Metal-Free Synthesis of High-Purity Triazole Intermediates: Scalable Manufacturing for Pharmaceutical Supply Chains
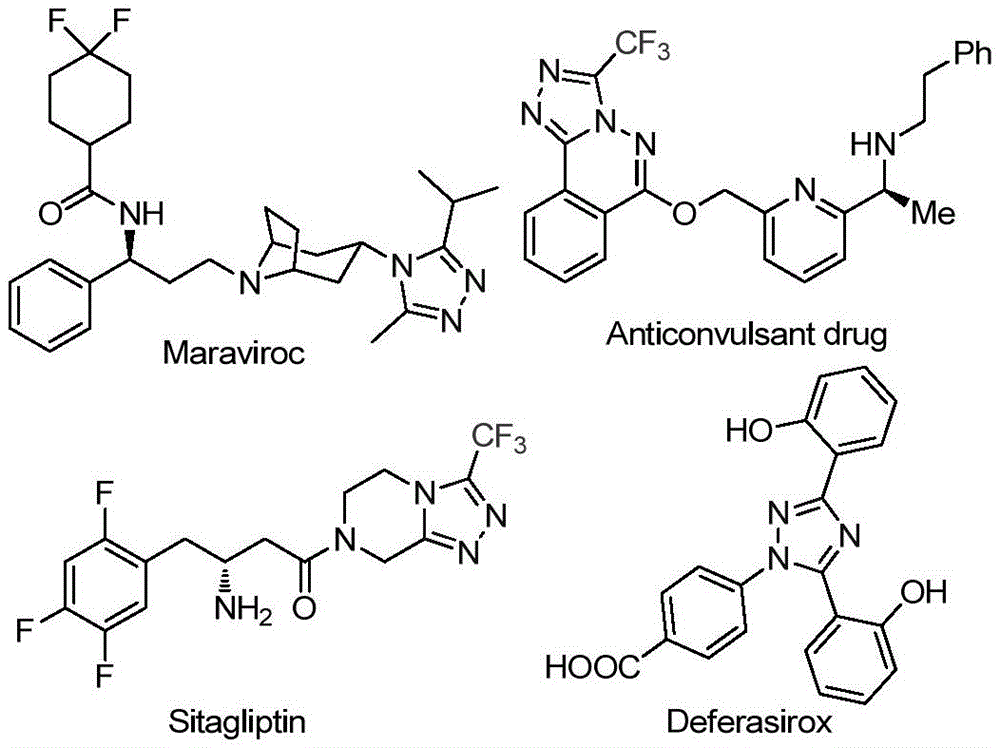
The recently granted Chinese patent CN113105402B discloses an innovative methodology for synthesizing 3,4,5-trisubstituted 1,2,4-triazole compounds, representing a significant advancement in the production of fluorinated heterocyclic intermediates critical to modern pharmaceutical development. This metal-free synthetic route addresses longstanding challenges in producing high-purity triazole derivatives that incorporate both trifluoromethyl and acyl functional groups—structural motifs found in numerous blockbuster drugs including antiviral agents and diabetes therapeutics.
Advanced Reaction Mechanism and Purity Control
The patented process begins with the iodine-mediated Kornblum oxidation of aryl ethyl ketones in dimethyl sulfoxide solvent at 90-110°C, forming aryl diketone intermediates without requiring anhydrous or oxygen-free conditions—a significant departure from conventional methods that typically demand stringent environmental controls. This initial transformation proceeds through a well-documented iodination pathway followed by oxidation, generating the key α-diketone species that subsequently react with trifluoroethylimide hydrazides under mild thermal conditions. The reaction mechanism involves sequential dehydration and cyclization steps promoted by the iodine/sodium dihydrogen phosphate/pyridine system at 120°C, which facilitates the formation of the triazole ring through intramolecular nucleophilic attack and aromatization without the need for transition metal catalysts that often complicate purification protocols.
