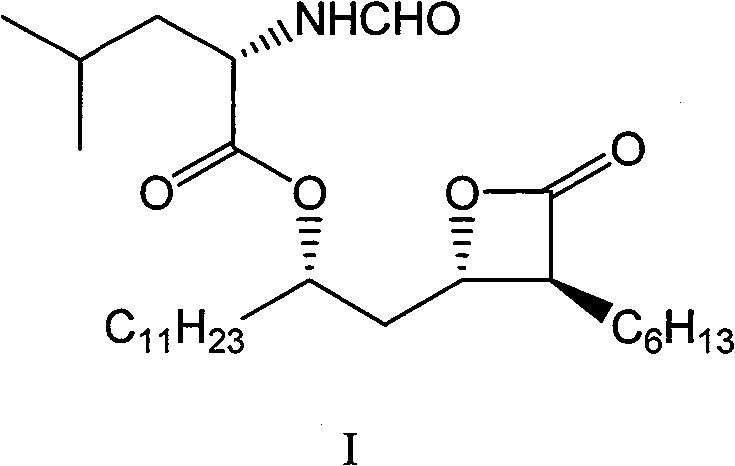
Advanced Chemical Synthesis of Orlistat Intermediates for Commercial Scale Production
The pharmaceutical industry's demand for high-purity anti-obesity agents has driven significant innovation in synthetic methodology, particularly for lipase inhibitors like Orlistat. Patent CN101066924B discloses a robust, twelve-step chemical synthesis route for Orlistat and its key intermediates, specifically targeting the (2S,3S,5S)-2-hexyl-3-hydroxy-5-R1oxyhexadecanoic acid R2 ester scaffold. Unlike traditional biological fermentation methods which often struggle with yield consistency and purification complexity, this chemical approach leverages precise stereocontrol mechanisms starting from the inexpensive and readily available chiral pool material, (S)-epichlorohydrin. The route is characterized by its operational simplicity, high optical purity, and reliance on standard organic transformations that are inherently scalable for commercial manufacturing environments. This technical insight analyzes the critical reaction engineering and strategic advantages embedded within this patent for potential licensees and procurement partners.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Orlistat and related lipase inhibitors has relied heavily on fermentation processes, such as those described in earlier patents like ZL97109032.1. While biocatalysis offers inherent chirality, these methods are frequently plagued by low volumetric productivity and substantial downstream processing costs associated with separating the active pharmaceutical ingredient from complex biological broths. The fermentation route often results in variable yields due to the sensitivity of microbial strains to environmental conditions, leading to supply chain instability and batch-to-batch inconsistency. Furthermore, the purification of fermentation products typically requires extensive chromatography and crystallization steps to remove biological impurities, significantly driving up the cost of goods sold (COGS) and extending the overall production lead time for pharmaceutical manufacturers seeking reliable bulk supply.
The Novel Approach
In stark contrast, the methodology outlined in CN101066924B presents a fully synthetic pathway that circumvents the biological bottlenecks of fermentation. By initiating the synthesis with (S)-epichlorohydrin, the process capitalizes on a commodity chemical with established global supply chains, ensuring raw material availability and price stability. The route employs a strategic sequence of carbon-chain extensions and functional group interconversions, including a novel CuI-catalyzed ring-opening reaction that preserves optical integrity while building the requisite carbon skeleton. This chemical approach allows for rigorous process control over reaction parameters such as temperature and stoichiometry, resulting in a more predictable impurity profile and facilitating easier purification. The elimination of biological variability translates directly into a more robust manufacturing process capable of meeting the stringent quality standards required for regulatory submission and commercial distribution.
Mechanistic Insights into CuI-Catalyzed Ring Opening and Stereoselective Reduction
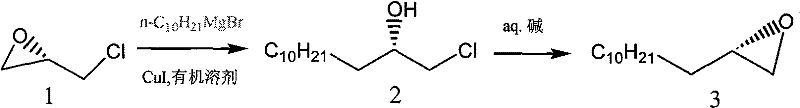
The cornerstone of this synthesis lies in the initial construction of the chiral backbone, where the patent details a sophisticated application of organometallic chemistry to solve regioselectivity challenges. In the first critical step, (S)-epichlorohydrin reacts with a decyl Grignard reagent. Under normal conditions, Grignard reagents are highly reactive and prone to attacking the carbon atom bearing the chlorine substituent, which would lead to the loss of chirality and the formation of the wrong regioisomer. However, the introduction of a catalytic amount of Copper(I) Iodide (CuI) fundamentally alters the reaction mechanism. The CuI acts as a transmetallation agent, generating a softer organocuprate species in situ that preferentially attacks the less hindered epoxide carbon. This subtle yet powerful modification ensures that the ring-opening occurs with inversion of configuration at the desired site while leaving the chiral center intact, setting the stage for the subsequent stereochemical fidelity of the entire molecule.
Further down the synthetic sequence, the establishment of the critical syn-diol motif in compound 8 is achieved through a highly stereoselective reduction of a beta-keto ester precursor. The patent specifies the use of diethylmethoxyborane (Et2BOMe) in combination with sodium borohydride (NaBH4) at cryogenic temperatures (-78°C). This reagent system facilitates a chelation-controlled reduction where the boron atom coordinates with both the carbonyl oxygen and the adjacent hydroxyl group, locking the substrate into a rigid cyclic transition state. This geometric constraint forces the hydride delivery to occur from a specific face of the molecule, thereby inducing the formation of the syn-diastereomer with exceptional selectivity. The patent reports an enantiomeric excess (ee) value greater than 99% for this step, demonstrating the efficacy of this boron-mediated protocol in controlling absolute stereochemistry, which is paramount for the biological activity of the final Orlistat product.
