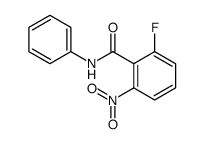
2-Fluoro-6-nitro-N-phenylbenzamide
- CAS No.870281-83-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate essential for the synthesis of Idelalisib. Available in bulk quantities with comprehensive quality documentation and global logistics support.
Request Bulk PricingProduct Technical Details
Product Overview
2-Fluoro-6-nitro-N-phenylbenzamide represents a critical specialized building block within the realm of modern pharmaceutical chemistry. Designed with precision for complex organic synthesis, this compound serves as a vital precursor in the manufacturing of advanced kinase inhibitors. Our facility produces this intermediate under strict quality control protocols to ensure consistency and reliability for downstream drug development processes. The chemical structure incorporates specific functional groups that facilitate efficient coupling reactions required for high-value therapeutic agents.
As a dedicated manufacturer, we understand the stringent requirements of the pharmaceutical supply chain. This product is engineered to meet the rigorous demands of medicinal chemists and process development teams. The presence of both fluoro and nitro substituents on the benzamide scaffold provides unique reactivity profiles, making it an indispensable component for constructing specific molecular architectures found in contemporary oncology treatments.
Technical Specifications
We maintain transparent and detailed specifications to guarantee product integrity. Each batch undergoes comprehensive analytical testing using state-of-the-art instrumentation including HPLC and NMR. The following table outlines the key physical and chemical parameters that define our standard commercial grade material.
| Parameter | Specification |
|---|---|
| CAS Number | 870281-83-7 |
| Molecular Formula | C13H9FN2O3 |
| Molecular Weight | 260.22 g/mol |
| Appearance | Light yellow powder |
| Assay (Purity) | ≥98.0% |
| Packing | 25 kg/drum or customized |
Industrial Applications
The primary application of this compound lies in the synthesis of Idelalisib, a potent phosphatidylinositol 3-kinase (PI3K) inhibitor. This therapeutic agent is utilized in the treatment of certain hematological malignancies. Consequently, the quality of this intermediate directly impacts the efficacy and safety profile of the final active pharmaceutical ingredient. Our manufacturing process is optimized to minimize impurities that could complicate subsequent purification steps.
Beyond its specific role in Idelalisib production, this benzamide derivative serves as a valuable research tool for exploring structure-activity relationships in kinase inhibitor development. Pharmaceutical companies utilize this building block to prototype new candidates targeting similar biological pathways. The stability of the nitro and fluoro groups under various reaction conditions allows for versatile synthetic routes, providing chemists with flexibility during process optimization.
Quality Assurance and Packaging
Quality is the cornerstone of our operation. Every production lot is accompanied by a Certificate of Analysis (COA) detailing test results against established specifications. We adhere to international safety standards for handling and shipping fine chemicals. Our quality assurance team conducts random sampling and retention testing to ensure long-term stability and performance consistency.
- Strict inbound raw material inspection to prevent contamination.
- In-process controls monitoring reaction progress and purity levels.
- Final product verification using multiple analytical methods.
- Secure packaging to protect against moisture and light exposure.
Standard packaging consists of 25 kg fiber drums lined with polyethylene bags, ensuring protection during transit. Customized packaging solutions are available upon request to accommodate specific logistical needs or smaller scale research requirements. We recommend storing the material in a cool, ventilated area away from direct sunlight to maintain optimal chemical stability over extended periods.



