Revolutionizing Biphenyl Synthesis: Ultra-Low Loading Nano Pd Catalyst for Commercial Scale-Up
The landscape of organic synthesis for biaryl scaffolds is undergoing a significant transformation driven by the need for greener, more cost-effective methodologies. Patent CN102267848A introduces a groundbreaking approach to synthesizing biphenyl compounds via a Suzuki coupling reaction that leverages a novel nano palladium catalyst. This technology addresses critical bottlenecks in traditional cross-coupling methods by utilizing aryl diazonium tetrafluoroborate salts as electrophilic partners instead of conventional aryl halides. The core innovation lies in the catalyst's exceptional stability and activity, which allows the reaction to proceed efficiently under aerobic conditions without the need for specialized ligands or rigorous anhydrous environments. For R&D directors and process chemists, this represents a paradigm shift towards simplified workflow integration, while procurement managers will recognize the immediate implications for raw material cost optimization and supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional Suzuki-Miyaura coupling reactions typically rely on aryl halides such as iodides or bromides as electrophilic substrates, which often necessitate the use of expensive phosphine ligands and strict inert atmosphere conditions to prevent catalyst deactivation. Furthermore, conventional palladium catalysts frequently require high loading levels, ranging from 1 mol% to 10 mol%, to achieve acceptable conversion rates, leading to significant residual metal contamination in the final product. This high metal burden imposes a heavy downstream burden on pharmaceutical manufacturers, who must implement costly and time-consuming purification steps to meet stringent regulatory limits for heavy metals in active pharmaceutical ingredients. Additionally, the sensitivity of many traditional catalyst systems to moisture and oxygen complicates the scale-up process, requiring specialized equipment and increasing the overall operational expenditure for large-scale manufacturing facilities.
The Novel Approach
In stark contrast, the methodology disclosed in this patent utilizes a uniquely prepared nano palladium catalyst that exhibits remarkable tolerance to air and water, thereby eliminating the need for complex inert gas setups. By employing aryl diazonium tetrafluoroborate salts, the reaction achieves superior reactivity even with electron-deficient or sterically hindered substrates, broadening the scope of accessible chemical space. The most striking advantage is the drastic reduction in catalyst loading to merely 0.1-0.3 mol%, which is substantially lower than the industry standard. This efficiency not only lowers the direct cost of the precious metal catalyst but also minimizes the environmental footprint associated with palladium waste. The process operates in benign solvents like methanol at mild temperatures, offering a streamlined pathway that aligns perfectly with modern principles of green chemistry and sustainable manufacturing practices.
Mechanistic Insights into Nano Pd-Catalyzed Suzuki Coupling
The efficacy of this synthetic route is rooted in the unique physicochemical properties of the nano palladium catalyst, which is synthesized through a controlled reduction process involving tetrakis triphenylphosphine palladium and polyol stabilizers. This preparation method yields nanometer-scale fibrous palladium particles that possess a high surface-area-to-volume ratio, maximizing the number of active sites available for the oxidative addition step of the catalytic cycle. Unlike homogeneous catalysts that may suffer from aggregation or decomposition under aerobic conditions, these nano-structured particles remain stable and dispersed, ensuring consistent catalytic performance throughout the reaction duration. The mechanism likely involves the facile oxidative addition of the aryl diazonium species to the Pd(0) surface, followed by transmetallation with the aryl boronic acid and subsequent reductive elimination to forge the carbon-carbon bond.
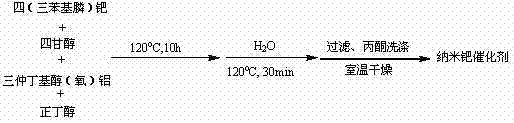
Furthermore, the use of aryl diazonium tetrafluoroborates as coupling partners offers distinct mechanistic advantages over aryl halides. The diazonium group is an excellent leaving group that facilitates rapid oxidative addition even at low temperatures, which helps suppress side reactions such as homocoupling of the boronic acid. The patent data indicates that the reaction proceeds smoothly with various substituents, including nitro, bromo, and methyl groups, demonstrating the robustness of the catalytic system against electronic variations. Impurity control is inherently improved because the mild reaction conditions and high selectivity minimize the formation of by-products, resulting in cleaner crude reaction mixtures. This high level of chemoselectivity is crucial for complex molecule synthesis where multiple functional groups might be present, reducing the need for protective group strategies and simplifying the overall synthetic sequence.
How to Synthesize Biphenyl Compounds Efficiently
The practical implementation of this technology involves a straightforward two-stage process beginning with the in-house or outsourced preparation of the specialized nano palladium catalyst, followed by the coupling reaction itself. The synthesis protocol is designed to be operator-friendly, utilizing standard laboratory glassware and avoiding the need for gloveboxes or Schlenk lines. Detailed standard operating procedures regarding stoichiometry, mixing rates, and temperature ramping are critical to reproducing the high yields reported in the patent literature. For process engineers looking to translate this from bench to plant, understanding the kinetics of the diazonium salt formation and its subsequent stability is key to ensuring safety and reproducibility. The following guide outlines the critical operational parameters derived from the patent examples to assist in method transfer.
