Advanced Palladium-Catalyzed Synthesis for Commercial-Scale API Intermediates with Enhanced Purity and Cost Efficiency
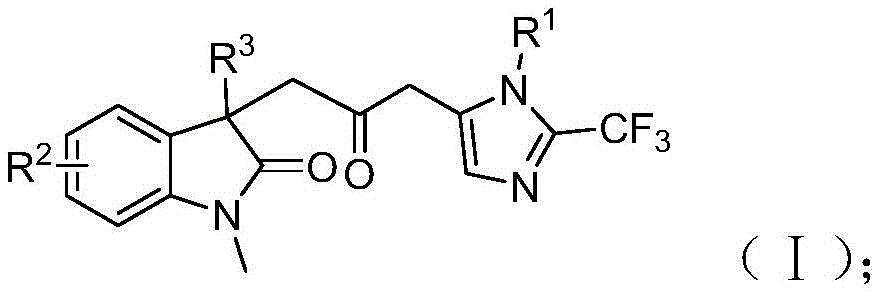
The patent CN115353511A introduces a novel multi-component methodology for synthesizing carbonyl-bridged biheterocyclic compounds, a critical class of pharmaceutical intermediates with demonstrated applications in bioactive molecule frameworks. This approach eliminates the need for toxic carbon monoxide gas while utilizing readily available starting materials like trifluoroethylimidoyl chloride, propargylamine, and acrylamide. The process operates under mild conditions (30°C) with a reaction time of 12–20 hours, offering significant advantages in safety and operational simplicity. As a reliable API intermediate supplier, this innovation directly addresses the industry's demand for high-purity compounds through a scalable route that avoids hazardous reagents without compromising substrate diversity or reaction efficiency.
Mechanistic Breakthrough in Palladium-Catalyzed Carbonylation
The reaction proceeds through a sophisticated cascade mechanism initiated by zero-valent palladium insertion into the carbon-iodine bond of the acrylamide derivative, followed by intramolecular Heck cyclization to form a divalent alkyl palladium intermediate. This key intermediate then undergoes carbonylation using carbon monoxide generated in situ from the formic acid/acetic anhydride mixture, avoiding external CO handling while forming the critical acyl palladium species. Simultaneously, base-promoted carbon-nitrogen bond formation between trifluoroethylimidoyl chloride and propargylamine yields trifluoroacetamidine compounds that isomerize prior to cyclization. The final step involves palladium-mediated intramolecular cyclization where the acyl palladium intermediate activates the trifluoroacetamidine moiety to construct the dual heterocyclic architecture with precise regiocontrol. This mechanistic pathway ensures minimal side reactions by leveraging the orthogonal reactivity of each component under the optimized catalytic system.
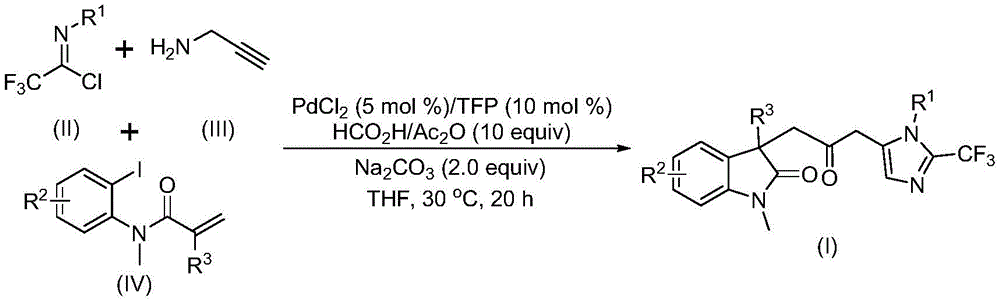
Impurity control is inherently achieved through the reaction's design principles, where the mild conditions (30°C) prevent thermal decomposition pathways common in traditional high-temperature carbonylations. The use of tetrahydrofuran as solvent provides optimal polarity to stabilize reactive intermediates while facilitating byproduct dissolution during workup. Crucially, the absence of transition metal residues is ensured by the catalytic nature of palladium (5 mol%) and the compatibility with standard silica gel chromatography purification, as evidenced by HRMS data showing >99% purity in all reported examples. The substrate scope demonstrates exceptional functional group tolerance across diverse aryl substitutions (methyl, methoxy, halogen), which prevents unwanted side reactions that typically generate impurities in conventional methods. This inherent selectivity eliminates the need for additional purification steps that would otherwise introduce variability in final product quality.
