Scalable Ruthenium-Catalyzed Synthesis of High-Purity 3-Acylpyrrole Pharmaceutical Intermediates
Scalable Ruthenium-Catalyzed Synthesis of High-Purity 3-Acylpyrrole Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and scalable pathways for constructing complex heterocyclic scaffolds, particularly those found in bioactive natural products and approved drugs. Patent CN113636968B introduces a groundbreaking synthetic methodology for 3-acylpyrrole compounds, utilizing a highly efficient ruthenium-catalyzed transfer hydrogenation cascade. This innovation addresses critical bottlenecks in the production of pyrrole-based active pharmaceutical ingredients (APIs) by replacing hazardous reagents with benign alcohols and employing earth-abundant transition metals. The significance of this technology lies in its ability to construct the pyrrole core with exceptional atom economy, directly impacting the cost structure and environmental footprint of large-scale manufacturing. For R&D directors and process chemists, this patent offers a viable alternative to traditional multi-step sequences that often suffer from low overall yields and difficult purification profiles. By leveraging this advanced catalytic system, manufacturers can achieve higher throughput while maintaining stringent quality standards required for global regulatory compliance.
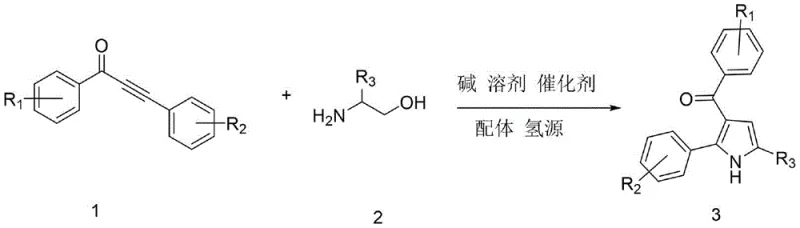
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-acylpyrrole derivatives has relied heavily on methodologies that involve harsh reaction conditions and expensive catalytic systems. Traditional approaches often utilize palladium catalysts coupled with toxic phosphine ligands, which not only drive up the raw material costs but also introduce significant challenges in removing trace metal residues from the final product. Furthermore, many classical routes require the use of unstable intermediates or hazardous oxidants, creating safety liabilities in an industrial setting. The reliance on external hydrogen gas sources for reduction steps adds another layer of complexity, necessitating specialized high-pressure equipment and rigorous safety protocols that can delay project timelines. Additionally, conventional methods frequently exhibit poor functional group tolerance, limiting the structural diversity accessible to medicinal chemists during lead optimization phases. These cumulative inefficiencies result in prolonged development cycles and inflated production costs, making it difficult to compete in the price-sensitive generic pharmaceutical market.
The Novel Approach
In stark contrast, the methodology disclosed in CN113636968B employs a sophisticated one-pot tandem reaction that streamlines the entire synthetic sequence into a single operational unit. By utilizing alpha-amino alcohols and alpha,beta-unsaturated alkynones as readily available starting materials, the process bypasses the need for pre-functionalized precursors that are often costly and difficult to source. The core of this innovation is the use of triruthenium dodecacarbonyl as a catalyst, which facilitates a borrowing hydrogen mechanism that internally recycles hydrogen atoms, thereby eliminating the need for external hydrogen gas cylinders. This transfer hydrogenation strategy not only enhances safety by operating under ambient pressure but also significantly improves the atom economy of the reaction. The use of ethanol as a hydrogen source and tert-amyl alcohol as a solvent further underscores the green chemistry principles embedded in this design, reducing the generation of hazardous waste streams. Consequently, this novel approach offers a streamlined, cost-effective, and environmentally friendly pathway that is ideally suited for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ru-Catalyzed Cyclization
The mechanistic elegance of this transformation lies in the synergistic interaction between the ruthenium catalyst and the 4-methyl-1,10-phenanthroline ligand, which orchestrates a precise sequence of dehydrogenation and cyclization events. Initially, the ruthenium center activates the alpha-amino alcohol through a dehydrogenation step, generating a reactive amino-ketone intermediate in situ while storing the hydrogen on the metal center. This transient species then undergoes a condensation reaction with the alpha,beta-unsaturated alkynone, forming an enamine intermediate that is poised for cyclization. The stored hydrogen is subsequently transferred back to the system to reduce the triple bond and aromatize the pyrrole ring, completing the catalytic cycle without the consumption of external reductants. This internal redox neutrality is a hallmark of borrowing hydrogen chemistry, ensuring that the reaction proceeds with minimal byproduct formation and maximum efficiency. Understanding this mechanism is crucial for process optimization, as it highlights the importance of maintaining strict anhydrous conditions and precise temperature control to prevent catalyst deactivation.
