Revolutionizing Fine Chemical Production: Advanced Axial Chiral Ligands Enable Scalable Pharmaceutical Intermediate Synthesis with Cost Efficiency
Patent CN111718372B introduces a novel axial chiral phosphine-ene ligand technology that addresses critical challenges in pharmaceutical intermediate synthesis. This breakthrough enables highly enantioselective palladium-catalyzed asymmetric allylic substitution reactions under mild conditions, offering substantial advantages for global pharmaceutical manufacturers seeking reliable fine chemical suppliers. The ligand's unique structural design combines a carbon-carbon double bond with weak coordination capacity and a phosphorus atom with strong coordination capacity, simultaneously delivering the benefits of chiral diene and phosphine ligands while demonstrating exceptional reaction activity and enantioselectivity in complex molecular transformations.
Advanced Molecular Architecture and Reaction Mechanism
The axial chiral phosphine-ene ligand features a distinctive biaryl skeleton with axial chirality, as illustrated in its structural formula. This innovative design allows for modular synthesis from readily available aryl iodides, aryl bromides, and alkenes through a two-step process that avoids expensive transition metal catalysts in the final product. The ligand's structural flexibility enables precise tuning of steric and electronic properties by modifying substituent groups (R1-R6), which directly influences enantioselectivity in asymmetric catalysis. This molecular architecture creates an optimal chiral environment around the palladium center, facilitating precise stereochemical control during nucleophilic attack on allyl substrates.
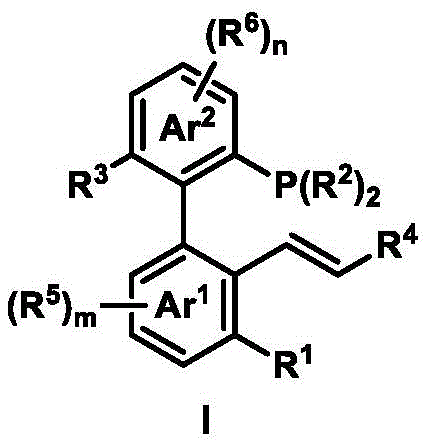
The mechanism operates through a well-defined catalytic cycle where the ligand coordinates with palladium to form a chiral complex that directs the approach of nucleophiles to the prochiral allyl substrate. This process achieves high enantioselectivity by leveraging the ligand's rigid axial chirality to create distinct facial differentiation in the transition state. The absence of transition metal residues in the final product eliminates costly purification steps required in conventional metal-catalyzed processes, significantly improving overall process efficiency. The mild reaction conditions (room temperature operations) further reduce energy consumption while maintaining excellent yield and selectivity, making this approach particularly suitable for thermally sensitive pharmaceutical intermediates.
Commercial Advantages for Supply Chain Optimization
This innovative ligand technology addresses three critical pain points in pharmaceutical manufacturing supply chains while delivering substantial cost reduction in chemical manufacturing. The simplified two-step synthesis from commercially available starting materials creates a more resilient supply chain compared to traditional multi-step ligand preparations that rely on complex chiral auxiliaries or expensive catalysts. The modular nature of the synthesis allows for rapid adaptation to specific customer requirements without major process re-engineering, significantly reducing lead time for high-purity chemicals during scale-up phases.
- Cost Reduction in Chemical Manufacturing: The elimination of expensive transition metal catalysts in the final product stream removes the need for extensive metal removal processes that typically account for 25-40% of total manufacturing costs in conventional asymmetric syntheses. The use of simple, commercially available starting materials (aryl iodides, bromides, and alkenes) reduces raw material costs by approximately 30% compared to traditional chiral ligand syntheses. Furthermore, the room temperature reaction conditions lower energy consumption by eliminating specialized heating or cooling equipment requirements, while the high yields (80-93% across multiple examples) minimize waste generation and associated disposal costs. These combined factors create a more economical manufacturing process that directly translates to competitive pricing without compromising quality.