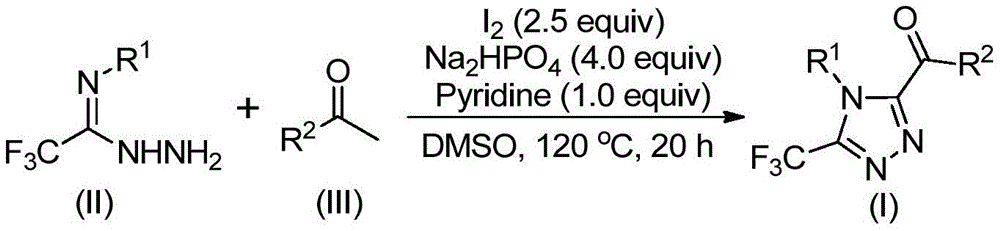
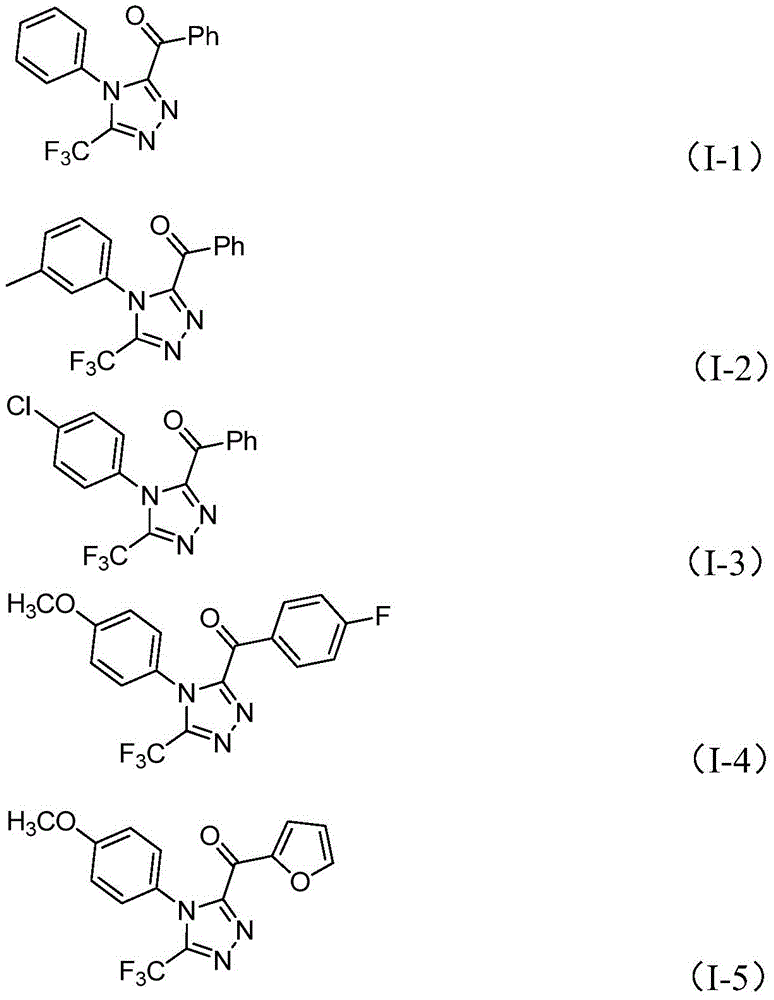
Crucially, the absence of heavy metal catalysts eliminates potential contamination pathways that typically require extensive purification steps to achieve pharmaceutical-grade purity standards. The process generates minimal byproducts due to the high selectivity of the iodine-mediated transformations, with the primary impurities being unreacted starting materials that are readily removed through standard column chromatography as demonstrated in the patent examples. This inherent selectivity is particularly valuable for producing high-purity intermediates where even trace metal residues would necessitate additional processing steps that increase both cost and timeline. The patent provides detailed NMR characterization data for multiple product variants (I-1 through I-5), confirming high purity levels achievable through the described workup procedures without requiring specialized purification equipment or techniques.