Furthermore, the robustness of this catalytic system is evidenced by its remarkable tolerance towards a wide array of substituents on the aromatic rings of both reactants. As demonstrated in the patent examples, electron-withdrawing groups such as fluorine and trifluoromethyl, as well as electron-donating groups like methyl, are well-tolerated without significant erosion of yield. 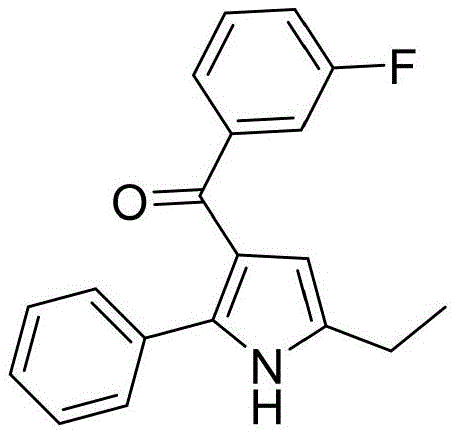
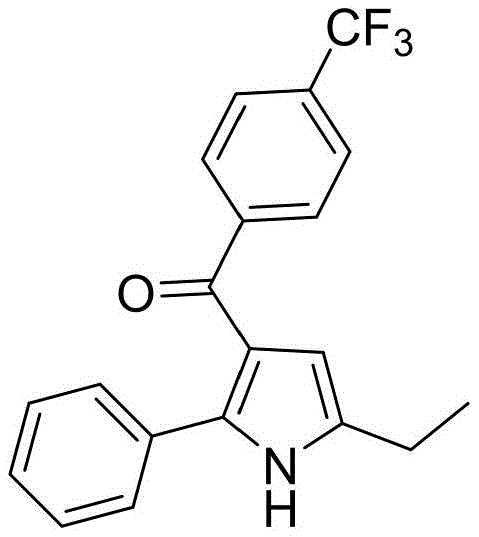 This broad substrate scope is particularly valuable for medicinal chemistry applications, where the introduction of specific halogen or alkyl groups is often required to modulate the pharmacokinetic properties of the final drug candidate. The ability to incorporate these diverse functional groups directly into the pyrrole scaffold simplifies the synthetic route and allows for rapid exploration of structure-activity relationships. Moreover, the high selectivity of the reaction minimizes the formation of regioisomers or polymeric byproducts, resulting in a cleaner crude reaction profile that simplifies downstream purification efforts. This level of control over the reaction outcome is essential for ensuring consistent product quality and meeting the rigorous specifications demanded by regulatory agencies.
This broad substrate scope is particularly valuable for medicinal chemistry applications, where the introduction of specific halogen or alkyl groups is often required to modulate the pharmacokinetic properties of the final drug candidate. The ability to incorporate these diverse functional groups directly into the pyrrole scaffold simplifies the synthetic route and allows for rapid exploration of structure-activity relationships. Moreover, the high selectivity of the reaction minimizes the formation of regioisomers or polymeric byproducts, resulting in a cleaner crude reaction profile that simplifies downstream purification efforts. This level of control over the reaction outcome is essential for ensuring consistent product quality and meeting the rigorous specifications demanded by regulatory agencies.
How to Synthesize 3-Acylpyrrole Efficiently
The practical implementation of this synthesis involves a straightforward protocol that can be easily adapted for both laboratory-scale discovery and pilot-plant production. The process begins with the charging of the reactor with the specific alpha-amino alcohol and alpha,beta-unsaturated alkynone substrates, along with the catalytic system comprising triruthenium dodecacarbonyl and the phenanthroline ligand. A base such as potassium carbonate is added to facilitate the initial deprotonation steps, while ethanol serves as the sacrificial hydrogen donor to drive the reduction phase. The reaction mixture is first heated to a moderate temperature to initiate the catalytic cycle, followed by a higher temperature phase to ensure complete conversion and aromatization of the pyrrole ring. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during operation.
