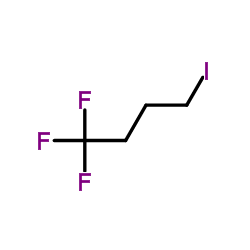
1-Iodo-4,4,4-trifluorobutane
- CAS No.461-17-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 1-iodo-4,4,4-trifluorobutane (CAS 461-17-6) is a versatile fluorinated building block widely used as an intermediate in advanced organic synthesis for pharmaceuticals, agrochemicals, and novel materials.
Request Bulk PricingProduct Technical Details
Product Overview
1-Iodo-4,4,4-trifluorobutane (CAS 461-17-6) is a high-value fluorinated alkyl halide employed extensively as a synthetic intermediate in modern organic chemistry. With its unique combination of a reactive iodine atom and a strongly electron-withdrawing trifluoromethyl group, this compound serves as a key building block for constructing complex fluorinated molecules essential in pharmaceutical R&D, agrochemical innovation, and advanced material science.
Specifications
| Molecular Formula | C₄H₆F₃I |
|---|---|
| Molecular Weight | 237.99 g/mol |
| Appearance | Clear liquid |
| Assay (GC) | ≥98.0% |
| Moisture | ≤0.1% |
| Density | 1.85 g/cm³ |
| Boiling Point | 126°C |
| Flash Point | >100°C |
| Refractive Index | 1.4326 |
Industrial Applications
This fluorinated intermediate plays a critical role in multi-step syntheses where controlled introduction of a -CF₃-containing alkyl chain is required. Its primary applications include:
- Serving as a precursor in the preparation of fluorinated heterocycles and bioactive compounds during early-stage drug discovery.
- Facilitating cross-coupling reactions (e.g., Suzuki, Negishi) to construct C–C bonds in fluorinated scaffolds.
- Acting as an alkylating agent in the development of novel agrochemicals with enhanced metabolic stability.
- Supporting the synthesis of specialty monomers for high-performance fluoropolymers.
Manufactured under stringent quality control protocols, our 1-iodo-4,4,4-trifluorobutane meets international standards for industrial-scale use. It is typically packed in 200 kg drums and should be stored in a cool, well-ventilated area away from light and moisture to maintain stability and reactivity.