Traditional vs. Novel Triazole Synthesis Pathways
The Limitations of Conventional Methods
Traditional approaches to synthesizing multi-substituted triazoles often rely on transition metal-catalyzed cyclizations or require harsh reaction conditions that limit scalability and increase production costs. Many established routes demand strictly anhydrous and oxygen-free environments to prevent catalyst deactivation or side reactions, necessitating specialized equipment and trained personnel that significantly extend lead times for intermediate production. The use of palladium or copper catalysts in conventional methods introduces substantial challenges for pharmaceutical manufacturers who must implement rigorous metal removal protocols to meet regulatory requirements for final drug substances—processes that typically require additional processing steps while adding complexity to quality control procedures. Furthermore, existing methodologies often exhibit narrow substrate scope with poor tolerance for functional groups commonly found in complex pharmaceutical intermediates, restricting their applicability for diverse molecular architectures required in modern drug discovery programs.
The Novel Approach
The patented methodology overcomes these limitations through a carefully designed iodine-promoted cascade reaction that operates under ambient atmospheric conditions without requiring specialized handling equipment. By utilizing inexpensive and readily available iodine as the sole promoter—replacing expensive transition metals—the process eliminates both the capital investment in catalyst recovery systems and the operational costs associated with metal removal from final products. The reaction sequence demonstrates exceptional functional group tolerance as evidenced by the successful synthesis of various substituted derivatives (I-1 through I-5), including compounds containing halogens, alkoxy groups, and heteroaromatic systems that would typically interfere with metal-catalyzed processes. The patent demonstrates scalability to gram quantities with consistent product quality, indicating straightforward translation to pilot and production scales without requiring significant process re-engineering—a critical advantage for pharmaceutical manufacturers seeking reliable supply of complex intermediates.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic route addresses three critical pain points in pharmaceutical intermediate procurement: cost structure complexity, supply chain vulnerability, and scalability limitations that have historically constrained the commercial viability of fluorinated triazole compounds. By eliminating expensive catalyst systems and simplifying reaction conditions, the process creates immediate value for procurement teams seeking cost-effective solutions without compromising quality requirements essential for pharmaceutical manufacturing.
- Cost Reduction in Catalyst System: The elimination of transition metal catalysts represents a fundamental cost advantage as it removes both the raw material expense of precious metals and the downstream processing costs associated with metal removal. Traditional routes requiring palladium or copper catalysts typically incur additional expenses for specialized purification equipment and validation protocols to ensure metal residues fall below regulatory thresholds—costs that can account for significant portions of total manufacturing expenses for sensitive intermediates. The iodine-based system described in the patent utilizes commodity chemicals that are significantly less expensive than transition metal catalysts while avoiding the need for costly metal scavenging steps that extend production timelines and increase waste disposal requirements. This cost structure simplification directly translates to more competitive pricing without sacrificing quality standards required for pharmaceutical applications.
- Reduced Lead Time: The simplified reaction protocol enables faster production cycles by eliminating time-consuming setup procedures required for anhydrous/oxygen-free conditions and reducing post-reaction processing steps. Without the need for specialized glovebox environments or inert gas purging systems, manufacturing facilities can implement this process using standard equipment with minimal reconfiguration time between batches. The patent demonstrates straightforward scalability from laboratory to production scale without intermediate development phases, potentially reducing lead times compared to conventional methods that require extensive process optimization before commercial implementation. This accelerated timeline is particularly valuable for pharmaceutical companies working under tight development schedules where intermediate availability can become a critical path constraint affecting overall drug development timelines.
- Scalability Without Specialized Equipment: The process operates effectively under standard atmospheric conditions using common laboratory equipment that is readily available in most chemical manufacturing facilities worldwide. This compatibility with existing infrastructure eliminates capital expenditure requirements for new reactor systems or specialized purification units that would otherwise be necessary for implementing traditional metal-catalyzed routes. The demonstrated ability to scale directly to gram quantities with consistent product quality suggests straightforward translation to multi-kilogram production without requiring significant process re-engineering—a critical advantage for supply chain managers seeking reliable sources of complex intermediates without lengthy qualification periods. This equipment compatibility also enhances supply chain resilience by enabling multiple manufacturing sites to produce the intermediate without substantial capital investment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pharmaceutical Intermediate Supplier
While the advanced methodology detailed in patent CN113105402B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
