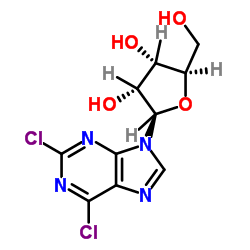
2,6-Dichloropurine-9-β-D-riboside
- CAS No.13276-52-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-quality pharmaceutical intermediate used in antiviral and antitumor drug synthesis with ≥99% purity.
Request Bulk PricingProduct Technical Details
Product Overview
2,6-Dichloropurine-9-β-D-riboside is a specialized nucleoside analog characterized by its unique purine backbone structure. This compound serves as a critical building block in the pharmaceutical industry, specifically designed for the development of advanced therapeutic agents. Our manufacturing process ensures high chemical stability and consistent quality, making it an ideal choice for complex organic synthesis pathways. The presence of chlorine atoms at the 2 and 6 positions of the purine ring provides excellent reactivity for further functionalization, allowing medicinal chemists to derive a wide range of bioactive molecules.
Technical Specifications
We adhere to strict quality control protocols to guarantee the integrity of every batch. The following table outlines the key physical and chemical parameters verified through our internal laboratory testing and third-party validation.
| Parameter | Specification |
|---|---|
| CAS Number | 13276-52-3 |
| Molecular Formula | C10H10Cl2N4O4 |
| Molecular Weight | 321.12 g/mol |
| Appearance | White or off-white crystalline powder |
| Assay (HPLC) | ≥99.0% |
| Loss on Drying | ≤1.0% |
| Residue on Ignition | ≤0.5% |
| Heavy Metals | ≤20ppm |
| Melting Point | >190ºC |
Industrial Applications
This purine derivative is primarily utilized as a key intermediate in the synthesis of nucleoside-based pharmaceuticals. Its applications span across several critical therapeutic areas, including the development of antiviral medications and antitumor agents. Researchers value this compound for its reliable performance in coupling reactions and its compatibility with various protecting group strategies. Whether used in early-stage drug discovery or scaled-up manufacturing, 2,6-Dichloropurine-9-β-D-riboside provides a robust foundation for creating next-generation medicines. It is particularly effective in pathways requiring precise stereochemistry and high regioselectivity.
Quality Assurance and Storage
Our facility operates under rigorous quality management systems to ensure product safety and consistency. Each shipment is accompanied by a Certificate of Analysis (COA) detailing batch-specific test results. To maintain optimal stability, the product should be stored in a cool, ventilated environment away from direct sunlight and moisture. We offer flexible packaging options, including standard 25 kg drums, to accommodate both laboratory-scale research and large-scale industrial production needs. Our global logistics network ensures timely delivery while maintaining the integrity of the chemical structure during transit.



