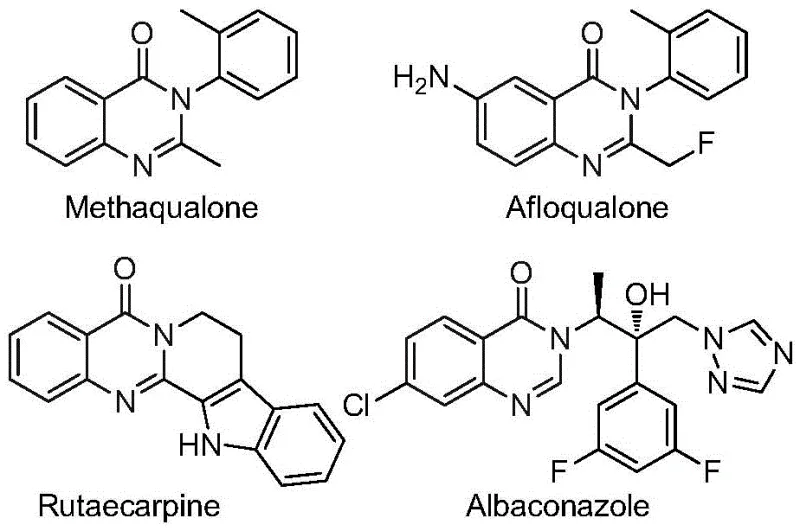
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Applications
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that possess significant biological activity, particularly within the quinazolinone class which is foundational to numerous therapeutic agents. As detailed in the recent intellectual property disclosure CN112480015B, a groundbreaking multi-component one-pot method has been developed for the efficient synthesis of 2-trifluoromethyl substituted quinazolinones. This technological advancement addresses critical bottlenecks in traditional heterocycle construction by utilizing cheap and readily available nitro compounds alongside trifluoroethylimidoyl chloride. The integration of a trifluoromethyl group is strategically vital for drug design, as it markedly enhances metabolic stability, lipophilicity, and bioavailability of the parent molecule. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a paradigm shift towards more economical and operationally simple manufacturing processes that bypass the need for hazardous high-pressure carbon monoxide gas.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied heavily on methodologies that impose severe logistical and economic burdens on the supply chain. Traditional synthetic pathways often necessitate the use of pre-activated substrates such as 2-bromoformylaniline or 2-iodoaniline, which are significantly more expensive and less atom-economical than their nitro counterparts. Furthermore, many established protocols require ruthenium or platinum catalysts operating under high-pressure carbon monoxide conditions, creating substantial safety hazards and requiring specialized reactor infrastructure that limits scalability. Other iron-catalyzed condensation reactions frequently suffer from narrow substrate scope and inconsistent yields, while palladium-catalyzed cyclizations involving acid anhydrides often demand rigorous exclusion of moisture and oxygen. These cumulative disadvantages result in elevated production costs, complex waste management profiles, and extended lead times for high-purity pharmaceutical intermediates, thereby hindering the rapid development of new drug candidates.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a transition metal palladium-catalyzed carbonylation cascade reaction that operates under remarkably mild and accessible conditions. By employing nitro compounds as the nitrogen source and trifluoroethylimidoyl chloride as the carbon framework builder, the process eliminates the need for pre-functionalized halogenated anilines. The reaction proceeds efficiently in 1,4-dioxane at a moderate temperature of 120°C, utilizing molybdenum hexacarbonyl as a safe, solid carbon monoxide surrogate instead of toxic gas cylinders. This multi-component one-pot strategy not only simplifies the operational workflow by combining reduction, coupling, and cyclization into a single vessel but also demonstrates exceptional functional group tolerance. Consequently, this methodology offers a streamlined pathway for cost reduction in pharmaceutical intermediate manufacturing by reducing unit operations and minimizing the consumption of precious metal catalysts through optimized ligand systems.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic elegance of this transformation lies in its intricate cascade sequence which effectively merges reduction and carbonylation chemistries. The reaction initiates with the reduction of the nitro compound to the corresponding amine species mediated by molybdenum hexacarbonyl, which simultaneously serves as the CO source. Following this in situ reduction, a base-promoted intermolecular carbon-nitrogen bond coupling occurs between the generated amine and the trifluoroethylimidoyl chloride to form a trifluoroacetamidine derivative. Subsequently, the palladium catalyst, coordinated by the dppp ligand, inserts into the carbon-iodine bond of the intermediate to generate a divalent palladium species. As the temperature is maintained at 120°C, the molybdenum complex releases carbon monoxide which inserts into the carbon-palladium bond, forming a crucial acyl-palladium intermediate. This sequence culminates in the formation of a seven-membered cyclic palladium intermediate facilitated by the base, followed by reductive elimination to release the final 2-trifluoromethyl substituted quinazolinone product.
