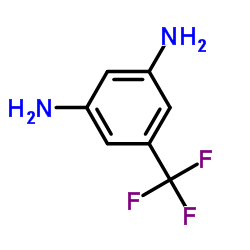
3,5-Diaminobenzotrifluoride
- CAS No.368-53-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 3,5-Diaminobenzotrifluoride (CAS 368-53-6), a key fluorinated aromatic diamine intermediate for advanced pharmaceutical and agrochemical synthesis.
Request Bulk PricingProduct Technical Details
Product Overview
3,5-Diaminobenzotrifluoride (CAS 368-53-6) is a high-value fluorinated aromatic diamine widely employed as a critical building block in the synthesis of complex active pharmaceutical ingredients (APIs) and specialty agrochemicals. Its unique molecular architecture—featuring two amino groups in a meta configuration relative to a strongly electron-withdrawing trifluoromethyl group—imparts exceptional reactivity and selectivity in heterocyclic and cross-coupling reactions.
Specifications
| Molecular Formula | C₇H₇F₃N₂ |
|---|---|
| Molecular Weight | 176.14 g/mol |
| Purity | ≥98.0% |
| Appearance | Solid |
| Melting Point | 87–89 °C (lit.) |
| Boiling Point | 275 °C at 760 mmHg |
| Density | 1.381 g/cm³ |
| Flash Point | 122.5 °C |
| Refractive Index | 1.54 |
| Storage | Stable at room temperature in closed containers under normal storage and handling conditions. |
| Packaging | 200 kg per drum |
Industrial Applications
This compound serves primarily as a versatile intermediate in multi-step organic syntheses within the pharmaceutical and fine chemical industries. Its dual amine functionality enables sequential derivatization, while the trifluoromethyl group enhances metabolic stability and lipophilicity in downstream molecules—key attributes in modern drug design.
- Ideal for constructing nitrogen-containing heterocycles such as triazoles, benzimidazoles, and quinazolines.
- Used in the development of kinase inhibitors, anti-inflammatory agents, and antimicrobial scaffolds.
- Compatible with standard coupling protocols including Buchwald-Hartwig amination and Ullmann-type reactions.
As a non-controlled, non-hazardous (under normal use) specialty intermediate, 3,5-Diaminobenzotrifluoride aligns with global regulatory standards for industrial chemical supply chains. We provide comprehensive documentation including Certificate of Analysis (COA), safety data sheets, and batch-specific quality reports to support seamless integration into GMP-compliant manufacturing workflows.



