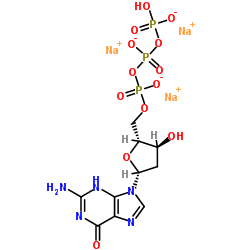
2'-Deoxyguanosine-5'-triphosphate trisodium salt
- CAS No.93919-41-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity 2'-Deoxyguanosine-5'-triphosphate trisodium salt for molecular biology and pharmaceutical research.
Request Bulk PricingProduct Technical Details
Product Overview
2'-Deoxyguanosine-5'-triphosphate trisodium salt is a high-quality nucleotide derivative essential for various biochemical and pharmaceutical applications. As a fundamental building block for DNA synthesis, this compound plays a critical role in molecular biology research, enzymatic reactions, and the development of advanced therapeutic intermediates. Our manufacturing process ensures exceptional stability and purity, making it an ideal choice for laboratories and industrial facilities requiring reliable reagents.
We specialize in the production of complex pharmaceutical intermediates and biochemical reagents. This specific trisodium salt form offers enhanced solubility and stability compared to free acid forms, facilitating easier handling in aqueous solutions. Each batch undergoes rigorous quality control testing to meet international standards for research and development purposes.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | 2'-Deoxyguanosine-5'-triphosphate trisodium salt |
| CAS Number | 93919-41-6 |
| Molecular Formula | C10H13N5Na3O13P3 |
| Molecular Weight | 573.13 g/mol |
| Appearance | White to off-white crystalline powder |
| Purity (HPLC) | ≥98.0% |
| Storage Conditions | Cool, ventilated place away from moisture |
Industrial Applications
This nucleotide is widely utilized in the field of molecular biology and genetic engineering. It serves as a crucial substrate for DNA polymerases during polymerase chain reaction (PCR) processes, DNA sequencing, and in vitro transcription assays. Researchers rely on its high purity to ensure accurate results in genomic studies and diagnostic development.
In the pharmaceutical sector, this compound acts as a key intermediate for synthesizing nucleoside analogs and antiviral agents. Its consistent quality supports the development of new drugs targeting viral infections and genetic disorders. Our facility adheres to strict manufacturing protocols to maintain batch-to-batch consistency, which is vital for large-scale production needs.
Quality Assurance and Packaging
Quality is our top priority. Every production lot is tested using high-performance liquid chromatography (HPLC) to verify purity levels. We provide comprehensive documentation, including Certificates of Analysis (COA), to ensure transparency and compliance with client requirements. Our quality management system is designed to meet global standards for chemical manufacturing.
- Advanced purification techniques ensure minimal impurities.
- Stable packaging protects product integrity during transit.
- Customized packaging options available upon request.
- Fast delivery services for global customers.
Standard packaging includes 25 kg drums, but we offer flexible solutions to accommodate specific client needs. Proper storage in a cool and ventilated environment is recommended to maintain optimal chemical stability over extended periods. Contact our sales team for detailed technical data sheets and bulk pricing information.



