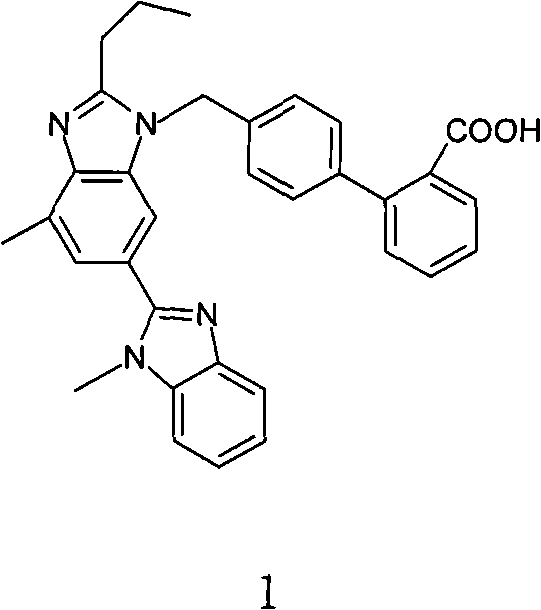
Advanced Telmisartan Refining via Salt Recrystallization for Commercial Scale-up
Advanced Telmisartan Refining via Salt Recrystallization for Commercial Scale-up
The pharmaceutical industry continuously demands higher purity standards for Angiotensin II receptor antagonists, particularly for blockbuster drugs like Telmisartan. A pivotal advancement in this domain is detailed in patent CN101838243A, which discloses a robust method for refining Telmisartan that circumvents the limitations of traditional ester-based purification routes. This innovative approach leverages a direct salt formation and recrystallization strategy to achieve exceptional purity levels exceeding 99.8 percent while maintaining a pristine color profile. For R&D directors and procurement specialists, understanding this technology is critical for securing a reliable Telmisartan supplier capable of delivering high-quality active pharmaceutical ingredients without the baggage of solvent residues. The following analysis dissects the technical merits and commercial viability of this purification protocol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of clinical-grade Telmisartan has relied heavily on pathways involving the purification of Telmisartan esters, such as methyl, ethyl, or tert-butyl esters, followed by alkaline hydrolysis. While effective in generating the core structure, these conventional methods suffer from significant downstream processing drawbacks. A primary concern is the frequent necessity of using high-boiling polar aprotic solvents, most notably dimethylformamide (DMF), during the refining stages. The presence of DMF creates a persistent challenge in manufacturing, as its high boiling point makes complete removal difficult, often leading to unacceptable solvent residues in the final API. Furthermore, products derived from these ester-based routes frequently exhibit poor appearance, characterized by a yellowish tint and lower purity profiles generally hovering below 99.5 percent, necessitating additional, costly reprocessing steps to meet pharmacopoeial standards.
The Novel Approach
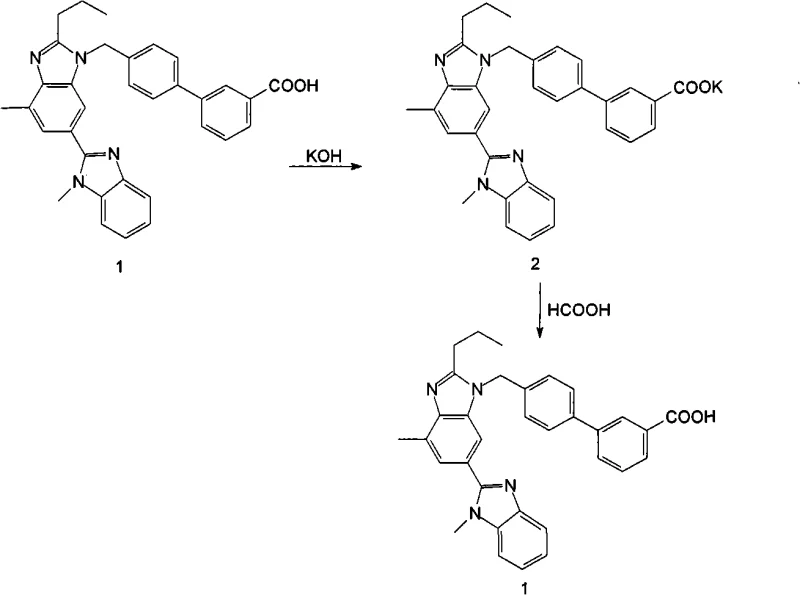
In stark contrast, the novel methodology outlined in the patent data introduces a streamlined three-step process that bypasses esterification entirely, focusing instead on the manipulation of the Telmisartan acid salt. This approach begins with the reaction of crude Telmisartan with an alkali to form a crude salt, followed by a dedicated recrystallization of this salt, and concludes with acidification to regenerate the pure free acid. By shifting the purification focus to the salt form, the process exploits differences in solubility and crystallization behavior that are more favorable than those of the free acid or its esters. This strategic pivot allows for the use of low-boiling, environmentally friendlier solvents like isopropanol and ethyl acetate, effectively eliminating the risk of high-boiling solvent residues and significantly enhancing the visual quality of the final crystal lattice.
Mechanistic Insights into Salt-Mediated Purification
The core mechanism driving the success of this refining technique lies in the physicochemical properties of the Telmisartan salt intermediates. Telmisartan possesses a strongly acidic carboxylic acid group due to the conjugated system within its molecular architecture, allowing it to react readily with various inorganic and organic bases to form stable salts. In the first stage, reacting the crude material with a base like potassium hydroxide in a solvent such as isopropanol converts the impure acid into a salt form that is more amenable to purification. This conversion is not merely a protection step but a purification vector; the ionic nature of the salt alters its interaction with impurities, facilitating their separation during the subsequent crystallization phase. The addition of activated carbon during this salt formation stage further adsorbs colored organic impurities, which are then mechanically removed via filtration, setting the stage for a high-purity outcome.