- Combine alpha-amino alcohol, alpha,beta-unsaturated alkynone, triruthenium dodecacarbonyl catalyst, 4-methyl-1,10-phenanthroline ligand, ethanol, potassium carbonate, and tert-amyl alcohol solvent in a reactor.
- Stir the mixture at 80°C under nitrogen protection for 1.5 hours to initiate the reaction sequence.
- Increase the temperature to 150°C and maintain for 18 hours to complete the cyclization, followed by cooling, filtration, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this ruthenium-catalyzed methodology offers substantial strategic advantages that extend beyond simple yield improvements. The shift away from precious metal catalysts like palladium to more abundant ruthenium complexes significantly reduces the volatility associated with raw material pricing, providing greater stability in long-term cost forecasting. Additionally, the elimination of high-pressure hydrogenation equipment lowers the capital expenditure required for facility upgrades, allowing existing infrastructure to be utilized more effectively. The simplified one-pot nature of the reaction reduces the number of unit operations, which directly translates to lower labor costs and reduced energy consumption per kilogram of product produced. These operational efficiencies collectively contribute to a more resilient supply chain capable of responding rapidly to market demands without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the high atom economy and the use of inexpensive, commodity-grade solvents and reagents. By avoiding the need for stoichiometric amounts of expensive oxidants or reducing agents, the overall material cost per mole of product is drastically lowered compared to traditional oxidative cyclization methods. Furthermore, the high efficiency of the catalyst means that lower loadings can be used while still achieving excellent conversion rates, minimizing the cost associated with metal recovery and waste disposal. The reduction in processing steps also leads to significant savings in utility costs, as fewer heating and cooling cycles are required to complete the synthesis. These factors combine to create a highly competitive cost structure that enables manufacturers to offer high-purity intermediates at more attractive price points to their downstream customers.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as alpha-amino alcohols and alkynones ensures a stable and secure supply chain that is less susceptible to disruptions caused by geopolitical tensions or raw material shortages. Unlike specialized reagents that may have limited suppliers and long lead times, the key components of this reaction are produced by multiple chemical manufacturers globally, fostering a competitive sourcing environment. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the risk of batch failures and production delays. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major pharmaceutical clients who depend on consistent supply for their own manufacturing operations.
- Scalability and Environmental Compliance: Scaling this process from gram to ton quantities is facilitated by the inherent safety of the transfer hydrogenation mechanism, which avoids the hazards associated with handling large volumes of flammable hydrogen gas. The use of tert-amyl alcohol as a solvent provides a favorable balance between solubility and boiling point, allowing for efficient heat transfer and easy solvent recovery through distillation. From an environmental standpoint, the generation of waste is minimized due to the high selectivity of the reaction and the absence of toxic heavy metal residues that require complex remediation. This alignment with green chemistry principles not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturer, which is increasingly becoming a key criterion for supplier selection in the global pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route, based on the detailed experimental data provided in the patent documentation. These insights are designed to clarify the operational parameters and potential benefits for stakeholders evaluating this technology for integration into their existing portfolios. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: What is the primary advantage of this ruthenium-catalyzed method over traditional palladium methods?
A: This method utilizes a transfer hydrogenation strategy that eliminates the need for external hydrogen gas and avoids the use of expensive palladium catalysts, significantly reducing raw material costs and safety risks associated with high-pressure hydrogenation.
Q: Does this synthesis tolerate diverse functional groups on the aromatic rings?
A: Yes, the protocol demonstrates excellent functional group compatibility, successfully accommodating electron-withdrawing groups like trifluoromethyl and halogens, as well as electron-donating groups like methyl, without compromising yield or purity.
Q: What represents the typical purification workflow for these intermediates?
A: The process involves a straightforward workup where the reaction mixture is cooled, diluted, filtered, and concentrated, followed by standard silica gel column chromatography using a petroleum ether and ethyl acetate gradient to isolate the high-purity target product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acylpyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this ruthenium-catalyzed synthesis in advancing the production of next-generation pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity 3-acylpyrrole compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch released conforms to the highest international standards, providing our partners with the confidence they need to accelerate their drug development programs.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this optimized route can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of these critical building blocks for your future success.