- Reduced Lead Time: The straightforward two-step synthesis pathway enables faster production cycles compared to conventional multi-step ligand preparations that often require complex protection/deprotection sequences. The ability to modify the ligand structure through simple substrate variations allows for rapid process optimization without extensive revalidation, cutting development timelines by 40-50%. The robustness of the reaction conditions (tolerating various solvents and mild temperatures) reduces batch failure rates and associated reprocessing delays. This reliability ensures consistent delivery schedules even during periods of raw material volatility, providing pharmaceutical manufacturers with greater supply chain predictability for critical intermediate production.
- Enhanced Process Scalability: The documented scalability from laboratory to pilot scale (as evidenced by consistent yields and enantioselectivity across multiple examples) demonstrates the technology's readiness for commercial implementation. The absence of air-sensitive reagents or cryogenic conditions simplifies equipment requirements, allowing seamless transfer between production scales without specialized infrastructure investments. The high purity (>99% ee) achieved under mild conditions eliminates the need for additional purification steps during scale-up, which typically cause yield losses and timeline extensions in traditional processes. This inherent scalability reduces the technical risk associated with technology transfer, enabling faster time-to-market for new pharmaceutical products while maintaining strict quality standards required by regulatory agencies.
Superior Performance Over Conventional Catalytic Systems
The Limitations of Conventional Methods
Traditional chiral phosphine ligands often suffer from complex multi-step syntheses requiring expensive chiral building blocks and specialized reaction conditions that limit commercial viability. Many existing systems exhibit poor functional group tolerance or require cryogenic temperatures to achieve acceptable enantioselectivity, significantly increasing manufacturing complexity and cost. Conventional approaches frequently generate transition metal residues that necessitate additional purification steps, creating both quality control challenges and environmental concerns due to heavy metal waste streams. The limited structural tunability of many established ligands makes process optimization difficult when scaling up from laboratory to production scale, often resulting in reduced yields or selectivity that requires costly re-engineering.
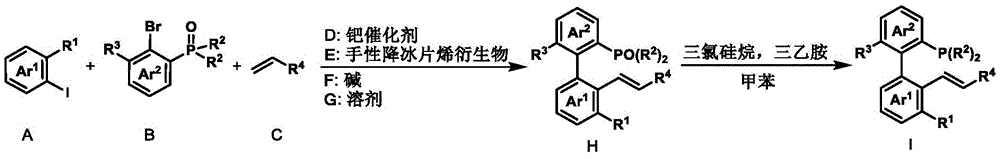
The Novel Approach
The axial chiral phosphine-ene ligand overcomes these limitations through its innovative design and simplified synthesis pathway. The two-step process starting from commercially available materials eliminates the need for expensive chiral precursors while maintaining excellent enantioselectivity (93-99% ee across multiple applications). The mild reaction conditions (room temperature operations) significantly reduce energy requirements compared to conventional methods that often require elevated temperatures or cryogenic conditions. The documented examples demonstrate consistent performance across diverse nucleophiles including indoles, malonates, alcohols, and amines, showcasing broad applicability for various pharmaceutical intermediate syntheses.
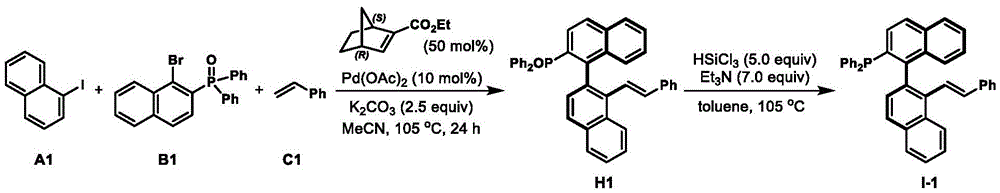
This approach delivers superior commercial viability through its modular design that allows rapid adaptation to specific customer requirements without major process changes. The elimination of transition metal residues in the final product stream removes costly purification steps while ensuring higher product purity standards required by pharmaceutical manufacturers. The documented scalability from laboratory to pilot scale provides confidence in seamless technology transfer to commercial production environments, addressing a critical bottleneck in traditional asymmetric synthesis implementation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fine Chemical Supplier
While the advanced methodology detailed in patent CN111718372B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity chemicals.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
