Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Intermediates for Pharma
Introduction to Patent CN115353511A Technology
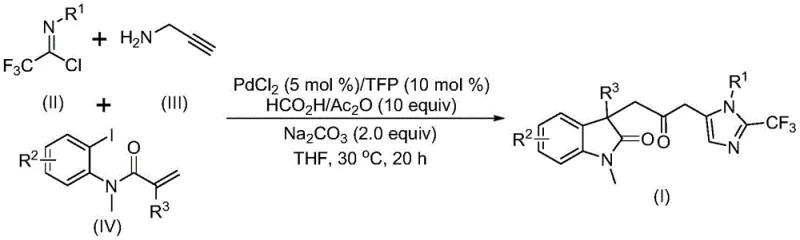
The pharmaceutical industry continuously seeks robust synthetic routes for complex heterocyclic scaffolds, particularly those incorporating metabolically stable motifs like trifluoromethyl groups. Patent CN115353511A discloses a groundbreaking multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole cores. This technology represents a significant leap forward in organic synthesis by enabling the construction of these privileged structures through a one-pot palladium-catalyzed cascade reaction. The process elegantly bypasses the need for hazardous carbon monoxide gas by utilizing a safe formic acid and acetic anhydride mixture as an in situ CO source. For R&D directors and process chemists, this methodology offers a streamlined pathway to access diverse libraries of bioactive molecules with high atom economy and operational simplicity, addressing critical bottlenecks in the development of next-generation therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-bridged biheterocycles has been fraught with significant challenges that hinder efficient production. Traditional approaches often rely on the direct coupling of two pre-formed heterocyclic substrates, which typically suffers from low regioselectivity and poor yields due to steric hindrance. Alternatively, oxidative cyclization strategies require harsh reaction conditions and stoichiometric amounts of toxic oxidants, generating substantial waste streams that complicate downstream purification. Most critically, classical carbonylation reactions necessitate the use of high-pressure carbon monoxide gas, posing severe safety risks and requiring specialized, expensive reactor infrastructure that many contract manufacturing organizations lack. These limitations collectively result in prolonged development timelines, inflated production costs, and restricted substrate scope, making the commercialization of such complex intermediates economically unviable for many potential drug candidates.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes a transition metal palladium-catalyzed carbonylation cascade that operates under remarkably mild conditions. By employing cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, the reaction achieves high efficiency without the need for toxic CO gas cylinders. The system leverages a formic acid and acetic anhydride mixture to release carbon monoxide in situ, ensuring a steady, controlled supply of the carbonyl source directly within the reaction medium. This innovation not only enhances laboratory safety but also drastically simplifies the engineering requirements for scale-up. Furthermore, the reaction exhibits exceptional functional group tolerance, accommodating various substituents like halogens, alkyls, and electron-withdrawing groups, thereby providing a versatile platform for medicinal chemistry optimization.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation involves a sophisticated sequence of organometallic steps initiated by the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This is followed by an intramolecular Heck-type reaction that generates a divalent alkyl-palladium intermediate, setting the stage for ring closure. Crucially, the subsequent carbonylation step occurs when this alkyl-palladium species intercepts the carbon monoxide released from the formic acid/acetic anhydride activation, forming a reactive acyl-palladium intermediate. Simultaneously, a base-promoted intermolecular reaction between the trifluoroethylimidoyl chloride and propargylamine constructs the trifluoroacetamidine moiety, which subsequently undergoes isomerization. The final cyclization is driven by the nucleophilic attack of the amidine nitrogen onto the activated acyl-palladium center, releasing the final carbonyl-bridged biheterocyclic product and regenerating the active palladium catalyst for the next cycle.
From an impurity control perspective, the use of specific ligands like tris(2-furyl)phosphine (TFP) plays a pivotal role in stabilizing the palladium center and preventing the formation of palladium black, which can lead to catalyst deactivation and metal contamination in the final API. The mild temperature of 30°C further suppresses side reactions such as polymerization of the acrylamide or decomposition of the sensitive imidoyl chloride. The compatibility of sodium carbonate as a mild base ensures that acid-sensitive functional groups on the substrate remain intact, while effectively neutralizing the HCl byproduct generated during the amidine formation. This precise balance of reactivity and selectivity ensures a clean reaction profile, minimizing the burden on purification teams and maximizing the overall yield of the desired high-purity intermediate.
How to Synthesize Carbonyl-Bridged Biheterocycles Efficiently
To implement this synthesis effectively, operators must adhere to strict stoichiometric ratios and mixing protocols as outlined in the patent examples. The standard procedure involves charging a reaction vessel with palladium chloride (5 mol%), TFP ligand (10 mol%), and sodium carbonate (2.0 equiv) in an anhydrous organic solvent such as tetrahydrofuran (THF). The CO source mixture, comprising formic acid and acetic anhydride (10 equiv total), is added alongside the three key coupling partners: trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative. The reaction mixture is then stirred at a controlled temperature of 30°C for a duration of 12 to 20 hours to ensure complete conversion. Post-reaction workup involves simple filtration to remove inorganic salts, followed by silica gel column chromatography to isolate the pure product.
- Mix palladium chloride catalyst, TFP ligand, sodium carbonate base, and the formic acid/acetic anhydride CO source in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and the acrylamide substrate to the reaction mixture under inert atmosphere.
