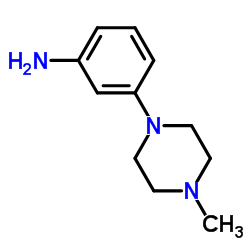
3-(4-Methylpiperazin-1-yl)aniline
- CAS No.148546-99-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate used in organic synthesis. Light grey powder with ≥98% purity.
Request Bulk PricingProduct Technical Details
Product Overview
3-(4-Methylpiperazin-1-yl)aniline represents a sophisticated heterocyclic building block extensively utilized within the realm of modern pharmaceutical research and organic synthesis. Characterized by its unique combination of an aniline moiety and a methylated piperazine ring, this compound serves as a critical precursor for the development of various therapeutic agents. Our facility produces this intermediate under strict quality control protocols to ensure consistency and reliability for large-scale manufacturing processes. The structural integrity of the piperazine ring offers enhanced solubility and bioavailability potential when incorporated into final drug candidates, making it a preferred choice for medicinal chemists.
The physical properties of this chemical make it suitable for a wide range of reaction conditions. Typically appearing as a light grey powder, it exhibits stable thermal characteristics with a defined melting point range. The high purity level ensures minimal side reactions during subsequent synthetic steps, which is vital for optimizing yield and reducing downstream purification costs. Density and boiling point data indicate robust stability under standard processing conditions, facilitating safe handling in industrial environments.
Technical Specifications
| Items of Analysis | Standard of Analysis | Test Results |
|---|---|---|
| Appearance | Light grey powder | Conform |
| Purity | ≥98% | 98.73% |
| Melting point | 97-100℃ | 98-99℃ |
| Conclusion | Conforms to Factory Standard | |
Applications in Synthesis
In terms of applications, this piperazine derivative is frequently employed in the construction of kinase inhibitors and other small molecule drugs targeting neurological and oncological pathways. The nucleophilic nature of the amine groups allows for versatile functionalization, enabling chemists to introduce complex structural motifs efficiently. This versatility supports rapid structure-activity relationship (SAR) studies during the early stages of drug discovery.
- Used as a key intermediate in pharmaceutical API synthesis.
- Facilitates the construction of complex heterocyclic systems.
- Compatible with various organic synthesis routes and solvents.
- Supports scale-up from laboratory to industrial production.
Quality Assurance and Storage
From an industrial perspective, the manufacturing process involves advanced organic synthesis techniques to minimize impurities and maximize yield. Our commitment to environmental safety ensures that production methods adhere to international regulatory standards. This focus on sustainable chemistry aligns with the growing demand for responsible sourcing in the global pharmaceutical industry.
Quality Assurance and Storage: We provide comprehensive documentation including Certificates of Analysis (COA) for every batch shipped. Each lot undergoes rigorous testing against internal factory standards to verify purity and identity. Proper storage in a cool, dry place away from direct sunlight and moisture is recommended to maintain long-term stability. Personnel should handle this compound using standard laboratory safety equipment, including gloves and eye protection, to ensure occupational safety.
Furthermore, the chemical stability of 3-(4-Methylpiperazin-1-yl)aniline allows for extended shelf life when stored correctly, reducing waste and improving cost-efficiency for our partners. The compound's compatibility with various solvents makes it adaptable to different synthetic routes, whether in solution-phase or solid-phase synthesis strategies. This flexibility is particularly valuable in process chemistry where scaling up from milligram to kilogram quantities requires robust and forgiving reaction conditions.
Our technical support team is available to assist with customization requests or specific purity requirements tailored to your project needs. By partnering with us, clients gain access to decades of expertise in chemical manufacturing and a dedication to quality that supports your innovation goals. Key advantages of sourcing this material from our established supply chain include consistent batch-to-batch reproducibility, competitive bulk pricing, and timely global logistics. We understand the critical nature of supply continuity in pharmaceutical manufacturing and maintain significant inventory levels to meet urgent production demands.



