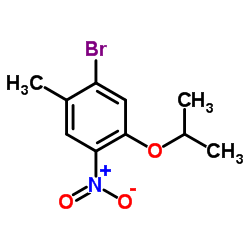
1-Bromo-5-isopropoxy-2-methyl-4-nitrobenzene
- CAS No.1202858-68-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate essential for Ceritinib synthesis. Available in bulk with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
1-Bromo-5-isopropoxy-2-methyl-4-nitrobenzene is a specialized organic compound designed for advanced pharmaceutical synthesis applications. This brominated nitrobenzene derivative serves as a critical building block in the production of kinase inhibitors, specifically functioning as a key intermediate in the manufacturing pathway of Ceritinib. Our facility produces this chemical under stringent Good Manufacturing Practice (GMP) guidelines to ensure consistent molecular integrity and batch-to-batch reproducibility.
The compound features a unique substitution pattern on the benzene ring, combining bromo, isopropoxy, methyl, and nitro functional groups. This specific arrangement facilitates selective coupling reactions required in multi-step organic synthesis. We prioritize high purity levels to minimize downstream purification burdens for our clients engaged in complex drug substance manufacturing.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | 1-Bromo-5-isopropoxy-2-methyl-4-nitrobenzene |
| CAS Registry Number | 1202858-68-1 |
| Molecular Formula | C10H12BrNO3 |
| Molecular Weight | 274.11 g/mol |
| Appearance | White crystalline powder |
| Purity (HPLC) | ≥98.0% |
| Packaging | 25 kg/drum or custom |
Industrial Applications
This intermediate is primarily utilized within the pharmaceutical sector for the synthesis of anaplastic lymphoma kinase (ALK) inhibitors. The presence of the bromine atom allows for palladium-catalyzed cross-coupling reactions, while the nitro group can be reduced to an amine for further functionalization. The isopropoxy moiety provides necessary lipophilicity and steric properties required for the final active pharmaceutical ingredient (API) bioactivity.
Our clients leverage this material for process development, clinical trial material production, and commercial-scale API manufacturing. The chemical stability of this compound ensures it withstands various reaction conditions without significant degradation, making it a reliable choice for robust synthetic routes.
Quality Assurance and Logistics
Quality control is paramount in our production process. Every batch undergoes rigorous testing using High-Performance Liquid Chromatography (HPLC), Nuclear Magnetic Resonance (NMR), and Mass Spectrometry (MS) to confirm identity and purity. We provide a comprehensive Certificate of Analysis (COA) with each shipment, detailing all relevant physicochemical properties and test results.
- Strict adherence to international safety and quality standards.
- Secure packaging to prevent moisture absorption and contamination.
- Global shipping capabilities with appropriate hazardous material documentation.
- Custom packaging solutions available upon request to suit specific workflow needs.
Storage recommendations include keeping the material in a cool, ventilated area away from direct sunlight and incompatible substances. Proper handling procedures should be followed to ensure personnel safety and environmental compliance. We are committed to supporting our partners with reliable supply chains and technical documentation to facilitate regulatory submissions.