- Prepare the nano Pd catalyst by reacting tetrakis triphenylphosphine palladium with tetraglycol and aluminum sec-butoxide in n-butanol at 120°C, followed by hydrolysis and filtration to obtain the active olive-green powder.
- Combine aryl diazonium tetrafluoroborate, aryl boronic acid (1.2 equivalents), and the nano Pd catalyst (0.3 mol%) in methanol solvent under ambient conditions without inert gas protection.
- Stir the reaction mixture at 25°C until TLC indicates completion, then remove solvent under reduced pressure and purify the residue via silica gel column chromatography to isolate the high-purity biphenyl product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this nano Pd-catalyzed protocol offers substantial strategic advantages for organizations managing the supply of fine chemical intermediates. The primary value driver is the significant reduction in raw material costs associated with the catalyst system. By lowering the palladium loading to trace levels, manufacturers can drastically reduce the consumption of this precious metal, which is subject to volatile market pricing. Moreover, the elimination of expensive ligands and the ability to use technical grade methanol instead of anhydrous solvents further contribute to a leaner cost structure. These savings can be passed down the supply chain or reinvested into R&D, providing a competitive edge in price-sensitive markets such as generic pharmaceuticals and agrochemicals.
- Cost Reduction in Manufacturing: The ultra-low catalyst loading of 0.3 mol% fundamentally alters the cost equation for biphenyl production. Traditional methods often require stoichiometric or near-stoichiometric amounts of additives and high catalyst loads, which inflate the bill of materials. By minimizing palladium usage, the process reduces the burden on downstream metal scavenging units, which are often capital-intensive and require consumable resins. This streamlined approach leads to a more efficient use of resources and a lower cost of goods sold, enhancing overall profit margins for high-volume production runs without compromising on product quality or purity specifications.
- Enhanced Supply Chain Reliability: The robustness of the catalyst against air and moisture translates directly into improved supply chain continuity. Facilities do not need to invest in specialized inert atmosphere infrastructure, reducing the risk of batch failures due to equipment malfunction or operator error. The use of readily available starting materials like aryl boronic acids and easily synthesized diazonium salts ensures that raw material sourcing remains stable and diversified. This resilience is vital for maintaining consistent delivery schedules to downstream customers, particularly in the pharmaceutical sector where supply interruptions can have severe regulatory and commercial consequences.
- Scalability and Environmental Compliance: Scaling this reaction is inherently safer and more environmentally friendly due to the mild operating conditions and the use of alcohol-based solvents. The reduction in heavy metal waste aligns with increasingly stringent global environmental regulations regarding effluent discharge and waste disposal. Companies adopting this technology can position themselves as sustainability leaders, appealing to eco-conscious clients and regulators. The simplified workup procedure, which often requires only filtration and crystallization rather than complex chromatographic separations, further enhances the throughput capacity of existing manufacturing plants, allowing for rapid response to market demand fluctuations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nano palladium catalytic system. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this method into their existing production portfolios. The answers highlight the practical benefits and operational considerations that distinguish this technology from legacy coupling methods.
Q: What are the advantages of using aryl diazonium tetrafluoroborate over traditional aryl halides in Suzuki coupling?
A: Aryl diazonium tetrafluoroborates serve as highly reactive electrophilic reagents that often eliminate the need for expensive ligands or harsh activation conditions required by aryl chlorides or bromides. This patent demonstrates that they enable efficient coupling under mild, aerobic conditions with significantly lower catalyst loading.
Q: How does the nano Pd catalyst improve supply chain stability for biphenyl production?
A: The catalyst is stable to both water and air, removing the stringent requirement for anhydrous solvents and inert atmosphere setups. This simplifies the manufacturing infrastructure, reduces operational complexity, and ensures consistent batch-to-batch reliability essential for large-scale supply chains.
Q: Is this method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes common solvents like methanol and operates at mild temperatures (0-50°C). The ultra-low catalyst loading of 0.3 mol% drastically reduces heavy metal contamination risks and downstream purification costs, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the nano Pd system described in CN102267848A for the production of high-value biphenyl intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of biphenyl derivative meets the exacting standards required by the global pharmaceutical and agrochemical industries. Our commitment to technical excellence allows us to offer customized solutions that optimize both yield and cost-efficiency for our partners.
We invite procurement leaders and R&D directors to collaborate with us on optimizing their supply chains for biphenyl-based scaffolds. By leveraging our expertise in nano-catalysis and continuous flow processing, we can help you achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. Together, we can drive innovation and efficiency in the manufacturing of next-generation fine chemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