Following the initial salt formation, the recrystallization step serves as the critical polishing operation. By dissolving the crude salt in a specific mixed solvent system, such as isopropanol and ethyl acetate, and controlling the temperature profile, the process induces the selective precipitation of the Telmisartan salt while leaving soluble impurities in the mother liquor. The patent highlights that this step can also incorporate activated carbon treatment to ensure maximum decolorization. Finally, the refined salt is dissolved in a methanol-water mixture and treated with a mild acid, such as formic acid, to adjust the pH to the isoelectric range of 4-6. This controlled acidification triggers the precipitation of the Telmisartan free acid in a highly ordered, colorless crystalline form, ensuring that the final product meets stringent specifications for both chemical purity and physical appearance.
How to Synthesize Telmisartan Efficiently
The synthesis of high-purity Telmisartan via this salt-mediated route offers a practical and scalable solution for manufacturers aiming to optimize their production lines. The process is designed to be operationally simple, avoiding the complex multi-step sequences associated with ester hydrolysis methods. By focusing on direct salt manipulation, facilities can reduce cycle times and minimize the handling of hazardous high-boiling solvents. The detailed standardized synthesis steps for implementing this purification protocol are provided in the guide below, outlining the precise stoichiometric ratios and thermal conditions required to replicate the high yields and purity reported in the patent literature.
- React crude Telmisartan with alkali (e.g., KOH) in a low-boiling solvent like isopropanol to form the crude telmisartan salt, optionally using activated carbon for decolorization.
- Recrystallize the crude telmisartan salt in a mixed solvent system (e.g., isopropanol and ethyl acetate) to remove impurities and obtain refined telmisartan salt.
- Dissolve the refined salt in a methanol-water mixture, adjust pH to 4-6 using formic acid to precipitate the final colorless to white Telmisartan crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this refining technology translates into tangible operational efficiencies and risk mitigation. The elimination of high-boiling solvents like DMF removes the need for energy-intensive distillation processes required for solvent recovery, thereby reducing utility costs and environmental compliance burdens. Furthermore, the simplified workflow enhances the reliability of the supply chain by reducing the number of unit operations where yield losses or quality deviations could occur. This robustness ensures a more consistent output of high-purity Telmisartan, safeguarding against production delays that could impact downstream formulation schedules.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and difficult-to-remove solvents with commodity chemicals like isopropanol and ethyl acetate. By avoiding the use of DMF, manufacturers eliminate the substantial costs associated with solvent recovery systems and the rigorous testing required to certify low residual levels in the final API. Additionally, the high yields reported in the patent examples suggest a more efficient utilization of raw materials, meaning less waste is generated per kilogram of finished product. This efficiency directly lowers the cost of goods sold (COGS), providing a competitive pricing advantage in the global market for antihypertensive agents.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on common, widely available solvents mitigates the risk of supply disruptions often associated with specialized reagents. The process operates under relatively mild thermal conditions, typically ranging from ambient to reflux temperatures of common alcohols, which reduces the strain on reactor equipment and minimizes maintenance downtime. The ability to produce a colorless, high-purity product consistently means that batches are less likely to be rejected during quality control, ensuring a steady flow of inventory to meet market demand. This reliability is crucial for maintaining long-term contracts with generic drug manufacturers who require uninterrupted supply of key intermediates.
- Scalability and Environmental Compliance: The simplicity of the crystallization and filtration steps makes this process inherently scalable from pilot plant to commercial production volumes. The absence of toxic high-boiling solvents simplifies waste stream management, as the effluent is easier to treat and dispose of in accordance with environmental regulations. The use of activated carbon for decolorization is a well-established, scalable technology that integrates seamlessly into existing infrastructure without requiring major capital investment in new equipment. Consequently, facilities can ramp up production capacity to meet surging demand for Telmisartan while maintaining a smaller environmental footprint, aligning with modern green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this Telmisartan refining method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on purity benchmarks, solvent choices, and process scalability. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial manufacturing.
Q: How does this purification method improve product color compared to conventional routes?
A: By utilizing a salt formation and recrystallization strategy with activated carbon treatment at multiple stages, this method effectively removes colored impurities that typically persist when using high-boiling solvents like DMF, resulting in a colorless to white final product.
Q: What solvents are used to avoid residue issues in the final API?
A: The process exclusively employs low-boiling solvents such as methanol, ethanol, isopropanol, and ethyl acetate, which are easily removed during drying, ensuring the finished product is free from problematic high-boiling solvent residues.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the operational path is simplified by eliminating complex esterification and hydrolysis steps, relying instead on straightforward crystallization and filtration unit operations that are highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Telmisartan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of cardiovascular therapeutics. Our technical team has extensively analyzed advanced purification routes, including the salt-recrystallization method described herein, to ensure our manufacturing capabilities align with the highest industry standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver Telmisartan with stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify that every batch meets the colorless, high-purity criteria demanded by global regulatory bodies, ensuring your supply chain remains secure and compliant.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your specific project requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that demonstrates how our efficient solvent systems and high-yield protocols can reduce your overall procurement expenses. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on verified performance metrics and our commitment to excellence in fine chemical manufacturing.