- Stir the reaction at 30°C for 12 to 20 hours, then filter and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, this technology offers compelling economic and logistical benefits that directly impact the bottom line. The elimination of high-pressure carbon monoxide gas removes the need for specialized storage facilities and safety monitoring systems, leading to significant capital expenditure savings and reduced insurance premiums. Moreover, the starting materials, including propargylamine and various substituted acrylamides, are commodity chemicals available from multiple global suppliers, ensuring a resilient supply chain that is not dependent on single-source vendors. The ability to run the reaction at near-ambient temperatures (30°C) drastically reduces energy consumption compared to traditional high-temperature reflux processes, contributing to lower utility costs and a smaller carbon footprint for the manufacturing site.
- Cost Reduction in Manufacturing: The process utilizes inexpensive palladium chloride and common phosphine ligands rather than exotic, high-cost catalyst systems, and the low catalyst loading minimizes precious metal recovery costs. By avoiding the use of toxic gas cylinders and complex high-pressure reactors, the facility overheads are substantially reduced, allowing for more competitive pricing of the final intermediates. The high atom economy of the multicomponent reaction means less raw material is wasted as byproducts, further optimizing the cost of goods sold (COGS) for large-scale production campaigns.
- Enhanced Supply Chain Reliability: Since the key reagents are stable solids or liquids that do not require cryogenic storage or special transport permits for hazardous gases, logistics are simplified and lead times are shortened. The robustness of the reaction against moisture and air (to a reasonable extent given the Pd catalyst) allows for flexible scheduling and reduces the risk of batch failures due to minor environmental fluctuations. This reliability ensures consistent delivery schedules for downstream API manufacturers, mitigating the risk of production stoppages caused by raw material shortages or quality deviations.
- Scalability and Environmental Compliance: The protocol has been successfully demonstrated on a gram scale with clear pathways for expansion to kilogram and tonnage levels using standard stainless steel reactors. The absence of toxic CO emissions and the use of relatively benign solvents like THF simplify waste treatment and regulatory compliance, aligning with modern green chemistry principles. The straightforward workup involving filtration and chromatography avoids complex distillation or crystallization steps that often bottleneck scale-up, facilitating a smoother transition from pilot plant to commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity for technical teams evaluating this route for their specific projects. Understanding these nuances is essential for assessing the feasibility of adapting this chemistry to novel substrates or integrating it into existing manufacturing workflows.
Q: How does this method improve safety compared to traditional carbonylation?
A: Unlike conventional methods requiring high-pressure carbon monoxide gas cylinders, this protocol generates CO in situ from a formic acid and acetic anhydride mixture, significantly reducing explosion risks and infrastructure costs.
Q: What is the substrate scope for the R1 and R3 groups?
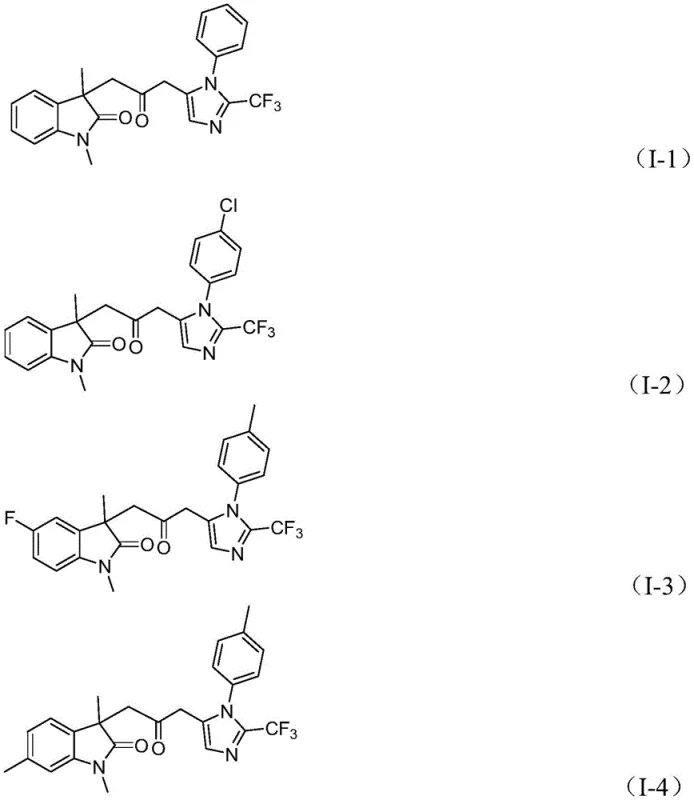
A: The method demonstrates excellent tolerance for various substituents including alkyl, halogen (Cl, Br, F), alkoxy, nitro, and trifluoromethyl groups on both the aryl rings and the acrylamide moiety.
Q: Is the palladium catalyst loading cost-effective for large scale?
A: Yes, the process utilizes low catalyst loading (5 mol% PdCl2) with inexpensive ligands like TFP, and the mild reaction conditions (30°C) minimize energy consumption, making it viable for industrial scaling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN115353511A for accelerating drug discovery pipelines. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with rigorous safety protocols, and our stringent purity specifications guarantee that every batch meets the highest quality standards required for pharmaceutical applications. With our rigorous QC labs and dedicated process development team, we are prepared to optimize this route for your specific target molecules, ensuring maximum yield and minimal impurity profiles.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this CO-free protocol for your specific API intermediate. We encourage you to contact us today to obtain specific COA data for similar structures and comprehensive route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain.