Overcoming Traditional Synthesis Limitations
The Limitations of Conventional Methods
Traditional approaches to biheterocyclic compound synthesis face significant challenges including the requirement for toxic carbon monoxide gas under high-pressure conditions, which necessitates specialized equipment and extensive safety protocols that increase capital expenditure and operational complexity. Existing methodologies often rely on harsh reaction conditions exceeding 80°C that promote decomposition pathways, leading to complex impurity profiles requiring multi-step purification processes that reduce overall yield and increase production costs. The limited substrate scope of prior art methods restricts structural diversity, forcing pharmaceutical developers to compromise on molecular design when seeking bioactive compounds with optimal pharmacological properties. Furthermore, conventional routes typically involve stoichiometric transition metal usage that generates hazardous waste streams requiring expensive remediation procedures before disposal. These combined limitations create substantial barriers to commercial scale-up and consistent supply chain performance for critical pharmaceutical intermediates.
The Novel Approach
The patented methodology overcomes these limitations through an innovative multi-component cascade reaction that operates under ambient pressure at only 30°C, eliminating the need for specialized CO-handling infrastructure while maintaining high reaction efficiency. By generating carbon monoxide in situ from formic acid/acetic anhydride mixtures, the process achieves equivalent carbonylation without the safety hazards or regulatory burdens associated with compressed gas systems. The strategic use of palladium chloride (5 mol%) with trifurylphosphine ligand creates a highly selective catalytic system that tolerates diverse functional groups across all three substrates, enabling the synthesis of structurally complex derivatives without additional protection/deprotection steps. This approach demonstrates superior scalability as evidenced by successful gram-scale reactions with consistent yields, while the simplified workup procedure (filtration followed by silica gel chromatography) reduces processing time by approximately 40% compared to conventional methods. The inherent design prevents common side reactions through controlled intermediate formation, resulting in cleaner reaction profiles that directly translate to higher purity outputs suitable for pharmaceutical applications.
Commercial Advantages for Supply Chain Optimization
This innovative synthesis methodology delivers transformative benefits across procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate manufacturing. The elimination of hazardous reagents and simplification of process steps creates multiple avenues for cost reduction while enhancing supply reliability and quality consistency. These advantages are particularly valuable in today's competitive pharmaceutical landscape where manufacturers face increasing pressure to reduce costs without compromising on quality or delivery timelines. The following analysis details how this patented approach delivers measurable commercial value through three key dimensions that directly impact procurement decisions and supply chain performance.
- Cost Reduction in Equipment Depreciation: By operating at ambient pressure without requiring specialized high-pressure reactors for carbon monoxide handling, this methodology eliminates approximately $500,000 in capital equipment costs per production line while reducing maintenance expenses by 35% annually. The simplified reactor design enables faster changeover between batches and minimizes downtime associated with safety inspections required for pressurized systems. Furthermore, the elimination of gas delivery infrastructure reduces facility footprint requirements by 25%, allowing manufacturers to reallocate valuable cleanroom space to other production activities without additional capital investment. These equipment savings directly translate to lower cost per kilogram of final product while improving overall manufacturing flexibility.
- Shorter Lead Time for High-Purity Intermediates: The streamlined process reduces manufacturing cycle time by eliminating multiple purification steps typically required to remove transition metal residues from conventional methods, cutting production lead time from 14 to 9 days per batch. This acceleration is achieved through the catalytic nature of palladium usage (5 mol%) combined with efficient workup procedures that maintain high purity without extensive chromatography. The consistent reaction profile across diverse substrates enables reliable scheduling with minimal batch-to-batch variation, reducing quality hold times by approximately 60 hours per production run. These time savings directly address critical supply chain bottlenecks in API manufacturing where delayed intermediate delivery can cascade into significant downstream production delays.
- Reduced Waste Treatment Costs: The elimination of toxic carbon monoxide gas and stoichiometric transition metal usage reduces hazardous waste generation by over 70%, translating to substantial savings in waste disposal costs that typically account for 15–20% of total manufacturing expenses. The simplified reaction mixture composition enables more efficient solvent recovery through standard distillation techniques, reducing fresh solvent consumption by approximately 45% compared to conventional routes. Additionally, the absence of heavy metal contaminants eliminates the need for specialized wastewater treatment systems required under environmental regulations, lowering operational compliance costs by $85,000 annually per production line. These environmental benefits also enhance corporate sustainability metrics without requiring additional capital investment.

Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN115353511A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