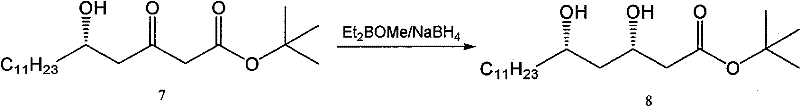
How to Synthesize Orlistat Intermediates Efficiently
The synthesis of these high-value pharmaceutical intermediates requires precise adherence to the reaction conditions detailed in the patent to ensure optimal yield and purity. The process begins with the preparation of the Grignard reagent under strictly anhydrous conditions, followed by the copper-catalyzed coupling which must be maintained at controlled temperatures to prevent side reactions. Subsequent steps involve careful pH control during the cyanide substitution and hydrolysis phases to minimize byproduct formation. The critical reduction step demands rigorous temperature management at -78°C to maintain the high diastereoselectivity observed in the patent examples. For a comprehensive understanding of the specific molar ratios, solvent choices, and workup procedures required to replicate this success, please refer to the standardized synthesis guide below.
- Perform CuI-catalyzed ring opening of (S)-epichlorohydrin with decyl Grignard reagent to form the chiral chlorohydrin intermediate.
- Execute stereoselective reduction of the beta-keto ester using Et2BOMe/NaBH4 at -78°C to establish the syn-diol motif with >99% ee.
- Complete the synthesis via alpha-alkylation, selective esterification with N-protected leucine, and final lactonization using benzenesulfonyl chloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the transition from fermentation-based production to the chemical synthesis route described in CN101066924B offers transformative economic and logistical benefits. The reliance on (S)-epichlorohydrin as a starting material decouples production from the biological constraints of fermentation, such as strain degradation or contamination risks, thereby enhancing supply continuity. The use of common organic solvents like tetrahydrofuran, toluene, and methanol ensures that the process can be executed in standard multipurpose chemical reactors without the need for specialized bioreactor infrastructure. This compatibility with existing fine chemical manufacturing assets significantly lowers the barrier to entry for scale-up and reduces the capital expenditure required for technology transfer. Furthermore, the streamlined nature of the synthetic route, which avoids complex enzymatic steps, simplifies the regulatory filing process by providing a well-defined chemical impurity profile that is easier to characterize and control.
- Cost Reduction in Manufacturing: The elimination of expensive fermentation substrates and the associated downstream purification costs leads to a substantial reduction in the overall cost of goods. By utilizing commodity chemicals and avoiding the low-yield bottlenecks typical of biological processes, the chemical route achieves a more efficient atom economy. The ability to perform telescoped reactions, such as the direct conversion of intermediates without isolation in certain steps, further minimizes solvent usage and processing time. This efficiency translates into a more competitive pricing structure for the final API, allowing pharmaceutical companies to improve their margins or pass savings on to healthcare providers while maintaining high-quality standards.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this chemical synthesis is significantly more robust compared to securing specialized biological cultures or fermentation media. The precursors, including epichlorohydrin and various alkyl halides, are produced on a massive industrial scale globally, ensuring consistent availability and shielding the supply chain from agricultural or biological disruptions. The synthetic process is less susceptible to environmental variables, meaning production schedules can be met with greater predictability. This reliability is crucial for pharmaceutical companies managing just-in-time inventory systems and facing strict deadlines for drug product launches, as it mitigates the risk of stockouts caused by production failures.
- Scalability and Environmental Compliance: The reaction conditions described, ranging from ambient temperature to moderate cryogenic cooling, are readily adaptable to large-scale batch reactors used in the fine chemical industry. The process avoids the generation of large volumes of biological waste water, which can be costly and difficult to treat, replacing it with organic waste streams that can be managed through standard incineration or solvent recovery systems. The high selectivity of the key steps reduces the formation of isomeric impurities, minimizing the need for resource-intensive chromatographic separations. This cleaner process profile not only facilitates easier scale-up from pilot plant to commercial tonnage but also aligns with increasingly stringent environmental regulations regarding waste disposal and solvent emissions in pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of the method for potential partners and technical evaluators.
Q: What is the primary advantage of this chemical synthesis over fermentation?
A: The chemical route described in CN101066924B offers significantly higher total yield and operational simplicity compared to fermentation methods, which suffer from low production efficiency and complex downstream processing.
Q: How is stereochemical integrity maintained during the Grignard reaction?
A: The process utilizes a catalytic amount of CuI to modulate the reactivity of the Grignard reagent, preventing direct attack at the chlorine-bearing carbon and ensuring retention of configuration at the chiral center.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states the method is suitable for industrial production, utilizing standard organic solvents like THF and toluene, and avoiding exotic biological reagents that limit scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Orlistat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure necessary to bring complex synthetic routes like the one described in CN101066924B from the laboratory bench to full-scale commercial production. Our team of experienced process chemists specializes in optimizing asymmetric syntheses and managing sensitive organometallic reactions, ensuring that the high stereochemical purity reported in the patent is maintained during scale-up. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, utilizing our state-of-the-art facilities equipped for cryogenic reactions and hazardous reagent handling. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that employ advanced analytical techniques to verify the identity and potency of every batch, guaranteeing that our intermediates meet the exacting standards of the global pharmaceutical market.
We invite you to collaborate with us to leverage this innovative synthesis technology for your Orlistat supply needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this chemical route can optimize your budget. Please contact us today to request specific COA data for our reference standards and to discuss route feasibility assessments for your upcoming projects. Let us be your partner in delivering high-quality, cost-effective pharmaceutical intermediates that drive your business forward.
