodating both aryl-substituted olefins (such as styrenes with various electronic groups) and alkyl-substituted olefins, allowing for the synthesis of diverse 2-aryl and 3-alkyl substituted derivatives.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is designed for scalability, utilizing commercially available catalysts and reagents under relatively mild conditions (110 °C), making it highly practical for expanding from gram-level laboratory synthesis to multi-kilogram commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into industrial reality. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging technologies such as the palladium-catalyzed carbonylation described here, we can offer our clients a competitive edge through efficient, safe, and cost-effective manufacturing solutions tailored to their specific project needs.
We invite you to collaborate with us to explore the full potential of this synthetic route for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis for your target molecules. We are prepared to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions about your supply chain strategy. Let us be your partner in turning complex chemical challenges into commercial successes.