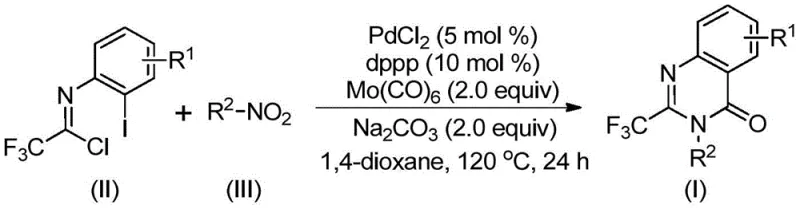
From an impurity control perspective, the selection of the 1,3-bis(diphenylphosphino)propane (dppp) ligand plays a pivotal role in stabilizing the palladium center and preventing the formation of homocoupling byproducts or dehalogenated side products. The use of sodium carbonate as a mild base ensures that the reaction environment remains conducive to cyclization without promoting hydrolysis of the sensitive imidoyl chloride starting material. Furthermore, the stoichiometric balance between the trifluoroethylimidoyl chloride and the nitro compound is carefully optimized, typically employing a slight excess of the nitro substrate to drive the equilibrium forward. This precise control over the catalytic cycle ensures that the resulting high-purity OLED material or pharmaceutical precursor meets stringent quality specifications with minimal downstream purification requirements, thereby enhancing the overall process mass intensity.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The execution of this synthesis requires careful attention to reagent quality and reaction parameters to maximize yield and reproducibility across different substrate classes. The protocol is designed to be robust, accommodating a wide variety of electronic and steric environments on both the nitroarene and the imidoyl chloride components. Operators should ensure that the molybdenum hexacarbonyl is fresh and handled under inert atmosphere conditions prior to charging, as its efficacy as a CO source is critical for the carbonylation step. The detailed standardized synthesis steps below outline the precise molar ratios and thermal conditions required to achieve the reported yields ranging from 69% to 96% across diverse examples.
- Charge a reaction vessel with palladium chloride, dppp ligand, sodium carbonate, molybdenum hexacarbonyl, trifluoroethylimidoyl chloride, and the specific nitro compound substrate in 1,4-dioxane solvent.
- Heat the reaction mixture to 120°C and maintain stirring for a duration of 16 to 30 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits regarding cost structure and logistical reliability. The primary driver of value is the substitution of expensive, specialized starting materials with commodity chemicals; nitro compounds are ubiquitous in the fine chemical market and are produced at massive scales, ensuring a stable and competitive pricing landscape. By eliminating the dependency on high-pressure gas infrastructure and exotic catalysts, the capital expenditure required for manufacturing is drastically simplified, allowing for production in standard glass-lined or stainless steel reactors. This flexibility translates directly into enhanced supply chain resilience, as the risk of disruption due to specialized equipment failure or scarce reagent availability is significantly mitigated. Moreover, the one-pot nature of the reaction reduces solvent consumption and waste generation, aligning with modern environmental compliance standards and reducing the burden on waste treatment facilities.
- Cost Reduction in Manufacturing: The economic advantage of this process is derived principally from the utilization of low-cost nitro compounds and the avoidance of high-pressure carbon monoxide equipment. By replacing expensive pre-activated halides with readily available nitro substrates, the raw material cost profile is substantially optimized. Additionally, the use of a solid CO surrogate removes the safety costs and regulatory overhead associated with handling toxic gases, leading to further indirect savings in insurance and facility maintenance. The high atom economy of the one-pot design minimizes solvent usage and purification steps, which collectively contribute to a lower cost of goods sold without compromising product quality.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for long-term production contracts, and this method leverages starting materials that are widely manufactured and stocked globally. Nitro compounds and simple palladium salts are standard inventory items for most chemical distributors, reducing the lead time for raw material acquisition. The robustness of the reaction conditions means that production schedules are less susceptible to delays caused by sensitive reagent degradation or strict storage requirements. This reliability ensures consistent delivery timelines for downstream customers, fostering stronger partnerships and enabling just-in-time manufacturing strategies for complex pharmaceutical intermediates.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is seamless due to the absence of hazardous high-pressure steps and the use of common organic solvents like 1,4-dioxane. The process generates minimal heavy metal waste relative to the product output, and the palladium catalyst can potentially be recovered or scavenged using standard industry practices. This environmental friendliness simplifies the permitting process for new manufacturing lines and reduces the ecological footprint of the synthesis. The ability to scale this reaction to multi-kilogram or tonnage levels without significant re-engineering makes it an ideal candidate for the commercial scale-up of complex polymer additives or drug substances.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation to provide clarity for potential licensees and manufacturing partners. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production portfolios.
Q: What are the key advantages of using nitro compounds in this synthesis?
A: Nitro compounds serve as inexpensive and readily available starting materials that replace costly pre-activated substrates like 2-bromoformylaniline, significantly reducing raw material costs while maintaining high reaction efficiency.
Q: How does the molybdenum hexacarbonyl function in this reaction system?
A: Molybdenum hexacarbonyl acts as a solid carbon monoxide surrogate, releasing CO in situ under heating conditions to facilitate the carbonylation step without requiring high-pressure gas equipment.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method has been demonstrated to be scalable to gram levels with simple post-treatment procedures involving filtration and column chromatography, making it viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes in accelerating drug development timelines and optimizing production costs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial reality is smooth and compliant. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-trifluoromethyl quinazolinone meets the highest international standards. Our commitment to quality and consistency makes us a trusted partner for multinational corporations seeking to secure their supply chains for critical pharmaceutical intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver high-value chemical solutions that drive your business forward.
