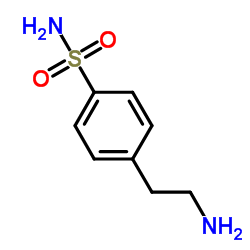
4-(2-Aminoethyl)benzenesulfonamide
- CAS No.35303-76-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 4-(2-Aminoethyl)benzenesulfonamide is a critical pharmaceutical intermediate used primarily in the synthesis of antidiabetic agents like Glipizide.
Request Bulk PricingProduct Technical Details
Product Overview
4-(2-Aminoethyl)benzenesulfonamide represents a sophisticated organic compound essential to modern pharmaceutical manufacturing. Identified by CAS number 35303-76-5, this chemical serves as a foundational building block in the production of sulfonylurea derivatives. Our facility specializes in the large-scale synthesis of this intermediate, ensuring consistent quality and reliability for global research and development teams. The compound is characterized by its stable chemical structure and high reactivity, making it ideal for complex organic synthesis pathways.
We understand the critical nature of supply chain consistency in the pharmaceutical industry. Therefore, our production processes are designed to maintain strict batch-to-batch uniformity. Each lot undergoes rigorous analytical testing to verify identity, purity, and physical properties. This commitment to quality ensures that downstream manufacturers can rely on our material for their own synthesis protocols without compromising final product integrity.
Key Specifications
The following table outlines the standard technical specifications for our premium grade material. These parameters are verified using advanced analytical instrumentation including HPLC and spectroscopic methods.
| Parameter | Specification |
|---|---|
| CAS Number | 35303-76-5 |
| Molecular Formula | C8H12N2O2S |
| Molecular Weight | 200.26 g/mol |
| Appearance | White or off-white powder |
| Purity (HPLC) | ≥99.5% |
| Melting Point | 146°C - 153°C |
| Loss on Drying | ≤0.5% |
| Residue on Ignition | ≤0.12% |
Industrial Applications
This sulfonamide derivative is predominantly utilized as a key intermediate in the pharmaceutical sector. Its primary application lies in the synthesis of antidiabetic medications, specifically serving as a precursor for Glipizide. The chemical structure allows for efficient functionalization, enabling chemists to construct complex molecular architectures required for therapeutic efficacy. Beyond pharmaceuticals, this compound may also find utility in agrochemical research and fine chemical synthesis where sulfonamide motifs are required.
Our technical team supports clients with detailed documentation regarding safe handling and integration into existing synthesis routes. We provide comprehensive data on solubility profiles and reaction conditions to facilitate smooth scale-up processes. Whether for pilot plant operations or commercial manufacturing, our material is engineered to meet demanding performance benchmarks.
Quality Assurance and Packaging
Quality control is the cornerstone of our manufacturing operation. Every batch is accompanied by a Certificate of Analysis (COA) detailing test results against internal and pharmacopoeial standards. We maintain full traceability from raw material sourcing to final dispatch. Our quality assurance protocols include testing for related substances, heavy metals, and residual solvents to ensure safety and compliance.
- Standard packaging includes 25kg cardboard drums.
- Custom packaging solutions available upon request.
- Stored in cool, dry, well-closed containers.
- Protected from moisture and strong light during transit.
We offer global shipping capabilities with appropriate hazard classification and documentation. Our logistics partners ensure timely delivery while maintaining product stability. For large volume requirements, we provide competitive bulk pricing structures tailored to long-term partnerships. Contact our sales team today to request samples or discuss your specific procurement needs.



